Abstract
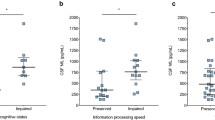
Cognitive impairment (CI) is a frequent and disabling symptom in Multiple Sclerosis (MS). Axonal damage may contribute to CI development from early stages. Nevertheless, no biomarkers are at the moment available to track CI in MS patients. We aimed to explore the correlation of cerebrospinal fluid (CSF) axonal biomarkers, in particular: light-chain neurofilaments (NFL), Tau, and Beta-amyloid protein (Abeta) in MS patients with CI at the diagnosis. 62 newly diagnosed MS patients were enrolled, and cognition was evaluated using the Brief International Cognitive Assessment for MS (BICAMS) battery. CSF NFL, Abeta, and Tau levels were determined with commercial ELISA. Patients with CI (45.1%) did not differ for demographic, clinical, and MRI characteristics (except for lower educational level), but they displayed greater neurodegeneration, exhibiting higher mean CSF Tau protein (162.1 ± 52.96 pg/ml versus 132.2 ± 63.86 pg/ml p:0.03). No differences were observed for Abeta and NFL. The number of impaired tests and Tau were significantly correlated (r:0.32 p:0.01). Tau was higher in particular in patients with slowed information processing speed (IPS) (p:0.006) and a linear regression analysis accounting for EDSS, MRI, and MS subtype confirmed Tau as a weak predictor of IPS and cognitive impairment. In conclusion, CI has an important burden on the quality of life of MS patients and should be looked for even at diagnosis. Axonal damage biomarkers, and in particular Tau, seem to reflect cognition impairment in the early stages.

Similar content being viewed by others
Data availability statement
Data are available upon reasonable request.
References
Benedict RHB, Amato MP, DeLuca J, Geurts JJG (2020) Cognitive impairment in multiple sclerosis: clinical management, MRI, and therapeutic avenues. Lancet Neurol 19(10):860–871. https://doi.org/10.1016/s1474-4422(20)30277-5
Ruet A, Deloire M, Charré-Morin J, Hamel D, Brochet B (2013) Cognitive impairment differs between primary progressive and relapsing-remitting MS. Neurology 80(16):1501–1508. https://doi.org/10.1212/WNL.0b013e31828cf82f
Amato MP, Hakiki B, Goretti B, Rossi F, Stromillo ML, Giorgio A, Roscio M, Ghezzi A, Guidi L, Bartolozzi ML, Portaccio E, De Stefano N (2012) Association of MRI metrics and cognitive impairment in radiologically isolated syndromes. Neurology 78(5):309–314. https://doi.org/10.1212/WNL.0b013e31824528c9
Kalb R, Beier M, Benedict RH, Charvet L, Costello K, Feinstein A, Gingold J, Goverover Y, Halper J, Harris C, Kostich L, Krupp L, Lathi E, LaRocca N, Thrower B, DeLuca J (2018) Recommendations for cognitive screening and management in multiple sclerosis care. Mult Scler 24(13):1665–1680. https://doi.org/10.1177/1352458518803785
Portaccio E, Goretti B, Zipoli V, Siracusa G, Sorbi S, Amato MP (2009) A short version of Rao’s brief repeatable battery as a screening tool for cognitive impairment in multiple sclerosis. Clin Neuropsychol 23(2):268–275. https://doi.org/10.1080/13854040801992815
Rocca MA, Amato MP, De Stefano N, Enzinger C, Geurts JJ, Penner IK, Rovira A, Sumowski JF, Valsasina P, Filippi M (2015) Clinical and imaging assessment of cognitive dysfunction in multiple sclerosis. Lancet Neurol 14(3):302–317. https://doi.org/10.1016/s1474-4422(14)70250-9
Grossi P, Portaccio E, Bellomi F, Bianchi V, Cilia S, Falautano M, Goretti B, Pietrolongo E, Viterbo RG, Messmer Uccelli M (2020) The minimal neuropsychological assessment of MS patients (MACFIMS): normative data of the Italian population. Neurol Sci 41(6):1489–1496. https://doi.org/10.1007/s10072-020-04251-6
Goretti B, Niccolai C, Hakiki B, Sturchio A, Falautano M, Minacapelli E, Martinelli V, Incerti C, Nocentini U, Murgia M, Fenu G, Cocco E, Marrosu MG, Garofalo E, Ambra FI, Maddestra M, Consalvo M, Viterbo RG, Trojano M, Losignore NA, Zimatore GB, Pietrolongo E, Lugaresi A, Langdon D, Portaccio E, Amato MP (2014) The Brief International Cognitive Assessment for Multiple Sclerosis (BICAMS): normative values with gender, age and education corrections in the Italian population. BMC Neurol 14:171. https://doi.org/10.1186/s12883-014-0171-6
