Abstract
Background
Endogenous angiogenesis inhibitors act as natural negative feedback in the focal area during the neovascularization process, and have less interference on physiological angiogenesis, and thus fewer negative side-effects. These inhibitors are potential candidates to combine with or substitutes for current popular anti-angiogenesis treatments to have synergistic effect. In this study, the effects of recombinant endothelial growth inhibitor protein (rhEDI-8t), a novel endogenous protein originated from collagen VIII, was investigated on ocular neovascularization (NV). Endostatin, a well-identified endogenous angiogenesis inhibitor, was compared in parallel and served as a positive control.
Methods
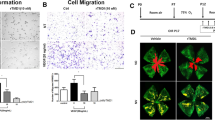
The inhibitory effect of rhEDI-8t on vascular endothelial cells was evaluated by a human umbilical vascular endothelial cells (HUVEC) proliferation test and a bovine aortic endothelial cells (BAEC) migration experiment. The effect of rhEDI-8t on ocular NV was further investigated in mice with choroidal neovascularization (choroidal NV) induced by laser, ischemic retinopathy and transgenic mice with expression of VEGF in photoreceptors (rho/VEGF) respectively.
Results
RhEDI-8t inhibited the growth of HUVECs and migration of BAECs stimulated by basic fibroblast growth factor (bFGF). Mice intravitreally treated with rhEDI-8t showed a significant reduction of choroidal NV, retinal NV and subretinal NV.
Conclusion
Endogenous angiogenesis inhibitor rhEDI-8t showed a potent anti-angiogenesis effect in both in vitro and in vivo experiments. It contributed to the suppression of ocular NV. The study suggested that rhEDI-8t could be a subsidiary potent therapeutic medicine in addition to anti-VEGF therapy in future clinical anti-angiogenesis treatment.





Similar content being viewed by others
Abbreviations
- NV:
-
Neovascularization
- VEGF:
-
Vascular endothelial growth factor
- rho/VEGF:
-
Rhodopsin promoter/ VEGF
- AMD:
-
Age-related macular degeneration
- HUVEC:
-
Human umbilical vascular endothelial cells
- BAEC:
-
Bovine aortic endothelial cells
Reference
Folkman J (1971) Tumor angiogenesis: therapeutic implications. N Engl J Med 285(21):1182–1186
Hosseini H, Nejabat M (2007) A potential therapeutic strategy for inhibition of corneal neovascularization with new anti-VEGF agents. Med Hypotheses 68(4):799–801
Tobe T, Okamoto N, Vinores MA, Derevjanik NL, Vinores SA, Zack DJ, Campochiaro PA (1998) Evolution of neovascularization in mice with overexpression of vascular endothelial growth factor in photoreceptors. Invest Ophthalmol Vis Sci 39(1):180–188
O’Reilly MS, Boehm T, Shing Y, Fukai N, Vasios G, Lane WS, Flynn E, Birkhead JR, Olsen BR, Folkman J (1997) Endostatin: an endogenous inhibitor of angiogenesis and tumor growth. Cell 88(2):277–285
Abdollahi A, Hahnfeldt P, Maercker C, Gröne HJ, Debus J, Ansorge W, Folkman J, Hlatky L, Huber PE (2004) Endostatin’s antiangiogenic signaling network. Mol Cell 13(5):649–663
Ramchandran R, Dhanabal M, Volk R, Waterman MJ, Segal M, Lu H, Knebelmann B, Sukhatme VP (1999) Antiangiogenic activity of restin, NC10 domain of human collagen XV: comparison to endostatin. Biochem Biophys Res Commun 255(3):735–739
Colorado PC, Torre A, Kamphaus G, Maeshima Y, Hopfer H, Takahashi K, Volk R, Zamborsky ED, Herman S, Sarkar PK, Ericksen MB, Dhanabal M, Simons M, Post M, Kufe DW, Weichselbaum RR, Sukhatme VP, Kalluri R (2000) Anti-angiogenic cues from vascular basement membrane collagen. Cancer Res 60(9):2520–2526
Xu R, Yao ZY, Xin L, Zhang Q, Li TP, Gan RB (2001) NC1 domain of human type VIII collagen (alpha 1) inhibits bovine aortic endothelial cell proliferation and causes cell apoptosis. Biochem Biophys Res Commun 289(1):264–268
Risau W (1997) Mechanisms of angiogenesis. Nature 386:671–674
Shono T, Kanetake H, Kanda S (2001) The role of mitogen activated protein kinase activation within focal adhesions in chemotaxis toward FGF-2 by murine brain capillary endothelial cells. Exp Cell Res 264:275–283
Kiec-Wilk B, Grzybowska-Galuszka J, Polus A, Pryjma J, Knapp A, Kristiansen K (2010) The MAPK-dependent regulation of the Jagged/notch gene expression by VEGF, bFGF or PPAR gamma mediated angiogenesis in HUVEC. J Physiol Pharmacol 61(2):217–225
Okamoto N, Tobe T, Hackett SF, Ozaki H, Vinores MA, LaRochelle W, Zack DJ, Campochiaro PA (1997) Transgenic mice with increased expression of vascular endothelial growth factor in the retina: a new model of intraretinal and subretinal neovascularization. Am J Pathol 151(1):281–291
Mendrinos E, Petropoulos IK, Mangioris G, Papadopoulou DN, Stangos AN, Pournaras CJ (2008) Lactate-induced retinal arteriolar vasodilation implicates neuronal nitric oxide synthesis in minipigs. Invest Ophthalmol Vis Sci 49(11):5060–5066
