Abstract
Background
Myopic maculopathy is the leading cause of subfoveal choroidal neovascularization (CNV) among patients under 50 years of age. New antiangiogenic drugs are being used off-label to treat myopic CNV and the short-term outcome of these therapies has been reported. The aim of this study is to report the changes in best-corrected visual acuity (BCVA) and optical coherence tomography (OCT) in highly myopic CNV treated by intravitreal bevacizumab at 2 years.
Methods
Prospective non-randomized, interventional case series study of 19 highly myopic eyes from 18 patients with subfoveal and juxtafoveal CNV treated by three monthly intravitreal injections of 1.25 mg bevacizumab. Patients were evaluated for BCVA and OCT at baseline and then monthly for 2 years.
Results
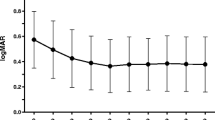
Eleven eyes were naive for treatment and eight eyes had been previously treated by photodynamic therapy. LogMAR BCVA averaged 0.54 (SD 0.25, range 0.2–1.0; Snellen 20/69) at baseline; 0.40 (SD 0.35, range 0.0–1.2; Snellen 20/50) at 1 year; and 0.47 (SD 0.31, range 0.0–1.0; Snellen 20/59) at 2 years (p = 0.04 and p = 0.20, respectively, Student's t test paired data). Re-treatment was performed in four eyes during the first year: three eyes at month six and one eye at month 12. Four eyes required one re-injection during the second year at months 14, 18, 20, and 24. Neither ocular nor systemic adverse reactions were detected.
Conclusions
Intravitreal bevacizumab seems to be effective for subfoveal and juxtafoveal CNV in highly myopic eyes. BCVA gain decreases and is no longer significant by the end of the second year.
Similar content being viewed by others
References
Cohen SY, Laroche A, Leguen Y, Soubrane G, Coscas GJ (1996) Etiology of choroidal neovascularization in young patients. Ophthalmology 103:1241–1244
Yamamoto I, Rogers AH, Reichel E, Yates PA, Duker JS (2007) Intravitreal bevacizumab (Avastin) as treatment for subfoveal choroidal neovascularisation secondary to pathological myopia. Br J Ophthalmol 91:157–160
Tewari A, Dhalla MS, Apte RS (2006) Intravitreal bevacizumab for treatment of choroidal neovascularization in pathologic myopia. Retina 26:1093–1094
Sakaguchi H, Ikuno Y, Gomi F, Kamei M, Sawa M, Tsujikawa M, Oshima Y, Kusaka S, Tano Y (2007) Intravitreal injection of bevacizumab for choroidal neovascularisation associated with pathological myopia. Br J Ophthalmol 91:161–165
Ruiz-Moreno JM, Gomez-Ulla F, Montero JA, Ares S, Lopez-Lopez F, Rodriguez M, Fernandez M (2009) Intravitreous bevacizumab to treat subfoveal choroidal neovascularization in highly myopic eyes: short-term results. Eye 23:334–338
Nguyen QD, Shah S, Tatlipinar S, Do DV, Anden EV, Campochiaro PA (2005) Bevacizumab suppresses choroidal neovascularisation caused by pathological myopia. Br J Ophthalmol 89:1368–1370
Mandal S, Venkatesh P, Sampangi R, Garg S (2007) Intravitreal bevacizumab (Avastin) as primary treatment for myopic choroidal neovascularization. Eur J Ophthalmol 17:620–626
Laud K, Spaide RF, Freund KB, Slakter J, Klancnik JM Jr (2006) Treatment of choroidal neovascularization in pathologic myopia with intravitreal bevacizumab. Retina 26:960–963
Chan WM, Lai TY, Liu DT, Lam DS (2007) Intravitreal bevacizumab (Avastin) for myopic choroidal neovascularization: six-month results of a prospective pilot study. Ophthalmology 114:2190–2196
Arias L, Planas N, Prades S, Caminal JM, Rubio M, Pujol O, Roca G (2008) Intravitreal bevacizumab (Avastin) for choroidal neovascularisation secondary to pathological myopia: 6-month results. Br J Ophthalmol 92:1035–1039
Wu PC, Chen YJ (2009) Intravitreal injection of bevacizumab for myopic choroidal neovascularization: 1-year follow-up. Eye
Ruiz-Moreno JM, Montero JA, Gomez-Ulla F, Ares S (2009) Intravitreal bevacizumab to treat subfoveal choroidal neovascularisation in highly myopic eyes: 1-year outcome. Br J Ophthalmol 93:448–451
Ikuno Y, Sayanagi K, Soga K, Sawa M, Tsujikawa M, Gomi F, Tano Y (2009) Intravitreal bevacizumab for choroidal neovascularization attributable to pathological myopia: one-year results. Am J Ophthalmol 147(94–100):e101