Gaetani L, Salvadori N, Chipi E, Gentili L, Borrelli A, Parnetti L, Di Filippo M (2021) Cognitive impairment in multiple sclerosis: lessons from cerebrospinal fluid biomarkers. Neural Regen Res 16(1):36–42. https://doi.org/10.4103/1673-5374.286949
Petracca M, Pontillo G, Moccia M, Carotenuto A, Cocozza S, Lanzillo R, Brunetti A, Brescia Morra V (2021) Neuroimaging correlates of cognitive dysfunction in adults with multiple sclerosis. Brain Sci. https://doi.org/10.3390/brainsci11030346
Aktas O, Renner A, Huss A, Filser M, Baetge S, Stute N, Gasis M, Lepka K, Goebels N, Senel M, Graf J, Enzinger C, Pinter D, Antoch G, Turowski B, Hartung HP, Albrecht P, Otto M, Tumani H, Penner IK (2020) Serum neurofilament light chain: no clear relation to cognition and neuropsychiatric symptoms in stable MS. Neurol Neuroimmunol Neuroinflamm. https://doi.org/10.1212/nxi.0000000000000885
Giedraitiene N, Drukteiniene E, Kizlaitiene R, Cimbalas A, Asoklis R, Kaubrys G (2021) Cognitive decline in multiple sclerosis is related to the progression of retinal atrophy and presence of oligoclonal bands: a 5-year follow-up study. Front Neurol 12:678735. https://doi.org/10.3389/fneur.2021.678735
Virgilio E, Vecchio D, Crespi I, Serino R, Cantello R, Dianzani U, Comi C (2021) Cerebrospinal Tau levels as a predictor of early disability in Multiple Sclerosis. Multiple Scler Relat Disord. https://doi.org/10.1016/j.msard.2021.103231
Mori F, Rossi S, Sancesario G, Codeca C, Mataluni G, Monteleone F, Buttari F, Kusayanagi H, Castelli M, Motta C, Studer V, Bernardi G, Koch G, Bernardini S, Centonze D (2011) Cognitive and cortical plasticity deficits correlate with altered amyloid-beta CSF levels in multiple sclerosis. Neuropsychopharmacology 36(3):559–568. https://doi.org/10.1038/npp.2010.187
Kuhle J, Kropshofer H, Haering DA, Kundu U, Meinert R, Barro C, Dahlke F, Tomic D, Leppert D, Kappos L (2019) Blood neurofilament light chain as a biomarker of MS disease activity and treatment response. Neurology 92(10):e1007–e1015. https://doi.org/10.1212/wnl.0000000000007032
Gaetani L, Salvadori N, Lisetti V, Eusebi P, Mancini A, Gentili L, Borrelli A, Portaccio E, Sarchielli P, Blennow K, Zetterberg H, Parnetti L, Calabresi P, Di Filippo M (2019) Cerebrospinal fluid neurofilament light chain tracks cognitive impairment in multiple sclerosis. J Neurol 266(9):2157–2163. https://doi.org/10.1007/s00415-019-09398-7
Quintana E, Coll C, Salavedra-Pont J, Muñoz-San Martín M, Robles-Cedeño R, Tomàs-Roig J, Buxó M, Matute-Blanch C, Villar LM, Montalban X, Comabella M, Perkal H, Gich J, Ramió-Torrentà L (2018) Cognitive impairment in early stages of multiple sclerosis is associated with high cerebrospinal fluid levels of chitinase 3-like 1 and neurofilament light chain. Eur J Neurol 25(9):1189–1191. https://doi.org/10.1111/ene.13687
Modvig S, Degn M, Roed H, Sørensen TL, Larsson HB, Langkilde AR, Frederiksen JL, Sellebjerg F (2015) Cerebrospinal fluid levels of chitinase 3-like 1 and neurofilament light chain predict multiple sclerosis development and disability after optic neuritis. Mult Scler 21(14):1761–1770. https://doi.org/10.1177/1352458515574148
Kalatha T, Arnaoutoglou M, Koukoulidis T, Hatzifilippou E, Bouras E, Baloyannis S, Koutsouraki E (2019) Does cognitive dysfunction correlate with neurofilament light polypeptide levels in the CSF of patients with multiple sclerosis? J Int Med Res 47(5):2187–2198. https://doi.org/10.1177/0300060519840550
Cruz-Gomez ÁJ, Forero L, Lozano-Soto E, Cano-Cano F, Sanmartino F, Rashid-López R, Paz-Expósito J, Gómez Ramirez JD, Espinosa-Rosso R, González-Rosa JJ (2021) Cortical thickness and serum NfL explain cognitive dysfunction in newly diagnosed patients with multiple sclerosis. Neurology - Neuroimmunology Neuroinflammation 8(6):e1074. https://doi.org/10.1212/nxi.0000000000001074
