Abstract
Background
Dopamine (DA) storage and release are reduced in form deprivation myopia (FDM) in a wide range of species, from chicks to primates. FDM can be prevented by treatment with DA agonists such as apomorphine, and paradoxically by the dopamine neurotoxin 6-hydroxydopamine. In this study, we increased the DA levels by direct intravitreal DA injections to learn if FDM can be suppressed in a rabbit model.
Methods
Seven-day-old rabbits were deprived of pattern vision by the suturing the right eyelids after natural eye opening. In the first group (FD, n=20), the right eye received form deprivation (FD) alone. In the second group (DA-FD, n=16), the deprived eye of 7-day-old rabbits received four intravitreal injections of 20 μg dopamine every 5 days. In the third group (saline-FD, n=16), the deprived eye received saline injections with the same schedule. The contralateral eye remained untreated as a control. At the end of the 8-week deprivation period, the effects of DA on refractive error, corneal curvature and ocular dimensions were assessed by streak retinoscopy, keratometry and A-scan ultrasonography, respectively.
Results
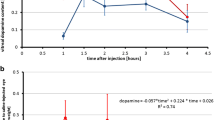
Eight weeks of FDM induced a myopic shift of −2.70±0.87 D (n=20) in treated eyes compared with contralateral eyes. The major structural correlate of the myopia appeared to be elongation of the vitreous chamber (0.7±0.3 mm, n=20) and axial elongation (0.9±0.3 mm, n=20), respectively. Repeated intravitreal injections of DA fully prevented the myopic shift (−0.06±0.37 D), elongation of the vitreous chamber (0.1±0.3 mm, n=16) and axial elongation (0.3±0.2 mm, n=16) due to lid suture, whereas saline injections had slight effect.
Conclusions
FD by suturing eyelids is an effective technique to induce a significant myopic shift, vitreous chamber and axial elongation in rabbits as a model of myopia development. These changes associated with FD were retarded by intravitreal injections of DA.

Similar content being viewed by others
References
Beresford JA, Crewther SG, Kiely PM, Crewther DP (2001) Comparison of refractive state and circumferential morphology of retina, choroid, and sclera in chick models of experimentally induced ametropia. Optom Vis Sci 78:40–49
Bryant MR, Kampmeier J, Er H, Kasetsuwan N, Sanchez-DiMartino D, Shah SS, McDonnell PJ (1999) PRK-induced anisometropia in the rabbit as a model of myopia. Graefe’s Arch Clin Exp Ophthalmol 237:161–165
Cottriall CL, Brew J, Vessey KA, McBrien NA (2001) Diisopropylfluoro-phosphate alters retinal neurotransmitter levels and reduces experimentally-induced myopia. Naunyn Schmiedeberg’s Arch Pharmacol 364:372–382
Feldkaemper M, Diether S, Kleine G, Schaeffel F (1999) Interactions of spatial and luminance information in the retina of chickens during myopia development. Exp Eye Res 68:105–115
Funata M, Tokoro T (1990) Scleral change in experimentally myopic monkeys. Graefe’s Arch Clin Exp Ophthalmol 228:174–179
Hendrickson P, Rosenblum W (1985) Accommodation demand and deprivation in kitten ocular development. Invest Ophthalmol Vis Sci 26:343–349
Guo SS, Sivak JG, Callender MG, Diehl-Jones B (1995) Retinal dopamine and lens induced refractive errors in chicks. Curr Eye Res 14:385–383
Iuvone PM, Tigges M, Fernandes A, Tigges J (1989) Dopamine synthesis and metabolism in rhesus monkey retina: development, aging, and the effects of monocular visual deprivation. Vis Neurosci 2:465–471
Iuvone PM, Tigges M, Stone RA, Lambert S, Laties AM (1991) Effects of apomorphine, a dopamine receptor agonist, on ocular refraction and axial elongation in a primate model of myopia. Invest Ophthalmol Vis Sci 32:1674–1677
Marsh-Tootle WL, Norton TT (1989) Refractive and structural measures of lid-suture myopia in tree shrew. Invest Ophthalmol Vis Sci 30:2245–2257
Maurice DM, Mushin AJ (1966) Production of myopia in rabbits by raised body temperature and increased intraocular pressure. Lancet 2:1160–1162
McBrien NA, Moghaddam HO, New R, Williams LR (1993) Experimental myopia in a diurnal mammal (Sciurus carolinensis) with no accommodative ability. J Physiol 469:427–441
McBrien NA, Norton TT (1992) The development of experimental myopia and ocular component dimensions in monocularly lid-sutured tree shrews (Tupaia belangeri). Vision Res 32:843–852
Megaw PL, Morgan IG, Boelen MK (1997) Dopaminergic behaviour in chicken retina and the effect of form deprivation. Aust N Z J Ophthalmol 25:76–78
Megaw P, Morgan I, Boelen M (2001) Vitreal dihydroxyphenylacetic acid (DOPAC) as an index of retinal dopamine release. J Neurochem 76:1636–1644