Gharbiya M, Allievi F, Mazzeo L, Gabrieli CB (2009) Intravitreal bevacizumab treatment for choroidal neovascularization in pathologic myopia: 12-month results. Am J Ophthalmol 147(84–93):e81
Chan WM, Lai TY, Liu DT, Lam DS (2009) Intravitreal bevacizumab (Avastin) for myopic choroidal neovascularisation: 1-year results of a prospective pilot study. Br J Ophthalmol 93:150–154
Tong JP, Chan WM, Liu DT, Lai TY, Choy KW, Pang CP, Lam DS (2006) Aqueous humor levels of vascular endothelial growth factor and pigment epithelium-derived factor in polypoidal choroidal vasculopathy and choroidal neovascularization. Am J Ophthalmol 141:456–462
Bennett MD, Yee W (2007) Pegaptanib for myopic choroidal neovascularization in a young patient. Graefes Arch Clin Exp Ophthalmol 245:903–905
Silva RM, Ruiz-Moreno JM, Nascimento J, Carneiro A, Rosa P, Barbosaa A, Carvalheira F, Abreu JR, Cunha-Vaz JG (2008) Short-term efficacy and safety of intravitreal ranibizumab for myopic choroidal neovascularization. Retina 28:1117–1123
Konstantinidis L, Mantel I, Pournaras JA, Zografos L, Ambresin A (2009) Intravitreal ranibizumab (Lucentis) for the treatment of myopic choroidal neovascularization. Graefes Arch Clin Exp Ophthalmol 247:311–318
Figurska M, Stankiewicz A (2008) Anty-VEGF therapy in the treatment of myopic macular choroidal neovascularization–cases report. Klin Oczna 110:387–391
Ruiz-Moreno JM, Amat P, Montero JA, Lugo F (2008) Photodynamic therapy to treat choroidal neovascularisation in highly myopic patients: 4 years’ outcome. Br J Ophthalmol 92:792–794
Ikuno Y, Nagai Y, Matsuda S, Arisawa A, Sho K, Oshita T, Takahashi K, Uchihori Y, Gomi F (2009) Two-year visual results for older Asian women treated with photodynamic therapy or bevacizumab for myopic choroidal neovascularization. Am J Ophthalmol 149:140–146
Krebs I, Binder S, Stolba U, Glittenberg C, Brannath W, Goll A (2005) Choroidal neovascularization in pathologic myopia: three-year results after photodynamic therapy. Am J Ophthalmol 140:416–425
Ruiz-Moreno JM, Montero JA (2003) Subretinal fibrosis after photodynamic therapy in subfoveal choroidal neovascularisation in highly myopic eyes. Br J Ophthalmol 87:856–859
Baba T, Kubota-Taniai M, Kitahashi M, Okada K, Mitamura Y, Yamamoto S (2009) Two-year comparison of photodynamic therapy and intravitreal bevacizumab for treatment of myopic choroidal neovascularization. Br J Ophthalmol. doi:10.1136/bjo.2009.166025
Ruiz-Moreno JM, Montero JA (2002) Long-term visual acuity after argon green laser photocoagulation of juxtafoveal choroidal neovascularization in highly myopic eyes. Eur J Ophthalmol 12:117–122
Rosenfeld PJ, Rich RM, Lalwani GA (2006) Ranibizumab: Phase III clinical trial results. Ophthalmol Clin North Am 19:361–372
Acknowledgments
The authors have full control of all primary data and they agree to allow Graefe’s Archive for Clinical and Experimental Ophthalmology to review their data if requested. This study was supported in part by a grant of the Spanish Ministry of Health, Instituto de Salud Carlos III, Red Temática de Investigación Cooperativa en Salud “Patología ocular del envejecimiento, calidad visual y calidad de vida” (RD07/0062/0019). The design of this study was reviewed and approved by the clinical ethics committee at Vissum Ophthalmological Institute of Alicante. Written informed consent and individualized approval from the National Ministry of Health was obtained prior to the procedure. This study has been performed in accordance with the ethical standards of the 1964 Declaration of Helsinki, and data gathering was performed after obtaining written informed consent. Patients were informed about the off-label situation of this therapy and women of child-bearing age were also informed about the possible risks of pregnancy and in utero exposition.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ruiz-Moreno, J.M., Montero, J.A. Intravitreal bevacizumab to treat myopic choroidal neovascularization: 2-year outcome. Graefes Arch Clin Exp Ophthalmol 248, 937–941 (2010). https://doi.org/10.1007/s00417-010-1340-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-010-1340-y