Abstract
Background
The purpose of this study was to investigate the effect of recombinant pigment epithelium-derived factor (rPEDF), secreted by ARPE-19 cells transfected with the human PEDF gene and transplanted subconjunctivally in normal and in rabbits in which corneal neovascularization was elicited by a chemical burn.
Methods
Twenty grey Chinchilla Bastard rabbits were randomly assigned to four groups; neovascularization was induced in groups A, B, and C by alkali cauterization. Seven days later, group A received no cell implantation, non-transfected ARPE-19 cells were implanted subconjunctivally in group B, and PEDF-transfected ARPE-19 cells were implanted subconjunctivally in groups C and D (non-cauterized). In-vivo rPEDF secretion was analyzed by immunoblotting, and ELISA of extracts of conjunctival tissue samples taken at different time points. Digital photographs acquired on days 7, 14, and 21 after cauterization were evaluated for lead vessel length, vascular invasion area, and overall neovascularization rate.
Results
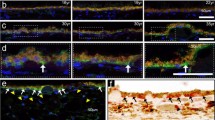
At days 14 and 21 after cauterization, significant differences were observed between groups A, B, and C in lead vessel length (day 21: 5.91 ± 0.45, 5.11 ± 1.22, 3.79 ± 0.59 mm, repectively), vascular invasion area (day 21: 35.5 ± 8.65, 34.86 ± 4.92, 19.2 ± 5.03 mm2 respectively), and rate of corneal neovascularization. Compared to controls, neovascularization was reduced by 37.5 % on day 14 and 47 % on day 21. Analysis of conjunctival tissue extracts showed that rPEDF was secreted by the transplanted PEDF-transfected cells.
Conclusion
Subconjunctivally transplanted, PEDF-transfected ARPE-19 cells secrete rPEDF, which inhibits the corneal neovascularization elicited by alkali cauterization.




Similar content being viewed by others
References
Cursiefen C, Wenkel H, Martus P et al (2001) Impact of short-term versus long-term topical steroids on corneal neovascularization after non-high-risk keratoplasty. Graefes Arch Clin Exp Ophthalmol 239:514–521
Qazi YWG, Monson B, Jack Stringham J, Ambati BK (2010) Corneal transparency: genesis, maintenance and dysfunction. Brain Res Bull 25:98–210
Chakravarti S, Magnuson T, Lass JH, Jepsen KJ, LaMantia C, Carroll H (1998) Lumican regulates collagen fibril assembly: skin fragility and corneal opacity in the absence of lumican. J Cell Biol 141(5):1277–1286
Chang JH, Gabison EE, Kato T, Azar DT (2001) Corneal neovascularization. Curr Opin Ophthalmol 12(4):242–249
Price MO, Thompson RW Jr, Price FW Jr (2003) Risk factors for various causes of failure in initial corneal grafts. Arch Ophthalmol 121(8):1087–1092. doi:10.1001/archopht.121.8.1087
Cursiefen C, Cao J, Chen L, Liu Y, Maruyama K, Jackson D, Kruse F, Wiegand S, Dana M, Streilein J (2004) Inhibition of hemangiogenesis and lymphangiogenesis after normal-risk corneal transplantation by neutralizing VEGF promotes graft survival. Invest Ophthalmol Vis Sci 45(8):2666–2673
Chen WL, Lin CT, Lin NT, Tu IH, Li JW, Chow LP, Liu KR, Hu FR (2009) Subconjunctival injection of bevacizumab (Avastin) on corneal neovascularization in different rabbit models of corneal angiogenesis. Invest Ophthalmol Vis Sci 50(4):1659–1665. doi:10.1167/iovs. 08-1997
Bollinger KE, Smith SD (2009) Prevalence and management of elevated intraocular pressure after placement of an intravitreal sustained-release steroid implant. Curr Opin Ophthalmol 20(2):99–103
Hashemian MN, Moghimi S, Kiumehr S, Riazi M, Amoli FA (2009) Prevention and treatment of corneal neovascularization: comparison of different doses of subconjunctival bevacizumab with corticosteroid in experimental rats. Ophthalmic Res 42(2):90–95. doi:10.1159/000224783
