Abstract
Background
Multiple sclerosis (MS) is associated with regulatory T cells (Tregs) insufficiency while low-dose interleukin-2 (IL2LD) activates Tregs and reduces disease activity in autoimmune diseases.
Methods
We aimed at addressing whether IL2LD improved Tregs from MS patients. MS-IL2 was a single-center double-blind phase-2 study. Thirty patients (mean [SD] age 36.8 years [8.3], 16 female) with relapsing–remitting MS with new MRI lesions within 6 months before inclusion were randomly assigned in a 1:1 ratio to placebo or IL-2 at 1 million IU, daily for 5 days and then fortnightly for 6 months. The primary endpoint was change in Tregs at day-5.
Results
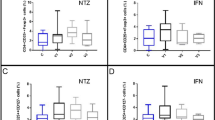
Unlike previous trials of IL2LD in more than 20 different autoimmune diseases, Tregs were not expanded at day-5 in IL2LD group, but only at day-15 (median [IQR] fold change from baseline: 1.26 [1.21–1.33] in IL2LD group; 1.01 [0.95–1.05] in placebo group, p < 0.001). At day-5, however, Tregs had acquired an activated phenotype (fold change of CD25 expression in Tregs: 2.17 [1.70–3.55] in IL2LD versus 0.97 [0.86–1.28] in placebo group, p < 0.0001). Regulator/effector T cells ratio remained elevated throughout treatment period in the IL2LD group (p < 0.001). Number of new active brain lesions and of relapses tended to be reduced in IL2LD treated patients, but the difference did not reach significance in this trial not powered to detect clinical efficacy.
Conclusion
The effect of IL2LD on Tregs in MS patients was modest and delayed, compared to other auto-immune diseases. This, together with findings that Tregs improve remyelination in MS models and recent reports of IL2LD efficacy in amyotrophic lateral sclerosis, warrants larger studies of IL2LD in MS, notably with increased dosages and/or modified modalities of administration.
Trial registration information
ClinicalTrials.gov: NCT02424396; EU Clinical trials Register: 2014-000088-42.





Similar content being viewed by others
Data availability
A deidentified dataset will be archived and upon request will be available from Assistance Publique Hôpitaux de Paris.
References
Baecher-Allan C, Kaskow BJ, Weiner HL (2018) Multiple sclerosis: mechanisms and immunotherapy. Neuron 97:742–768. https://doi.org/10.1016/j.neuron.2018.01.021
Viglietta V, Baecher-Allan C, Weiner HL, Hafler DA (2004) Loss of functional suppression by CD4+CD25+ regulatory T cells in patients with multiple sclerosis. J Exp Med 199:971–979. https://doi.org/10.1084/jem.20031579
Dominguez-Villar M, Baecher-Allan CM, Hafler DA (2011) Identification of T helper type 1-like, Foxp3+ regulatory T cells in human autoimmune disease. Nat Med 17:673–675. https://doi.org/10.1038/nm.2389
Goverman JM (2021) Regulatory T cells in multiple sclerosis. N Engl J Med 384:578–580. https://doi.org/10.1056/NEJMcibr2033544
Bar-Or A, Li R (2021) Cellular immunology of relapsing multiple sclerosis: interactions, checks, and balances. Lancet Neurol 20:470–483. https://doi.org/10.1016/S1474-4422(21)00063-6
Pompura SL, Wagner A, Kitz A et al (2021) Oleic acid restores suppressive defects in tissue-resident FOXP3 Tregs from patients with multiple sclerosis. J Clin Invest 131:e138519. https://doi.org/10.1172/JCI138519
Duffy SS, Keating BA, Moalem-Taylor G (2019) Adoptive transfer of regulatory T cells as a promising immunotherapy for the treatment of multiple sclerosis. Front Neurosci 13:1107. https://doi.org/10.3389/fnins.2019.01107
Sharabi A, Tsokos MG, Ding Y et al (2018) Regulatory T cells in the treatment of disease. Nat Rev Drug Discov. https://doi.org/10.1038/nrd.2018.148
Klatzmann D, Abbas AK (2015) The promise of low-dose interleukin-2 therapy for autoimmune and inflammatory diseases. Nat Rev Immunol 15:283–294. https://doi.org/10.1038/nri3823
Abbas AK, Trotta E, Simeonov RD et al (2018) Revisiting IL-2: Biology and therapeutic prospects. Sci Immunol 3:eaat1482. https://doi.org/10.1126/sciimmunol.aat1482
Frisullo G, Nociti V, Iorio R et al (2009) Regulatory T cells fail to suppress CD4+T-bet+ T cells in relapsing multiple sclerosis patients. Immunology 127:418–428. https://doi.org/10.1111/j.1365-2567.2008.02963.x
Mexhitaj I, Nyirenda MH, Li R et al (2019) Abnormal effector and regulatory T cell subsets in paediatric-onset multiple sclerosis. Brain 142:617–632. https://doi.org/10.1093/brain/awz017
Campbell DJ, Ziegler SF (2007) FOXP3 modifies the phenotypic and functional properties of regulatory T cells. Nat Rev Immunol 7:305–310. https://doi.org/10.1038/nri2061