Tobe T, Ortega S, Luna JD, Derevjanik NL, Vinores SA, Zack DJ, Vinores SA, Basilico C, Campochiaro PA (1998) Targeted disruption of the FGF2 gene does not prevent choroidal neovascularization in a murine model. Am J Pathol 153(5):1641–1646
Das A, McGuire PG (2003) Retinal and choroidal angiogenesis: pathophysiology and strategies for inhibition. Progr Retin Eye Res 22(6):721–748
Campochiaro PA, Hackett SF (2003) Ocular neovascularization: a valuable model system. Oncogene 22(42):6537–6548
Mori K, Ando A, Gehlbach P, Nesbitt D, Takahashi K, Goldsteen D, Penn M, Chen CT, Mori K, Melia M, Phipps S, Moffat D, Brazzell K, Liau G, Dixon KH, Campochiaro PA (2001) Inhibition of choroidal neovascularization by intravenous injection of adenoviral vectors expressing secretable endostatin. Am J Pathol 159(1):313–320
Noma H, Funatsu H, Yamashita H, Kitano S, Mishima HK, Hori S (2002) Regulation of angiogenesis in diabetic retinopathy: possible balance between vascular endothelial growth factor and endostatin. Arch Ophthalmol 120(8):1075–1080
Bhutto IA, Kim SY, McLeod DS, Merges C, Fukai N, Olsen BR, Lutty GA (2004) Localization of collagen XVIII and the endostatin portion of collagen XVIII in aged human control eyes and eyes with age-related macular degeneration. Invest Ophthalmol Vis Sci 45(5):1544–1552
Ferreras M, Felbor U, Lenhard T, Olsen BR, Delaissé J (2000) Generation and degradation of human endostatin proteins by various proteinases. FEBS Lett 486(3):247–251
Lee SJ, Jang JW, Kim YM, Lee HI, Jeon JY, Kwon YG, Lee ST (2002) Endostatin binds to the catalytic domain of matrix metalloproteinase-2. FEBS Lett 519(1–3):147–152
Nyberg P, Xie L, Kalluri R (2005) Endogenous inhibitors of angiogenesis. Cancer Res 65:3967–3979
Zatterstrom UK, Felbor U, Fukai N, Olsen BR (2000) Collagen XVIII/endostatin structure and functional role in angiogenesis. Cell Struct Funct 25:97–101
Ni Q, Ji H, Zhao Z, Fan X, Xu C (2009) Endostar, a modified endostatin inhibits non small cell lung cancer cell in vitro invasion through osteopontin-related mechanism. Eur J Pharmacol 614(1–3):1–6
Bai RZ, Wu Y, Liu Q, Xie K, Wei YQ, Wang YS, Liu K, Luo Y, Su JM, Hu B, Liu JY, Li Q, Niu T, Zhao ZW, Yang L (2009) Suppression of lung cancer in murine model: treated by combination of recombinant human endostsatin adenovirus with low-dose cisplatin. J Exp Clin Cancer Res 5:28–31
Ouyang XS, Wang X, Ling MT, Wong HL, Tsao SW, Wong YC (2002) Id-1 stimulates serum independent prostate cancer cell proliferation through inactivation of p16 (INK4a)/pRB pathway. Carcinogenesis 23(5):721–725
Ling MT, Wang X, Ouyang XS, Lee TK, Fan TY, Xu K, Tsao SW, Wong YC (2002) Activation of MAPK signaling pathway is essential for Id-1 induced serum independent prostate cancer cell growth. Oncogene 21(55):8498–8505
Seo MS, Kwak N, Ozaki H, Yamada H, Okamoto N, Yamada E, Fabbro D, Hofmann F, Wood JM, Campochiaro PA (1999) Dramatic inhibition of retinal and choroidal neovascularization by oral administration of a kinase inhibitor. Am J Pathol 154(6):1743–1753
Lu F, Adelman RA (2009) Are intravitreal bevacizumab and ranibizumab effective in a rat model of choroidal neovascularization? Graefes Arch Clin Exp Ophthalmol 247(2):171–177
O’Reilly MS, Holmgren L, Shing Y, Chen C, Rosenthal RA, Moses M, Lane WS, Cao Y, Sage EH, Folkman J (1994) Angiostatin: a novel angiogenesis inhibitor that mediates the suppression of metastases by a Lewis lung carcinoma. Cell 79(2):315–328
O’Reilly MS, Pirie-Shepherd S, Lane WS, Folkman J (1999) Antiangiogenic activity of the cleaved conformation of the serpin antithrombin. Science 285(5435):1926–1928
Maione TE, Gray GS, Petro J, Hunt AJ, Donner AL, Bauer SI, Carson HF, Sharpe RJ (1990) Inhibition of angiogenesis by recombinant human platelet factor-4 and related peptides. Science 247(4938):77–79
Dawson DW, Volpert OV, Gillis P, Crawford SE, Xu H, Benedict W, Bouck NP (1999) Pigment epithelium-derived factor: a potent inhibitor of angiogenesis. Science 285(5425):245–248
Acknowledgments
Supported by Shanghai Leading Academic Discipline Project S30205, Shanghai Municipal Education Committee Project 09YZ86, Shanghai Rising-Star Program 10QA1404600, Shanghai Nature Science Foundation 11ZR1422000, China.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Ling Zhang and Xi Shen contributed equally to the manuscript
Rights and permissions
About this article
Cite this article
Zhang, L., Shen, X., Lu, Q. et al. A potential therapeutic strategy for inhibition of ocular neovascularization with a new endogenous protein: rhEDI-8t. Graefes Arch Clin Exp Ophthalmol 250, 731–739 (2012). https://doi.org/10.1007/s00417-011-1765-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-011-1765-y