Abstract
Purpose
Although activated protein C (APC) is effective in preventing the death of retinal neurons in ischemic retinopathy, it is not known at what concentrations it becomes retinotoxic. The purpose of this study was to determine the concentrations of intravitreal APC that are safe and those that are toxic for the retina, using rabbit eyes.
Methods
The left eyes of 12 rabbits received an intravitreal injection of 1.5 to 150 μg of APC in 0.1 ml of saline. The fellow eyes were treated with an intravitreal injection of the same amount of saline. Slit-lamp examination, fundus examination, fluorescein angiography (FA), and electroretinograms (ERGs) were performed before and at different times after the injection. The eyes were enucleated at 6 months after the injection and examined histologically.
Results
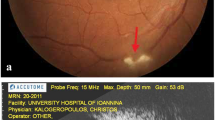
The clinical and histological differences between the control eyes and the eyes that had APC injections up to 15 μg were not significant. Localized retinal edema was observed in two of three eyes with 150 μg of APC immediately after the injection. In these two eyes, chorioretinal atrophy was observed in the area of the retinal edema at 6 months, which corresponded with a hyperfluorescent area in the FA images and focal retinal degeneration histologically. No significant changes were detected in the full-field ERGs in the eyes treated even with 150 μg of APC throughout the observation period.
Conclusion
Our results show that an intravitreal injection of APC at a dose ≤15 μg is safe, but 150 μg of APC can be toxic to the retina.




Similar content being viewed by others
References
Campochiaro PA (2000) Retinal and choroidal neovascularization. J Cell Physiol 184:301–310
Kaur C, Foulds WS, Ling EA (2008) Hypoxia–ischemia and retinal ganglion cell damage. Clin Ophthalmol 2:879–889
Fernández JA, Xu X, Liu D, Zlokovic BV, Griffin JH (2003) Recombinant murine-activated protein C is neuroprotective in a murine ischemic stroke model. Blood Cells Mol Dis 30:271–276
Guo H, Liu D, Gelbard H, Cheng T, Insalaco R, Fernandez JA, Griffin JH, Zlokovic BV (2004) Activated protein C prevents neuronal apoptosis via protease activated receptors 1 and 3. Neuron 41:563–572
Liu D, Cheng T, Guo H, Fernandez JA, Griffin JH, Song X, Zlokovic BV (2004) Tissue plasminogen activator neurovascular toxicity is controlled by activated protein C. Nat Med 10:1379–1383
Zlokovic BV, Zhang C, Liu D, Fernandez JA, Griffin JH, Chopp M (2005) Functional recovery after embolic stroke in rodents by activated protein C. Ann Neurol 58:474–477
Weiler H, Ruf W (2008) Activated protein C in sepsis: the promise of nonanticoagulant activated protein C. Curr Opin Hematol 15:487–493
Du Z, Yamamoto T, Ueda T, Suzuki M, Tano Y, Kamei M (2010) Activated protein C rescues the retina from ischemia-induced cell death. Invest Ophthalmol Vis Sci, doi:10.1167/iovs.10-5557
Morrison VL, Koh HJ, Cheng L, Bessho K, Davidson MC, Freeman WR (2006) Intravitreal toxicity of the kenalog vehicle (benzyl alcohol) in rabbits. Retina 26:339–344
Domotor E, Benzakour O, Griffin JH, Yule D, Fukudome K, Zlokovic BV (2003) Activated protein C alters cytosolic calcium flux in human brain endotherium via binding to endotherial protein C receptor and activation of protease activated receptor-1. Blood 101:4797–4801
Gorbacheva L, Pinelis V, Ishiwata S, Strukova S, Reiser G (2010) Activated protein C prevents glutamate-and thrombin-induced activation of nuclear factor-kappaB in cultured hippocamapal neurons. Neuroscience 165:1138–1146
Gupta A, Berg DT, Gerlitz B, Richardson MA, Galbreath E, Syed S, Sharma AC, Lowry SF, Grinnell BW (2007) Activated protein C suppresses adrenomedullin and ameliorate lipopolysaccharifde-induced hypotension. Shock 28:468–476
Zachary IG, Forster RK (1976) Experimental intravitreal gentamicin. Am J Ophthalmol 82:604–611
McDonald HR, Schatz H, Allen AW, Chenoweth RG, Cohen HB, Crawford JB, Klein R, May DR, Snider JD 3rd (1986) Retinal toxicity secondary to intraocular gentamicin injection. Ophthalmology 93:871–877
Rosenfeld PJ, Schwartz SD, Blumenkranz MS, Miller JW, Haller JA, Reimann JD, Greene WL, Shams N (2005) Maximum tolerated dose of a humanized anti-vascular endothelial growth factor antibody fragment for treating neovascular age-related macular degeneration. Ophthalmology 112:1048–1053
Rosenfeld PJ, Fung AE, Puliafito CA (2005) Optical coherence tomography findings after an intravitreal injection of bevacizumab (avastin) for macular edema from central retinal vein occlusion. Ophthalmic Surg Lasers Imaging 36:336–339
Avery RL, Pieramici DJ, Rabena MD, Castellarin AA, Nasir MA, Giust MJ (2006) Intravitreal bevacizumab (Avastin) for neovascular age-related macular degeneration. Ophthalmology 113:363–372
Heier JS, Antoszyk AN, Pavan PR, Leff SR, Rosenfeld PJ, Ciulla TA, Dreyer RF, Gentile RC, Sy JP, Hantsbarqer G, Shams N (2006) Ranibizumab for treatment of neovascular age-related macular degeneration: a phase I/II multicenter, controlled, multidose study. Ophthalmology 113:633
Jager RD, Aiello LP, Patel SC, Cunningham ET Jr (2004) Risks of intravitreous injection: a comprehensive review. Retina 24:676–698
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nishida, K., Kamei, M., Du, Zj. et al. Safety threshold of intravitreal activated protein-C. Graefes Arch Clin Exp Ophthalmol 249, 833–838 (2011). https://doi.org/10.1007/s00417-010-1566-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-010-1566-8