Huan J, Culbertson N, Spencer L et al (2005) Decreased FOXP3 levels in multiple sclerosis patients. J Neurosci Res 81:45–52. https://doi.org/10.1002/jnr.20522
Venken K, Hellings N, Hensen K et al (2006) Secondary progressive in contrast to relapsing-remitting multiple sclerosis patients show a normal CD4+CD25+ regulatory T-cell function and FOXP3 expression. J Neurosci Res 83:1432–1446. https://doi.org/10.1002/jnr.20852
Cerosaletti K, Schneider A, Schwedhelm K et al (2013) Multiple autoimmune-associated variants confer decreased IL-2R signaling in CD4+ CD25(hi) T cells of type 1 diabetic and multiple sclerosis patients. PLoS ONE 8:e83811. https://doi.org/10.1371/journal.pone.0083811
Lifshitz GV, Zhdanov DD, Lokhonina AV et al (2016) Ex vivo expanded regulatory T cells CD4+CD25+FoxP3+CD127Low develop strong immunosuppressive activity in patients with remitting-relapsing multiple sclerosis. Autoimmunity 49(6):388–396
Lubetzki C, Zalc B, Williams A et al (2020) Remyelination in multiple sclerosis: from basic science to clinical translation. Lancet Neurol 19:678–688. https://doi.org/10.1016/S1474-4422(20)30140-X
Burzyn D, Kuswanto W, Kolodin D et al (2013) A special population of regulatory T cells potentiates muscle repair. Cell 155:1282–1295. https://doi.org/10.1016/j.cell.2013.10.054
Ito M, Komai K, Mise-Omata S et al (2019) Brain regulatory T cells suppress astrogliosis and potentiate neurological recovery. Nature 565:246–250. https://doi.org/10.1038/s41586-018-0824-5
Dombrowski Y, O’Hagan T, Dittmer M et al (2017) Regulatory T cells promote myelin regeneration in the central nervous system. Nat Neurosci 20:674–680. https://doi.org/10.1038/nn.4528
McIntyre LL, Greilach SA, Othy S et al (2020) Regulatory T cells promote remyelination in the murine experimental autoimmune encephalomyelitis model of multiple sclerosis following human neural stem cell transplant. Neurobiol Dis 140:104868. https://doi.org/10.1016/j.nbd.2020.104868
De Paula PA, Schmidt A, Zhang A-H et al (2020) Engineered regulatory T cells expressing myelin-specific chimeric antigen receptors suppress EAE progression. Cell Immunol 358:104222. https://doi.org/10.1016/j.cellimm.2020.104222
Yu A, Snowhite I, Vendrame F et al (2015) Selective IL-2 responsiveness of regulatory T cells through multiple intrinsic mechanisms supports the use of low-dose IL-2 therapy in Type 1 diabetes. Diabetes 64:2172–2183. https://doi.org/10.2337/db14-1322
Graßhoff H, Comdühr S, Monne LR et al (2021) Low-dose IL-2 therapy in autoimmune and rheumatic diseases. Front Immunol. https://doi.org/10.3389/fimmu.2021.648408
He J, Zhang R, Shao M et al (2020) Efficacy and safety of low-dose IL-2 in the treatment of systemic lupus erythematosus: a randomised, double-blind, placebo-controlled trial. Ann Rheum Dis 79(1):141–149
Humrich JY, Cacoub P, Rosenzwajg M et al (2022) Low-dose interleukin-2 therapy in active systemic lupus erythematosus (LUPIL-2): a multicentre, double-blind, randomised and placebo-controlled phase II trial. Ann Rheum Dis 81:1685–1694. https://doi.org/10.1136/ard-2022-222501
Polman CH, Reingold SC, Banwell B et al (2011) Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol 69:292–302. https://doi.org/10.1002/ana.22366
Thompson AJ, Banwell BL, Barkhof F et al (2018) Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol 17:162–173. https://doi.org/10.1016/S1474-4422(17)30470-2
Churlaud G, Abbara C, Vinot P-A et al (2018) Pharmacodynamics of regulatory T cells in mice and humans treated with low-dose IL-2. J Allergy Clin Immunol 142:1344-1346.e3. https://doi.org/10.1016/j.jaci.2018.06.006
Noether GE (1987) Sample size determination for some common nonparametric tests. J Am Stat Assoc 82:645–647. https://doi.org/10.1080/01621459.1987.10478478
Hartemann A, Bensimon G, Payan CA et al (2013) Low-dose interleukin 2 in patients with type 1 diabetes: a phase 1/2 randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol 1:295–305. https://doi.org/10.1016/S2213-8587(13)70113-X
Van Gool F, Molofsky AB, Morar MM et al (2014) Interleukin-5-producing group 2 innate lymphoid cells control eosinophilia induced by interleukin-2 therapy. Blood 124:3572–3576. https://doi.org/10.1182/blood-2014-07-587493
Camu W, Mickunas M, Veyrune JL et al (2020) Repeated 5-day cycles of low dose aldesleukin in amyotrophic lateral sclerosis (IMODALS): a phase 2a randomised, double-blind, placebo-controlled trial. EBioMedicine 59:102844