Jaffe GJ, McCallum RM, Branchaud B, Skalak C, Butuner Z, Ashton P (2005) Long-term follow-up results of a pilot trial of a fluocinolone acetonide implant to treat posterior uveitis. Ophthalmology 112(7):1192–1198. doi:10.1016/j.ophtha.2005.03.013
Urban RC Jr, Cotlier E (1986) Corticosteroid-induced cataracts. Surv Ophthalmol 31(2):102–110. doi:10.1016/0039-6257(86)90077-9
Mastyugin V, Mosaed S, Bonazzi A, Dunn MW, Schwartzman ML (2001) Corneal epithelial VEGF and cytochrome P450 4B1 expression in a rabbit model of closed eye contact lens wear. Curr Eye Res 23(1):1–10
Kvanta A, Sarman S, Fagerholm P, Seregard S, Steen B (2000) Expression of matrix metalloproteinase-2 (MMP-2) and vascular endothelial growth factor (VEGF) in inflammation-associated corneal neovascularization. Exp Eye Res 70(4):419–428. doi:10.1006/exer.1999.0790
Amano S, Rohan R, Kuroki M, Tolentino M, Adamis AP (1998) Requirement for vascular endothelial growth factor in wound- and inflammation-related corneal neovascularization. Invest Ophthalmol Vis Sci 39(1):18–22
Qian CXBI, Levinger E, Rootman D (2008) Combined use of superficial keratectomy and subconjunctival bevacizumab injection for corneal neovascularization. Cornea 27:1090–1092
Perez-Santonja JJ, Campos-Mollo E, Lledo-Riquelme M, Javaloy J, Alio JL (2010) Inhibition of corneal neovascularization by topical bevacizumab (anti-VEGF) and sunitinib (anti-VEGF and anti-PDGF) in an animal model. Am J Ophthalmol 150(4):519–528. doi:10.1016/j.ajo.2010.04.024, e511
Papathanassiou M, Theodossiadis PG, Liarakos VS, Rouvas A, Giamarellos-Bourboulis EJ, Vergados IA (2008) Inhibition of corneal neovascularization by subconjunctival bevacizumab in an animal model. Am J Ophthalmol 145(3):424–431. doi:10.1016/j.ajo.2007.11.003
Kim WJ, Jeong HO, Chung SK (2010) The effect of bevacizumab on corneal neovascularization in rabbits. Korean J Ophthalmol 24(4):230–236. doi:10.3341/kjo.2010.24.4.230
Bock F, Onderka J, Dietrich T, Bachmann B, Kruse FE, Paschke M, Zahn G, Cursiefen C (2007) Bevacizumab as a potent inhibitor of inflammatory corneal angiogenesis and lymphangiogenesis. Invest Ophthalmol Vis Sci 48(6):2545–2552. doi:10.1167/iovs. 06-0570
Gerber HP, Wu X, Yu L, Wiesmann C, Liang XH, Lee CV, Fuh G, Olsson C, Damico L, Xie D, Meng YG, Gutierrez J, Corpuz R, Li B, Hall L, Rangell L, Ferrando R, Lowman H, Peale F, Ferrara N (2007) Mice expressing a humanized form of VEGF-A may provide insights into the safety and efficacy of anti-VEGF antibodies. Proc Natl Acad Sci U S A 104(9):3478–3483. doi:10.1073/pnas.0611492104
van der Flier M, Coenjaerts FE, Mwinzi PN, Rijkers E, Ruyken M, Scharringa J, Kimpen JL, Hoepelman AI, Geelen SP (2005) Antibody neutralization of vascular endothelial growth factor (VEGF) fails to attenuate vascular permeability and brain edema in experimental pneumococcal meningitis. J Neuroimmunol 160(1–2):170–177. doi:10.1016/j.jneuroim.2004.11.013
Dastjerdi MH, Al-Arfaj KM, Nallasamy N, Hamrah P, Jurkunas UV, Pineda R 2nd, Pavan-Langston D, Dana R (2009) Topical bevacizumab in the treatment of corneal neovascularization: results of a prospective, open-label, noncomparative study. Arch Ophthalmol 127(4):381–389. doi:10.1001/archophthalmol.2009.18
Bock F, Konig Y, Kruse F, Baier M, Cursiefen C (2008) Bevacizumab (Avastin) eye drops inhibit corneal neovascularization. Graefes Arch Clin Exp Ophthalmol 246(2):281–284. doi:10.1007/s00417-007-0684-4
Petsoglou C, Balaggan KS, Dart JK, Bunce C, Xing W, Ali RR, Tuft SJ (2013) Subconjunctival bevacizumab induces regression of corneal neovascularisation: a pilot randomised placebo-controlled double-masked trial. Br J Ophthalmol 97(1):28–32. doi:10.1136/bjophthalmol-2012-302137
Gueudry J, Richez F, Tougeron-Brousseau B, Genevois O, Muraine M (2010) Subconjunctival bevacizumab for corneal neovascularization. J Fr Ophtalmol 33(9):630–636. doi:10.1016/j.jfo.2010.07.007
