Abstract
Background
To establish an animal model of retinal neovascularization using vascular endothelial growth factor (VEGF165) and analyze the model using optical coherence tomography (OCT), fluorescein angiography (FA), and histopathologic evaluation.
Methods
Twelve rabbits were divided into groups as follows: group 1 (n = 3), sham intravitreous injections of 0.1 ml of balanced saline; group 2 (n = 6), one 10-μg intravitreal injection of VEGF165 on day 0; and group 3 (n = 3), two 10-μg intravitreal injections of VEGF165, one on day 0 and one on day 7. Follow-up evaluations (days 0, 3, 7, 14, 21, 28) included obtaining fundus color photographs and FA, OCT, and histopathologic examinations. Eyes were enucleated and stained with hematoxylin and eosin (H&E).
Results
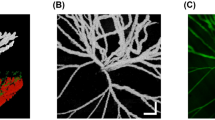
One injection of VEGF (group 2) was associated with dilatation and tortuosity of the retinal blood vessels that developed within 72 h. Retinal neovascularization was present by day 7 and regressed by day 14. However, even on day 28, the capillaries were still tortuous. Two VEGF injections (group 3) caused increased leakage and neovascularization up to day 14; severe capillary nonperfusion was seen during week 4. At the end of the follow-up period, OCT and histopathologic examination of group 3 showed peripapillary tractional retinal detachments. By day 7, the differences between the retinal thickness seen on OCT in groups 2 and 3 and the group 1 control group were significant (p < 0.001). The histologic findings showed increased vessel size in groups 2 and 3 by days 14 and 28 compared with the controls.
Conclusions
FA, OCT, and histopathologic findings showed that this retinal neovascularization model is efficient, sustainable, and reliable. One injection of VEGF165 created neovascularization that peaked after 1 week; two injections created more intense neovascularization that evolved to retinal detachments after 4 weeks.







Similar content being viewed by others
References
Friedman DS, O’Colmain BJ, Muñoz B, Tomany SC, McCarty C, de Jong PT, Nemesure B, Mitchell P, Kempen J, Congdon N (2004) Prevalence of age-related macular degeneration in the United States. Arch Ophthalmol 122:564–572
Kempen JH, O’Colmain BJ, Leske MC, Haffner SM, Klein R, Moss SE, Taylor HR, Hamman RF, Eye Diseases Prevalence Research Group (2004) The prevalence of diabetic retinopathy among adults in the United States. Arch Ophthalmol 122:552–563
Aiello LP, Avery RL, Arrigg PG, Arrigg PG, Keyt BA, Jampel HD, Shah ST, Pasquale LR, Thieme H, Iwamoto MA, Park JE, Nguyen HV, Aiello LM, Ferrara N, King GL (1994) Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Engl J Med 331:1480–1487
Wells JA, Murthy R, Chibber R, Nunn A, Molinatti PA, Kohner EM, Gregor ZJ (1996) Levels of vascular endothelial growth factor are elevated in the vitreous of patients with subretinal neovascularisation. Br J Ophthalmol 80:363–366
Galan A, Ferlin A, Caretti L, Buson G, Sato G, Frigo AC, Foresta C (2010) Association of age-related macular degeneration with polymorphisms in vascular endothelial growth factor and its receptor. Ophthalmology 117:1769–1774
Ferrara N, Houck K, Jakeman L, Leung DW (1992) Molecular and biological properties of the vascular endothelial growth factor family of proteins. Endocr Rev 13:18–32
Guerrin M, Moukadiri H, Chollet P, Moro F, Dutt K, Malecaze F, Plouet J (1995) Vasculotropin/vascular endothelial growth factor is an autocrine growth factor for human retinal pigment epithelial cells cultured in vitro. J Cell Physiol 164:385–394
Veikkola T, Alitalo K (1999) VEGFs, receptors and angiogenesis. Semin Cancer Biol 9:211–220
Park JE, Keller GA, Ferrara N (1993) The vascular endothelial growth factor (VEGF) isoforms: differential deposition into the subepithelial extracellular matrix and bioactivity of extracellular matrix-bound VEGF. Mol Biol Cell 4:1317–1326
Ishida S, Usui T, Yamashiro K, Kaji Y, Amano S, Ogura Y, Hida T, Oguchi Y, Ambati J, Miller JW, Gragoudas ES, Ng YS, D’Amore PA, Shima DT, Adamis AP (2003) VEGF164-mediated inflammation is required for pathological, but not physiological, ischemia-induced retinal neovascularization. J Exp Med 198:483–489
Ishida S, Usui T, Yamashiro K, Kaji Y, Ahmed E, Carrasquillo KG, Amano S, Hida T, Oguchi Y, Adamis AP (2003) VEGF 164 is proinflammatory in the diabetic retina. Invest Ophthalmol Vis Sci 44:2155–2162
Sampaio RB, Mendonca RJ, Simioni AR, Costa RA, Siqueira RC, Correa VM, Tedesco AC, Haddad A, Coutinho Netto J, Jorge R (2010) Rabbit retinal neovascularization induced by latex angiogenic-derived fraction: an experimental model. Curr Eye Res 35:56–62
Tano Y, Chandler DB, Machemer R (1981) Retinal neovascularization after intravitreal fibroblast injection. Am J Ophthalmol 92:103–109