Whangbo JS, Kim HT, Mirkovic N et al (2019) Dose-escalated interleukin-2 therapy for refractory chronic graft-versus-host disease in adults and children. Blood Adv 3:2550–2561. https://doi.org/10.1182/bloodadvances.2019000631
Koreth J, Matsuoka K, Kim HT et al (2011) Interleukin-2 and regulatory T cells in graft-versus-host disease. N Engl J Med 365:2055–2066. https://doi.org/10.1056/NEJMoa1108188
Alves S, Churlaud G, Audrain M et al (2017) Interleukin-2 improves amyloid pathology, synaptic failure and memory in Alzheimer’s disease mice. Brain 140:826–842. https://doi.org/10.1093/brain/aww330
Rosenzwajg M, Lorenzon R, Cacoub P et al (2019) Immunological and clinical effects of low-dose interleukin-2 across 11 autoimmune diseases in a single, open clinical trial. Ann Rheum Dis 78:209–217. https://doi.org/10.1136/annrheumdis-2018-214229
de la Vega GN, Dittmer M, Dombrowski Y, Fitzgerald DC (2019) Regenerating CNS myelin: Emerging roles of regulatory T cells and CCN proteins. Neurochem Int 130:1049. https://doi.org/10.1016/j.neuint.2018.11.024
de la Vega GN, Penalva R, Dittmer M et al (2020) Dynamic CCN3 expression in the murine CNS does not confer essential roles in myelination or remyelination. Proc Natl Acad Sci USA 117:18018–18028. https://doi.org/10.1073/pnas.1922089117
Yshii L, Pasciuto E, Bielefeld P et al (2022) Astrocyte-targeted gene delivery of interleukin 2 specifically increases brain-resident regulatory T cell numbers and protects against pathological neuroinflammation. Nat Immunol 23:878–891. https://doi.org/10.1038/s41590-022-01208-z
Acknowledgements
We thank the personnel from the Biotherapy Department, and particularly Michele Barbié, Nathalie Ferry and Cornelia Degbé, for their excellent work. We thank the personnel from the Neuroscience Clinical Investigation Center, and particularly research nurses Carine Lefort and Marie-France Le Labousse. We also thank Olivia Tran for the monitoring of the study. We thank all patients for their commitment in participating in this study.
Funding
This study was funded by a grant from the AFM/ARSEP. It was performed in the Clinical Investigation Centers of the Pitié-Salpêtrière Hospital which receive recurrent support from the DGOS and INSERM. Immune investigations were supported by an ANR grant iMAP (ANR-16-RHUS-0001). ILTOO pharma provided the investigational drug and the placebo.
Author information
Authors and Affiliations
Contributions
DK contributed to the concept of the study. CLo, MR, EV, JCC, CLu and DK contributed to the design of the study. All authors contributed to the acquisition, analysis, or interpretation of data. CLo, MR, CLu and DK drafted the first version of the manuscript. All authors contributed to critical revision of the manuscript for important intellectual content. Statistical analysis was performed by EV. CLu and DK had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Prof Alain Créange, Prof Jean Pelletier, Prof Sandra Vukusic, Dr Silvy Laporte were members of the Data Safety Monitoring Board.
Corresponding author
Ethics declarations
Conflicts of interest
DK and MR are inventors of a patent application owned by their institutions that protects the use of low-dose IL2 in autoimmune diseases. This patent has been licensed to ILTOO pharma in which they hold shares.
Ethical approval
The study was approved by the institutional review board of Pitié-Salpêtrière Hospital (CPP Ile de France VI) and was conducted in accordance with the Declaration of Helsinki and good clinical practice guidelines. The study protocol is registered on ClinicalTrials.gov (NCT02424396). Written, informed consent was obtained from all participants before enrolment in the study.
Ethical standard
The study was approved by the institutional review board of Pitié-Salpêtrière Hospital (CPP Ile de France VI) and was conducted in accordance with the Declaration of Helsinki and good clinical practice guidelines. The study protocol is registered on ClinicalTrials.gov (NCT02424396).
Informed consent
Written, informed consent was obtained from all participants before enrolment in the study.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Louapre, C., Rosenzwajg, M., Golse, M. et al. A randomized double-blind placebo-controlled trial of low-dose interleukin-2 in relapsing–remitting multiple sclerosis. J Neurol 270, 4403–4414 (2023). https://doi.org/10.1007/s00415-023-11690-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-023-11690-6