Abstract
Purpose
To describe characteristics of epiretinal cells at the vitreoretinal interface by correlative light and electron microscopy (CLEM).
Methods
Epiretinal membrane (ERM) specimens and internal limiting membrane (ILM) specimens were harvested by sequential peeling during vitrectomy from 27 eyes with idiopathic epiretinal gliosis, and processed for CLEM. Intraoperatively, the presence of posterior vitreous detachment (PVD) was documented. We used anti-vimentin, anti-α-smooth muscle actin (α-SMA), and anti-CD45 as primary antibodies. A fluorescein-tagged immunonanogold cluster was used as secondary antibody and visualized under the fluorescence and transmission electron microscope.
Results
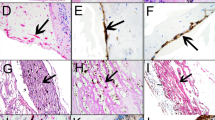
We demonstrated CD45-positive cells specifically labelled at their plasma membranes with ultrastructural features known for hyalocytes, such as oval nucleus with marginal chromatin, vacuoles, dense granules, and thin cytoplasmic protrusions. CD45-positive cells were mostly located on a thick layer of native vitreous collagen. They were covered by newly formed collagen strands with multilayered proliferation of myofibroblasts. We also demonstrated immunoreactivity for vimentin and alpha-SMA. Cell fragments with positive labelling for α-SMA and vimentin were not only found on the vitreal side of the ILM, but also on the retinal side.
Conclusions
By CLEM, the majority of CD45-positive cells in epiretinal cell proliferation were characterized as hyalocytes. In the context of anomalous PVD and vitreoschisis, ultrastructural features and topographic localization of hyalocytes suggest that these cells play a significant role in ERM formation. CLEM enables a more accurate characterization of epiretinal cell proliferation, and therefore, contributes to a better understanding of the pathogenesis of diseases at the vitreoretinal interface.




Similar content being viewed by others
References
Bochaton-Piallat ML, Kapetanios AD, Donati G, Redard M, Gabbiani G, Pournaras CJ (2000) TGF-ß1, TGF-ß receptor II and ED-A Fibronectin expression in myofibroblast of vitreoretinopathy. Invest Ophthalmol Vis Sci 41:2336–2342
Gandorfer A, Rohleder M, Kampik A (2002) Epiretinal pathology of vitreomacular traction syndrome. Br J Ophthalmol 86:902–909
Kampik A, Green WR, Michels RG, Nase PK (1980) Ultrastructural features of progressive idiopathic epiretinal membrane removed by vitreous surgery. Am J Ophthalmol 90:797–809
Kampik A, Kenyon KR, Michels RG, Green WR, de la Cruz ZC (1981) Epiretinal and vitreous membranes. Comparative study of 56 cases. Arch Ophthalmol 99:1445–1454
Shinoda K, Hirakata A, Hida T, Yamaguchi Y, Fukuda M, Maekawa S, Azuma N (2000) Ultrastructural and immunohistochemical findings in five patients with vitreomacular traction syndrome. Retina 20:289–293
Smiddy WE, Maguire AM, Green WR, Michels RG, de la Cruz ZC, Engels C, Jaeger M, Rice TA (1989) Idiopathic epiretinal membranes: ultrastructural characteristics and clinicopathologic correlation. Ophthalmology 96:811–821
Heidenkummer HP, Kampik A (1992) Proliferative activity and immunohistochemical cell differentiation in human epiretinal membranes. Ger J Ophthalmol 1:170–175
Zhao F, Gandorfer A, Haritoglou C, Scheler R, Schaumberger MM, Kampik A, Schumann RG (2013) Epiretinal cell proliferation in macular pucker and vitreomacular traction syndrome: analysis of flat-mounted internal limiting membrane specimens. Retina 33:77–88
Schumann RG, Eibl KH, Zhao F, Scheerbaum M, Scheler R, Schaumberger MM, Wehnes H, Walch AK, Haritoglou C, Kampik A, Gandorfer A (2011) Immunocytochemical and ultrastructural evidence of glial cells and hyalocytes in internal limiting membrane specimens of idiopathic macular holes. Invest Ophthalmol Vis Sci 52:7822–7834
Ogawa K (2002) Scanning electron microscopic study of hyalocytes in the guinea pig eye. Arch Histol Cytol 65:263–268
Saga T, Tagawa Y, Takeuchi T, Nerome K, Matsuda H (1984) Electron microscopic study of cells in vitreous of guinea pig. Jpn J Ophthalmol 28:239–247
Uerara M, Imagawa T, Kitagawa H (1996) Morphological studies of the hyalocytes in the chicken eye: scanning electron microscopy and inflammatory response after the intravitreous injection of carbon particles. J Anat 188:661–669
Salu P, Claeskens W, de Wilde A, Hijmans W, Wisse E (1985) Light and electron microscopic studies of the rat hyalocyte after perfusion fixation. Ophthalmic Res 17:125–130
Qiao H, Hisatomi T, Sonoda K-H et al (2005) The characterisation of hyalocytes: the origin, phenotype and turnover. Br J Ophthalmol 89:513–517
Gandorfer A, Rohleder M, Grosselfinger S, Haritoglou C, Ulbig M, Kampik A (2005) Epiretinal pathology of diffuse diabetic macular edema associated with vitreomacular traction. Am J Ophthalmol 139:638–652