Fallah MR, Khosravi K, Hashemian MN, Beheshtnezhad AH, Rajabi MT, Gohari M (2010) Efficacy of topical bevacizumab for inhibiting growth of impending recurrent pterygium. Curr Eye Res 35(1):17–22. doi:10.3109/02713680903395273
Chu HS, Hu FR, Yang CM, Yeh PT, Chen YM, Hou YC, Chen WL (2011) Subconjunctival injection of bevacizumab in the treatment of corneal neovascularization associated with lipid deposition. Cornea 30(1):60–66. doi:10.1097/ICO.0b013e3181e458c5
Bayar SA, Altinors DD, Kucukerdonmez C, Akova YA (2010) Severe corneal changes following intravitreal injection of bevacizumab. Ocul Immunol Inflamm 18(4):268–274. doi:10.3109/09273948.2010.490630
Galor A, Yoo SH (2010) Corneal melt while using topical bevacizumab eye drops. Ophthalmic Surg Lasers Imaging 9:1-3. doi:10.3928/15428877-20100215-07
Kim TI, Chung JL, Hong JP, Min K, Seo KY, Kim EK (2009) Bevacizumab application delays epithelial healing in rabbit cornea. Invest Ophthalmol Vis Sci 50(10):4653–4659. doi:10.1167/iovs. 08-2805
Rosenstein JM, Krum JM (2004) New roles for VEGF in nervous tissue–beyond blood vessels. Exp Neurol 187(2):246–253. doi:10.1016/j.expneurol.2004.01.022
Yu CQ, Zhang M, Matis KI, Kim C, Rosenblatt MI (2008) Vascular endothelial growth factor mediates corneal nerve repair. Invest Ophthalmol Vis Sci 49(9):3870–3878. doi:10.1167/iovs. 07-1418
Bock F, Onderka J, Rummelt C, Dietrich T, Bachmann B, Kruse FE, Schlotzer-Schrehardt U, Cursiefen C (2009) Safety profile of topical VEGF neutralization at the cornea. Invest Ophthalmol Vis Sci 50(5):2095–2102. doi:10.1167/iovs. 07-1129
Scappaticci FA, Skillings JR, Holden SN, Gerber HP, Miller K, Kabbinavar F, Bergsland E, Ngai J, Holmgren E, Wang J, Hurwitz H (2007) Arterial thromboembolic events in patients with metastatic carcinoma treated with chemotherapy and bevacizumab. J Natl Cancer Inst 99(16):1232–1239. doi:10.1093/jnci/djm086
Cannistra SA, Matulonis UA, Penson RT, Hambleton J, Dupont J, Mackey H, Douglas J, Burger RA, Armstrong D, Wenham R, McGuire W (2007) Phase II study of bevacizumab in patients with platinum-resistant ovarian cancer or peritoneal serous cancer. J Clin Oncol 25(33):5180–5186. doi:10.1200/JCO.2007.12.0782
Dawson DW, Volpert OV, Gillis P, Crawford SE, Xu H, Benedict W, Bouck NP (1999) Pigment epithelium-derived factor: a potent inhibitor of angiogenesis. Science 285(5425):245–248
Cursiefen C, Colin J, Dana R, Diaz-Llopis M, Faraj LA, Garcia-Delpech S, Geerling G, Price FW, Remeijer L, Rouse BT, Seitz B, Udaondo P, Meller D, Dua H (2012) Consensus statement on indications for anti-angiogenic therapy in the management of corneal diseases associated with neovascularisation: outcome of an expert roundtable. Br J Ophthalmol 96(1):3–9. doi:10.1136/bjo.2011.204701
Ambati BK, Nozaki M, Singh N, Takeda A, Jani PD, Suthar T, Albuquerque RJ, Richter E, Sakurai E, Newcomb MT, Kleinman ME, Caldwell RB, Lin Q, Ogura Y, Orecchia A, Samuelson DA, Agnew DW, St Leger J, Green WR, Mahasreshti PJ, Curiel DT, Kwan D, Marsh H, Ikeda S, Leiper LJ, Collinson JM, Bogdanovich S, Khurana TS, Shibuya M, Baldwin ME, Ferrara N, Gerber HP, De Falco S, Witta J, Baffi JZ, Raisler BJ, Ambati J (2006) Corneal avascularity is due to soluble VEGF receptor-1. Nature 443(7114):993–997. doi:10.1038/nature05249
Mirochnik Y, Aurora A, Schulze-Hoepfner FT, Deabes A, Shifrin V, Beckmann R, Polsky C, Volpert OV (2009) Short pigment epithelial-derived factor-derived peptide inhibits angiogenesis and tumor growth. Clin Cancer Res 15(5):1655–1663. doi:10.1158/1078-0432.CCR-08-2113
Zhang SX, Wang JJ, Gao G, Parke K, Ma JX (2006) Pigment epithelium-derived factor downregulates vascular endothelial growth factor (VEGF) expression and inhibits VEGF-VEGF receptor 2 binding in diabetic retinopathy. J Mol Endocrinol 37(1):1–12. doi:10.1677/jme.1.02008