Mohan M, Rao VA, Dada VK (1977) Experimental myopia in the rabbit. Exp Eye Res 25:33–38
Nathan J, Crewther SG, Crewther DP, Kiely PM (1984) Effects of retinal image degradation on ocular growth in cats. Invest Ophthalmol Vis Sci 25:1300–1306
Ni J, Smith EL 3rd (1989) Effects of chronic optical defocus on the kitten’s refractive status. Vision Res 29:929–938
Norton TT, McBrien NA (1992) Normal development of refractive state and ocular component dimensions in the tree shrew (Tupaia belangeri). Vision Res 32:833–842
Prince JH (1964) The rabbit in eye research. Thomas, Springfield, IlI
Rohrer B, Spira AW, Stell WK (1993) Apomorphine blocks form-deprivation myopia in chickens by a dopamine D2-receptor mechanism acting in retina or pigmented epithelium. Vis Neurosci 10:447–453
Rohrer B, Tao J, Stell WK (1997) Basic fibroblast growth factor, its high and-low-affinity receptors, and their relationship to form deprivation myopia in the chick. Neuroscience 79:775–787
Schaeffel F, Hagel G, Bartmann M, Kohler K, Zrenner E (1994) 6-Hydroxy dopamine does not affect lens-induced refractive errors but suppresses deprivation myopia. Vision Res 34:143–149
Schaeffel F, Howland HC (1991) Properties of the feedback loops controlling eye growth and refractive state in the chicken. Vision Res 31:717–734
Schmid KL, Wildsoet CF (2004) Inhibitory effects of apomorphine and atropine and their combination on myopia in chicks. Optom Vis Sci 81:137–147
Seko Y, Tanaka Y, Tokoro T (1997) Apomorphine inhibits the growth-stim-ulating effect of retinal pigment epithelium on scleral cells in vitro. Cell Bio Chem Funct 15:191–196
Siegwart JT Jr, Norton TT (2001) Steady state mRNA levels in tree shrew sclera with form deprivation myopia and during recovery. Invest Ophthalmol Vis Sci 42:1153–1159
Smith EL 3rd, Harwerth RS, Crawford ML, von Noorden GK (1987) Observations on the effects of form deprivation on the refractive status of the monkey. Invest Ophthalmol Vis Sci 28:1236–1245
Smith EL 3rd, Hung LF (2000) Form-deprivation myopia in monkeys is a graded phenomenon. Vision Res 40:371–381
Stone RA, Lin T, Iuvone PM, Laties AM (1990) Postnatal control of ocular growth: dopaminergic mechanisms. Ciba Found Symp 155:45–57; discussion 57–62
Stone RA, Lin T, Laties AM, Iuvone PM (1989) Retinal dopamine and form-deprivation myopia. Proc Natl Acad Sci USA 86:704–706
Tokoro T (1970) Experimental myopia in rabbits. Invest Ophthalmol Vis Sci 9:926–934
Verolino M, Nastri G, Sellitti L, Costagliola C (1999) Axial length increase in lid-sutured rabbits. Surv Ophthalmol 44(Suppl 1):s103–s108
Wallman J (1993) Retinal control of eye growth and refraction in progress in retinal research. Pergamon Press, London, 12:133–153
Wallman J, Winawer J (2004) Homeostasis of eye growth and the question of myopia. Neuron 43:447–468
Wildsoet C, Wallman J (1995) Choroidal and scleral mechanisms of compensation for spectacle lenses in chicks. Vision Res 35:1175–1194
Wioland N, Rodolf G, Bonavexture N (1990) Electrooculographic and electroretinographic study in the chicken after dopamine and haloperidol. Doc Ophthalmol 75:175–180
Yinon U (1984) Myopia induction in animals following alteration of the visual input during development: a review. Curr Eye Res 3:677–690
Zhong XW, Ge J, Deng WG, Chen XL, Huang J (2004) Expression of pax-6 in rhesus monkey of optical defocus induced myopia and form deprivation myopia. Chin Med J (Engl) 117:722–726
Zhong XW, Ge J, Nie HH, Chen XL, Huang J, Liu L (2004) Effects of photorefractive keratectomy-induced defocus on emmetropization of infant rhesus monkeys. Invest Ophthalmol Vis Sci 45:3806–3811
Acknowledgements
We would like to thank Dr. Jojo W. Du at SUNY State College of Optometry, for her through reading and constructive revision of the manuscript. Her invaluable comments and suggestions on this paper are gratefully acknowledged. This study was supported by the National Nature Science Foundation of China (30200306), the Science and Technology Foundation of Guang Dong Province of China (2003C32715) and the Science and Technology Foundation of Guang Zhou City of China (2002J1-C0381).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gao, Q., Liu, Q., Ma, P. et al. Effects of direct intravitreal dopamine injections on the development of lid-suture induced myopia in rabbits. Graefe's Arch Clin Exp Ophthalmo 244, 1329–1335 (2006). https://doi.org/10.1007/s00417-006-0254-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-006-0254-1