Friedova L, Motyl J, Srpova B, Oechtering J, Barro C, Vodehnalova K, Andelova M, Noskova L, Fialová L, Havrdova EK, Horakova D, Benedict RH, Kuhle J, Uher T (2020) The weak association between neurofilament levels at multiple sclerosis onset and cognitive performance after 9 years. Mult Scler Relat Disord 46:102534. https://doi.org/10.1016/j.msard.2020.102534
Jakimovski D, Zivadinov R, Ramanthan M, Hagemeier J, Weinstock-Guttman B, Tomic D, Kropshofer H, Fuchs TA, Barro C, Leppert D, Yaldizli Ö, Kuhle J, Benedict RH (2020) Serum neurofilament light chain level associations with clinical and cognitive performance in multiple sclerosis: a longitudinal retrospective 5-year study. Mult Scler 26(13):1670–1681. https://doi.org/10.1177/1352458519881428
Mattioli F, Bellomi F, Stampatori C, Mariotto S, Ferrari S, Monaco S, Mancinelli C, Capra R (2020) Longitudinal serum neurofilament light chain (sNfL) concentration relates to cognitive function in multiple sclerosis patients. J Neurol 267(8):2245–2251. https://doi.org/10.1007/s00415-020-09832-1
Polman CH, Reingold SC, Banwell B, Clanet M, Cohen JA, Filippi M, Fujihara K, Havrdova E, Hutchinson M, Kappos L, Lublin FD, Montalban X, O’Connor P, Sandberg-Wollheim M, Thompson AJ, Waubant E, Weinshenker B, Wolinsky JS (2011) Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol 69(2):292–302. https://doi.org/10.1002/ana.22366
Thompson AJ, Banwell BL, Barkhof F, Carroll WM, Coetzee T, Comi G, Correale J, Fazekas F, Filippi M, Freedman MS, Fujihara K, Galetta SL, Hartung HP, Kappos L, Lublin FD, Marrie RA, Miller AE, Miller DH, Montalban X, Mowry EM, Sorensen PS, Tintoré M, Traboulsee AL, Trojano M, Uitdehaag BMJ, Vukusic S, Waubant E, Weinshenker BG, Reingold SC, Cohen JA (2018) Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol 17(2):162–173. https://doi.org/10.1016/s1474-4422(17)30470-2
Filippi M, Rocca MA, Bastianello S, Comi G, Gallo P, Gallucci M, Ghezzi A, Marrosu MG, Minonzio G, Pantano P, Pozzilli C, Tedeschi G, Trojano M, Falini A, De Stefano N, Neuroimaging, Neurology MSSGotISo, Functional Neuroradiology Section of the Italian Association of N (2013) Guidelines from The Italian Neurological and Neuroradiological Societies for the use of magnetic resonance imaging in daily life clinical practice of multiple sclerosis patients. Neurol Sci 34(12):2085–2093. https://doi.org/10.1007/s10072-013-1485-7
Cree BAC, Bowen JD, Hartung HP, Vermersch P, Hughes B, Damian D, Hyvert Y, Dangond F, Galazka A, Grosso M, Jones DL, Leist TP (2021) Subgroup analysis of clinical and MRI outcomes in participants with a first clinical demyelinating event at risk of multiple sclerosis in the ORACLE-MS study. Mult Scler Relat Disord 49:102695. https://doi.org/10.1016/j.msard.2020.102695
Crespi I, Vecchio D, Serino R, Saliva E, Virgilio E, Sulas MG, Bellomo G, Dianzani U, Cantello R, Comi C (2019) K index is a reliable marker of intrathecal synthesis, and an alternative to igg index in multiple sclerosis diagnostic work-up. J Clin Med. https://doi.org/10.3390/jcm8040446
Vecchio D, Bellomo G, Serino R, Virgilio E, Lamonaca M, Dianzani U, Cantello R, Comi C, Crespi I (2020) Intrathecal kappa free light chains as markers for multiple sclerosis. Sci Rep 10(1):20329. https://doi.org/10.1038/s41598-020-77029-7
Vecchio D, Crespi I, Virgilio E, Naldi P, Campisi MP, Serino R, Dianzani U, Bellomo G, Cantello R, Comi C (2019) Kappa free light chains could predict early disease course in multiple sclerosis. Mult Scler Relat Disord 30:81–84. https://doi.org/10.1016/j.msard.2019.02.001
Sacco R, Santangelo G, Stamenova S, Bisecco A, Bonavita S, Lavorgna L, Trojano L, D’Ambrosio A, Tedeschi G, Gallo A (2016) Psychometric properties and validity of beck depression inventory II in multiple sclerosis. Eur J Neurol 23(4):744–750. https://doi.org/10.1111/ene.12932