Cai J, Jiang WG, Grant MB, Boulton M (2006) Pigment epithelium-derived factor inhibits angiogenesis via regulated intracellular proteolysis of vascular endothelial growth factor receptor 1. J Biol Chem 281(6):3604–3613. doi:10.1074/jbc.M507401200
Ogata N, Wada M, Otsuji T, Jo N, Tombran-Tink J, Matsumura M (2002) Expression of pigment epithelium-derived factor in normal adult Rat Eye and experimental choroidal neovascularization. Invest Ophthalmol Vis Sci 43:1168–1175
Petros C, Karakousis PC, John SK et al (2001) Localization of epithelium derived factor (PEDF) in developing and adult human ocular tissues. Mol Vis 7:154–163
Kuo CN, Yang LC, Yang CT, Lai CH, Chen MF, Chen CY, Chen CH, Wu PC, Kou HK, Chen YJ, Hung CH, Tsai CB (2009) Inhibition of corneal neovascularization with plasmid pigment epithelium-derived factor (p-PEDF) delivered by synthetic amphiphile INTeraction-18 (SAINT-18) vector in an experimental model of rat corneal angiogenesis. Exp Eye Res 89(5):678–685. doi:10.1016/j.exer.2009.06.021
Shao C, Sima J, Zhang SX, Jin J, Reinach P, Wang Z, Ma JX (2004) Suppression of corneal neovascularization by PEDF release from human amniotic membranes. Invest Ophthalmol Vis Sci 45(6):1758–1762
Jin J, Ma JX, Guan M, Yao K (2010) Inhibition of chemical cautery-induced corneal neovascularization by topical pigment epithelium-derived factor eyedrops. Cornea 29:1055–1061
Kuo CN, Chen CY, Chen SN, Yang LC, Lai LJ, Lai CH, Chen MF, Hung CH, Chen CH (2013) Inhibition of corneal neovascularization with the combination of bevacizumab and plasmid pigment epithelium-derived factor-synthetic amphiphile INTeraction-18 (p-PEDF-SAINT-18) vector in a Rat corneal experimental angiogenesis model. Int J Mol Sci 14(4):8291–8305. doi:10.3390/ijms14048291
Bai YJ, Huang LZ, Xu XL, Du W, Zhou AY, Yu WZ, Li XX (2012) Polyethylene glycol-modified pigment epithelial-derived factor: new prospects for treatment of retinal neovascularization. J Pharmacol Exp Ther 342(1):131–139. doi:10.1124/jpet.112.192575
Johnen S, Izsvak Z, Stocker M, Harmening N, Salz AK, Walter P, Thumann G (2012) Sleeping beauty transposon-mediated transfection of retinal and iris pigment epithelial cells. Invest Ophthalmol Vis Sci 53(8):4787–4796. doi:10.1167/iovs. 12-9951
Thumann G, Stöcker M, Maltusch C, Salz AK, Barth S, Walter P, Johnen S (2010) High efficiency non-viral transfection of retinal and iris pigment epithelial cells with pigment epithelium-derived factor. Gene Ther 17:181–189
Bahar I, Kaiserman I, McAllum P, Rootman D, Slomovic A (2008) Subconjunctival bevacizumab injection for corneal neovascularization. Cornea 27(2):142–147. doi:10.1097/ICO.0b013e318159019f
Olleveant NA, Humphris G, Roe B (1999) How big is a drop? A volumetric assay of essential oils. J Clin Nurs 8(3):299–304
Tomanek RJ, Schatteman GC (2000) Angiogenesis: new insights and therapeutic potential. Anat Rec 261(3):126–135. doi:10.1002/1097-0185(20000615)261:3<126::AID-AR7>3.0.CO;2-4
Grisanti S, Ishioka M, Kosiewicz M, Jiang LQ (1997) Immunity and immune privilege elicited by cultured retinal pigment epithelial cell transplants. Invest Ophthalmol Vis Sci 38(8):1619–1626
Acknowledgments
We would like to thank Claudia Etzkorn and Anna Dobias for their unremitting help in the experiments.
We would furthermore express our gratitude to Prof. N. Sorgente for his help in proof-reading this manuscript and for all the advice he has given us.
Conflict of Interest
All authors certify that they have NO affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kuerten, D., Johnen, S., Harmening, N. et al. Transplantation of PEDF-transfected pigment epithelial cells inhibits corneal neovascularization in a rabbit model. Graefes Arch Clin Exp Ophthalmol 253, 1061–1069 (2015). https://doi.org/10.1007/s00417-015-2954-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-015-2954-x