Saati S, Agrawal RN, Louie S, Chader GJ, Humayun MS (2010) Effect of multiple injections of small divided doses vs single injection of intravitreal bevacizumab on retinal neovascular model in rabbits. Graefes Arch Clin Exp Ophthalmol 248:457–466
Wong CG, Rich KA, Liaw LH, Hsu HT, Berns MW (2001) Intravitreal VEGF and bFGF produce florid retinal neovascularization and hemorrhage in the rabbit. Curr Eye Res 22:140–147
Ozaki H, Hayashi H, Vinores SA, Moromizato Y, Campochiaro PA, Oshima K (1997) Intravitreal sustained release of VEGF causes retinal neovascularization in rabbits and breakdown of blood–retinal barrier in rabbits and primates. Exp Eye Res 64:505–517
Deem CW, Futterman S, Kalina RE (1974) Induction of endothelial cell proliferation in rat retinal venules by chemical and indirect physical trauma. Invest Ophthalmol 13:580–585
Hamilton AM, Marshall J, Kohner EM, Bowbyes JA (1975) Retinal new vessel formation following experimental vein occlusion. Exp Eye Res 20:493–497
Tamura M (2001) Neovascularization in experimental retinal venous obstruction in rabbits. Jpn J Ophthalmol 45:144–150
Pournaras CJ, Tsacopoulos M, Strommer K, Gilodi N, Leunenberger PM (1990) Experimental retinal branch vein occlusion in miniature pigs induces local tissue hypoxia and vasoproliferative microangiopathy. Ophthalmology 97:1321–1328
Finkelstein D, Brem S, Patz A, Folkman J, Miller S, Ho-Chen C (1977) Experimental retinal neovascularization induced by intravitreal tumors. Am J Ophthalmol 83:660–664
Penn JS, Tolman BL, Lowery LA (1993) Variable oxygen exposure causes preretinal neovascularization in the newborn rat. Invest Ophthalmol Vis Sci 34:576–585
Andreoli CM, Miller JW (2007) Anti-vascular endothelial growth factor therapy for ocular neovascular disease. Curr Opin Ophthalmol 18:502–508
Mohamed Q, Gillies MC, Wong TY (2007) Management of diabetic retinopathy: a systematic review. JAMA 298:902–916
Ameri H, Chader GJ, Kim JG, Sadda SR, Rao NA, Humayun M (2007) The effects of intravitreous bevacizumab on retinal neovascular membrane and normal capillaries in rabbits. Invest Ophthalmol Vis Sci 48:5708–5715
Alikacem N, Yoshizawa T, Nelson KD, Wilson CA (2000) Quantitative MR imaging study of intravitreal sustained release of VEGF in rabbits. Invest Ophthalmol Vis Sci 41:1561–1569
Sato T, Kusaka S, Shimojo H, Fujikado T (2009) Simultaneous analyses of vitreous levels of 27 cytokines in eyes with retinopathy of prematurity. Ophthalmology 116:2165–2169
Pierce EA, Avery RL, Foley ED, Aiello LP, Smith LE (1995) Vascular endothelial growth factor/vascular permeability factor expression in a mouse model of retinal neovascularization. Proc Natl Acad Sci USA 92:905–909
Okamoto N, Tobe T, Hackett SF, Ozaki H, Vinores MA, La Rochele W, Zack DJ, Campochiaro PA (1997) Transgenic mice with increased expression of vascular endothelial growth factor in the retina: a new model of intraretinal and subretinal neovascularization. Am J Pathol 151:281–291
Suto C, Kitano S, Hori S (1992) An experimental model of intravitreal neovascularization in rabbits [Japanese]. Nihon Ganka Gakkai Zasshi 96:959–966
Pournaras CJ, Rungger-Brändle E, Riva CE, Hardarson SH, Stefansson E (2008) Regulation of retinal blood flow in health and disease. Prog Retin Eye Res 27:284–330
Hofman P, van Blijswijk BC, Gaillard PJ, Vrensen GF, Schlingermann RO (2001) Endothelial cell hypertrophy induced by vascular endothelial growth factor in the retina: new insights into the pathogenesis of capillary nonperfusion. Arch Ophthalmol 119:861–866
Tolentino MJ, Miller JW, Gragoudas ES, Jakobiec FA, Flynn E, Chatzistefanou K, Ferrara N, Adamis AP (1996) Intravitreous injections of vascular endothelial growth factor produce retinal ischemia and microangiopathy in an adult primate. Ophthalmology 103:1820–1828
McIntosh RL, Rogers SL, Lim L, Cheung N, Wang JJ, Mitchell P, Kowalski JW, Nguyen HP, Wong TY (2010) Natural history of central retinal vein occlusion: an evidence-based systematic review. Ophthalmology 117:1113–1123
Declaration of interest
The authors report no financial conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
This manuscript is part of Dr. Arana’s master thesis from the postgraduate program in clinical surgery of Federal University of Parana.
Rights and permissions
About this article
Cite this article
Arana, L.A., Pinto, A.T., Chader, G.J. et al. Fluorescein angiography, optical coherence tomography, and histopathologic findings in a VEGF165 animal model of retinal angiogenesis. Graefes Arch Clin Exp Ophthalmol 250, 1421–1428 (2012). https://doi.org/10.1007/s00417-012-1978-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-012-1978-8