Abstract
Background
To investigate how transient pressure applied to the retinal pigment epithelium (RPE) layer and choroid affects choroidal blood flow in rabbits.
Methods
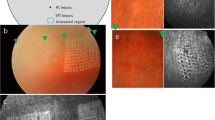
Twelve rabbits underwent vitrectomy and local retinectomy. In nine of the rabbits a glass rod was used to exert brief pressure on the RPE layer and choroid. Three of the rabbits had no pressure indentation and were considered to be controls. The choroidal circulation was studied by indocyanine green (ICG) angiography. The retina and choroid were studied by postmortem histology.
Results
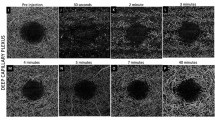
Pressure on the RPE layer and choroid caused nonfluorescence in segments of retinal arteries and veins and reduced fluorescence in adjacent choroidal capillaries, producing a black region at the pressure site in the angiograms. The size of this region decreased during the angiogram, often accompanied by the appearance of fine channels considered to be flow through the partially blocked vessels; the obstructed ends of the vessels became increasingly hyperfluorescent. These changes lasted for about 24 h before the choroidal circulation recovered. Histology showed evidence of thrombotic-like material in choroidal arteries and veins at the areas of absent perfusion. After local retinectomies, there was no evidence of thrombosis in control eyes where no pressure had been applied.
Conclusion
Brief pressure on the RPE and choroid causes immediate reduction in flow through choroidal vessels, which appears to be due to local thrombosis in small segments of these vessels that resolves slowly. This may reflect a tendency for thrombi to form rapidly in choroidal vessels; it may also depend on neural reflexes causing vasoconstriction. The long time course of recovery could result in retinal ischemia and may underlie the pathophysiology of other pressure insults to the choroid.






Similar content being viewed by others
References
Ambache N, Kavanagh L, Whiting J (1965) Effect of mechanical stimulation on rabbit eye: release of active substance in anterior chamber perfusate. J Physiol 176:378–408
Alm A (2003) Ocular circulation. In: Kaufman P, Alm A (eds) Adler’s physiology of the eye. Mosby, St. Louis, section 13
Berlin R (1873) Zur sogenannten commotio retinae. Klin Monatsbl Augenheilkd 1:42–78
Bouchenaki N, Cimino L, Auer C, Tao Tran V, Herbort CP (2002) Assessment and classification in posterior uveitis using indocyanine green angiography. Klin Monatsbl Augenheilkd 219:243–249
Cioffi GA, Alm A (2001) Measurement of ocular blood flow. J Glaucoma 10(Suppl 1):S62–S64
Flugel C, Tamm ER, Mayer B, Lutjen-Drecoll E (1994) Species differences in choroidal vasodilative innervation: evidence for specific intrinsic nitrergic and VIP-positive neurons in the human eye. Invest Ophthalmol Vis Sci 35:592–599
Flugel-Koch C, May CA, Lutjen-Drecoll E (1996) Presence of a contractile cell network in the human choroid. Ophthalmologica 210:296–302
Haefeli WE, Bargetzi MJ, Starnes HF, Blashke TR, Hoffman BB (1993) Evidence for activation of the sympathetic nervous system by recombinant human interleukin-1 beta in humans. J Immunother 13:136–140
Hayreh SS (2004) Posterior ciliary artery circulation in health and disease. The Weisenfeld Lecture. Invest Ophthalmol Vis Sci 45:749–757
Iijima H, Ida T, Murayama K, Imai M, Gohdo T (1999) Plasminogen activator inhibitor 1 in central serous chorioretinopathy. Am J Ophthalmol 127:477–488
Ivert L, Kjeldbye H, Gouras P (2002) Long term effects of short term retinal bleb detachments in rabbits. Graefes Arch Clin Exp Ophthalmol 240:232–237
Ivert L, Kong J, Gouras P (2003) Changes in the choroidal circulation of rabbit following RPE removal. Graefes Arch Clin Exp Ophthalmol 241:656–666
Kitaya N, Nagaoka T, Hikichi T, Sugawara R, Fukui K, Ishiko S, Yoshida A (2003) Features of abnormal choroidal circulation in central serous chorioretinopathy. Br J Ophthalmol 87:709–712
Luscher TF, Yang Z, Tschudi M, von Segesser L, Stulz P, Boulanger C, Siebenmann R, Turina M, Buhler FR (1990) Interaction between endothelin-1 and endothelium-derived relaxing factor in human arteries and veins. Circ Res 66:1088–1094
May CA, Horneber M, Lutjen-Drecoll E (1996) Quantitative and morphological changes of the choroid vasculature in RCS rats and their congenic controls. Exp Eye Res 63:75–84
Nilsson SFE, Linders J, Bill A (1985) Characteristics of uveal vasodilation produced by facial nerve stimulation in monkeys, cats, and rabbits. Exp Eye Res 40:841–852
Piccolino FC, Borgia L, Zinicola E, Zingirian M (1995) Indocyanine green angiographic findings in central serous chorioretinopathy. Eye 9:324–332
Prunte C, Flammer J (1996) Choroidal capillary and venous congestion in central serous chorioretinopathy. Am J Ophthalmol 121:26–34
Sears M (1960) Miosis and intraocular pressure changes during manometry. Arch Ophthalmol 63:707–714
Schneider U, Inhoffen W, Gelisken F (2003) Indocyanine green angiography in a case of recurrent posterior acute multifocal placoid pigment epitheliopathy. Acta Ophthalmol Scand 81:72–75
Torczynski E, Tso MO (1976) The architecture of the choriocapillaris at the posterior pole. Am J Ophthalmol 81:428–440
Trivino A, de Hoz B, Rojas JJ, Salazar JJ, Ramirez AI, Ramirez JM (2005) NPY and TH innervation in human choroidal whole-mounts. Histol Histopathol 20:302–393
Wilson CA, Choromokos EA, Sheppard R (1988) Acute posterior multifocal placoid pigment epitheliopathy. Brit J Ophthalmol 73:985–990
Acknowledgement
We thank Research to Prevent Blindness, Inc. for support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ivert, L., Kong, J. & Gouras, P. Alteration in choroidal blood flow produced by local pressure. Graefe's Arch Clin Exp Ophthalmo 244, 1339–1344 (2006). https://doi.org/10.1007/s00417-006-0292-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-006-0292-8