Hisatomi T, Enaida H, Sakamoto T, Kagimoto T, Ueno A, Nakamura T, Hata Y, Ishibashi T (2005) A new method for comprehensive bird’s-eye analysis of the surgically excised internal limiting membrane. Am J Ophthalmol 139:1121–1122
Hiscott PS, Grierson I, Trombetta CJ, Rahi AH, Marshall J, McLeod D (1984) Retinal and epiretinal glia–an immunochistochemical study. Br J Ophthalmol 68:698–707
Schumann RG, Schaumberger MM, Rohleder M, Haritoglou C, Kampik A, Gandorfer A (2006) Ultrastructure of the vitreomacular interface in full-thickness idiopathic macular holes: a consecutive analysis of 100 cases. Am J Ophthalmol 141:1112–1119
Schumann RG, Rohleder M, Schaumberger MM, Haritoglou C, Kampik A, Gandorfer A (2008) Idiopathic macular holes: ultrastructural aspects of surgical failure. Retina 28:340–349
Robinson JM, Takizawa T (2009) Correlative fluorescence and electron microscopy in tissues: immunocytochemistry. J Microsc 235:259–272
Takizawa T, Robinson JM (2003) Ultrathin cryosections: an important tool for immunofluorescence and correlative microscopy. J Histochem Cytochem 51:707–714
Asakawa H, Hiraoka Y, Haraguchi T (2014) A method for correlative light and electron microscopy for yeast cells. Micron 61C:51–63
Balazs EA, Toth LZ, Ozanics V (1980) Cytological studies on the developing vitreous as related to the hyaloid vessel system. Graefes Arch Clin Exp Ophthalmol 213:71–85
Llombart C, Nacher V, Ramos D, Luppo M, Carretero A, Navarro M, Melgarejo V, Armengol C, Rodriguez-Baeza R, Mendes-Jorge L, Ruberte J (2009) Morphological characterization of pecteneal hyalocytes in the developing quail retina. J Anat 215:280–291
Kohno R-I, Hata Y, Kawahara S, Kita T, Arita R, Mochizuki Y, Aiello LP, Ishibashi T (2009) Possible contribution of hyalocytes to idiopathic epiretinal membrane formation and its contraction. Br J Ophthalmol 93:1020–1026
Devarajan G, Chen M, Muckersie E, Xu H (2014) Culture and characterization of microglia from the adult murine retina. Sci World J. doi:10.1155/2014/894368
Maneu V, Noailles A, Megias J et al (2014) Retinal microglia are activated by systemic fungal infection. Invest Ophthalmol Vis Sci 55:3578–3585
Limb GA, Cole CJ, Earley O et al (1997) expression of hematopoietic cell markers by retinal pigment epithelial cells. Curr Eye Res 16:985–991
Lazarus HS, Hageman GS (1994) In situ characterization of the human hyalocyte. Arch Ophthalmol 112:1356–1362
Sakamoto T, Ishibashi T (2011) Hyalocytes: essential cells of the vitreous cavity in vitreoretinal pathophysiology. Retina 31:222–228
Sebag J, Gupta P, Rosen R, Garcia P, Sadun AA (2007) Macular holes and macular pucker: the role of vitreoschisis as imaged by optical coherence tomography/scanning laser ophthalmoscopy. Trans Am Ophthalmol Soc 105:121–129
Sebag J (2009) Vitreoschisis. Graefes Arch Clin Exp Ophthalmol 246:329–332
Newsome DA, Linsenmayer TF, Trelstad RL (1976) Vitreous body collagen. Evidence for a dual origin from the neural retina and hyalocytes. J Cell Biol 71:59–67
Nishitsuka K, Kashiwagi Y, Tojo N, Kanno C, Takahashi Y, Yamamoto T, Heldin P, Yamashita H (2007) Hyaluronan production regulation from porcine hyalocyte cell line by cytokines. Exp Eye Res 85:539–545
Vinores SA, Campochiaro PA, Conway BP (1990) Ultrastructural and electron-immunocytochemical characterization of cells in epiretinal membranes. Invest Ophthalmol Vis Sci 31:14–28
Vinores SA, Campochiaro PA, McGehee R, Orman W, Hackett SF, Hjelmeland LM (1990) Ultrastructural and immunocytochemical changes in retinal pigment epithelium, retinal glia, and fibroblasts in vitreous culture. Invest Ophthalmol Vis Sci 31:2529–2545
Gandorfer A, Schumann RG, Scheler R, Haritoglou C, Kampik A (2011) Pores of the inner limiting membrane in flat-mounted surgical specimens. Retina 31:977–981
Humbel BM, de Jong MD, Müller WH, Verkleij AJ (1998) Pre-embedding immunolabeling for electron microscopy: an evaluation of permeabilization methods and markers. Microsc Res Tech 42:43–58
Gandorfer A, Scheler R, Schumann R, Haritglou C, Kampik A (2009) Interference microscopy delineates cellular proliferations on flat mounted internal limiting membrane specimens. Br J Ophthalmol 93:120–122
Gandorfer A, Haritoglou C, Scheler R, Schumann R, Zhao F, Kampik A (2012) Residual cellular proliferation on the internal limiting membrane in macular pucker surgery. Retina 32:477–485
Financial support
This study was supported by the Ludwig-Maximilians-University Grant for Research and Education (FöFoLe, ID # 822).
Conflict of interest
The authors have no proprietary interest in any respect of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schumann, R.G., Gandorfer, A., Ziada, J. et al. Hyalocytes in idiopathic epiretinal membranes: a correlative light and electron microscopic study. Graefes Arch Clin Exp Ophthalmol 252, 1887–1894 (2014). https://doi.org/10.1007/s00417-014-2841-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-014-2841-x