Brochet B, Ruet A (2019) Cognitive impairment in multiple sclerosis with regards to disease duration and clinical phenotypes. Front Neurol 10:261. https://doi.org/10.3389/fneur.2019.00261
Sadigh-Eteghad S, Abbasi Garravnd N, Feizollahi M, Talebi M (2021) The expanded disability status scale score and demographic indexes are correlated with the severity of cognitive impairment in multiple sclerosis patients. J Clin Neurol 17(1):113–120. https://doi.org/10.3988/jcn.2021.17.1.113
Arce Rentería M, Vonk JMJ, Felix G, Avila JF, Zahodne LB, Dalchand E, Frazer KM, Martinez MN, Shouel HL, Manly JJ (2019) Illiteracy, dementia risk, and cognitive trajectories among older adults with low education. Neurology 93(24):e2247–e2256. https://doi.org/10.1212/wnl.0000000000008587
Di Filippo M, Portaccio E, Mancini A, Calabresi P (2018) Multiple sclerosis and cognition: synaptic failure and network dysfunction. Nat Rev Neurosci 19(10):599–609. https://doi.org/10.1038/s41583-018-0053-9
Meijer KA, van Geest Q, Eijlers AJC, Geurts JJG, Schoonheim MM, Hulst HE (2018) Is impaired information processing speed a matter of structural or functional damage in MS? Neuroimage Clin 20:844–850. https://doi.org/10.1016/j.nicl.2018.09.021
Frederiksen J, Kristensen K, Bahl JM, Christiansen M (2012) Tau protein: a possible prognostic factor in optic neuritis and multiple sclerosis. Mult Scler 18(5):592–599. https://doi.org/10.1177/1352458511424588
Hein Nee Maier K, Kohler A, Diem R, Sattler MB, Demmer I, Lange P, Bahr M, Otto M (2008) Biological markers for axonal degeneration in CSF and blood of patients with the first event indicative for multiple sclerosis. Neurosci Lett 436(1):72–76. https://doi.org/10.1016/j.neulet.2008.02.064
Jaworski J, Psujek M, Janczarek M, Szczerbo-Trojanowska M, Bartosik-Psujek H (2012) Total-tau in cerebrospinal fluid of patients with multiple sclerosis decreases in secondary progressive stage of disease and reflects degree of brain atrophy. Ups J Med Sci 117(3):284–292. https://doi.org/10.3109/03009734.2012.669423
Jiménez-Jiménez FJ, Zurdo JM, Hernanz A, Medina-Acebrón S, de Bustos F, Barcenilla B, Sayed Y, Ayuso-Peralta L (2002) Tau protein concentrations in cerebrospinal fluid of patients with multiple sclerosis. Acta Neurol Scand 106(6):351–354. https://doi.org/10.1034/j.1600-0404.2002.01370.x
Kapaki E, Paraskevas GP, Michalopoulou M, Kilidireas K (2000) Increased cerebrospinal fluid tau protein in multiple sclerosis. Eur Neurol 43(4):228–232. https://doi.org/10.1159/000008181
Cortese M, Munger KL, Martínez-Lapiscina EH, Barro C, Edan G, Freedman MS, Hartung HP, Montalbán X, Foley FW, Penner IK, Hemmer B, Fox EJ, Schippling S, Wicklein EM, Kappos L, Kuhle J, Ascherio A (2020) Vitamin D, smoking, EBV, and long-term cognitive performance in MS: 11-year follow-up of BENEFIT. Neurology 94(18):e1950–e1960. https://doi.org/10.1212/wnl.0000000000009371
Acknowledgements
None.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Contributions
Conceptualization, EV, DV and CC; methodology C.C; formal analysis, EV, IC, CP; investigation, EV, DV, PB, GG; resources, CC; data curation, E.V, D.V; writing—original draft preparation, EV; writing—review and editing, D.V, C.C., U.D, R.C; supervision, C.C, U.D, R.C. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest.
Institutional review board statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Ethics Committee of AOU Ospedale Maggiore della Carità di Novara (reference no: CE 190/19).
Informed consent statement
Informed consent was obtained from all subjects involved in the study.
Rights and permissions
About this article
Cite this article
Virgilio, E., Vecchio, D., Crespi, I. et al. Cerebrospinal fluid biomarkers and cognitive functions at multiple sclerosis diagnosis. J Neurol 269, 3249–3257 (2022). https://doi.org/10.1007/s00415-021-10945-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-021-10945-4