Abstract
Background
Patient-centered assessments have attracted increasing attention in the last decade in clinics and research. The purpose of this study was to examine the association between patients’ satisfaction with symptoms and several disease-specific and generic outcome measures in 100 patients with generalized myasthenia gravis (gMG).
Methods
In this cross-sectional study, patients with gMG followed at the Copenhagen Neuromuscular Center from October 2019 to June 2020 participated in one test. The patients completed commonly used MG-specific outcome measures and generic questionnaires for depression (Major Depression Inventory), comorbidities (Charlson Comorbidity Index), fatigue (Multidimensional Fatigue Inventory), overall health state (EQ-5D-3L), and satisfaction with MG treatment. The analyses were anchored in the Patient Acceptable Symptom State (PASS).
Results
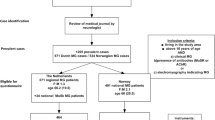
N = 190 patients were screened for the study, and 100 patients were included. One-third of the patients reported dissatisfaction (negative PASS status) with the current symptom state. Increasing MG symptoms, fatigue, depression, low MG-related quality of life, and shorter disease duration were associated with negative PASS status. Age, sex, BMI, MG treatment, and comorbidity did not influence PASS status.
Conclusions
This study shows that dissatisfaction with the current symptom level is high in patients with gMG and that dissatisfaction is associated with disease severity, disease length, depression, fatigue, and lower MG-related quality of life. The results emphasize the importance of a patient-centered approach to MG treatment to optimize patient satisfaction. The PASS question was useful in this study to investigate the causes of symptom dissatisfaction in gMG.
Similar content being viewed by others
References
Gilhus NE (2016) Myasthenia Gravis. N Engl J Med 375(26):2570–2581
Hansen JS, Danielsen DH, Somnier FE, Frøslev T, Jakobsen J, Johnsen SP et al (2016) Mortality in myasthenia gravis: A nationwide population-based follow-up study in Denmark. Muscle Nerve 53(1):73–77
Wijeysundera DN, Johnson SR (2016) How much better is good enough?: Patient-reported outcomes, minimal clinically important differences, and patient acceptable symptom states in perioperative research. Anesthesiology 125(1):7–10
Mendoza M, Tran C, Bril V, Katzberg HD, Barnett C (2020) Patient-acceptable symptom states in myasthenia gravis. Neurology. https://doi.org/10.1212/WNL.0000000000010574
Menon D, Barnett C, Bril V (2020) Comparison of the single simple question and the patient acceptable symptom state in myasthenia gravis. Eur J Neurol 27(11):2286–2291
Tindall RS, Rollins JA, Phillips JT, Greenlee RG, Wells L, Belendiuk G (1987) Preliminary results of a double-blind, randomized, placebo-controlled trial of cyclosporine in myasthenia gravis. N Engl J Med 316(12):719–724
Burns TM, Conaway MR, Cutter GR, Sanders DB (2008) Muscle study. Group construction of an efficient evaluative instrument for myasthenia gravis: the MG composite. Muscle Nerve 38(6):1553–1562
Wolfe GI, Herbelin L, Nations SP, Foster B, Bryan WW, Barohn RJ (1999) Myasthenia gravis activities of daily living profile. Neurology 52(7):1487–1489
Vissing J, Jacob S, Fujita KP, O’Brien F, Howard JF (2020) REGAIN study group “Minimal symptom expression” in patients with acetylcholine receptor antibody-positive refractory generalized myasthenia gravis treated with eculizumab. J Neurol 267(7):1991
Burns TM, Conaway MR, Cutter GR, Sanders DB (2008) Muscle study. Group Less is more, or almost as much: a 15-item quality-of-life instrument for myasthenia gravis. Muscle Nerve 38(2):957–963
Smets EM, Garssen B, Bonke B, De Haes JC (1995) The Multidimensional Fatigue Inventory (MFI) psychometric qualities of an instrument to assess fatigue. J Psychosom Res 39(3):315–325
Mikula P, Nagyova I, Krokavcova M, Vitkova M, Rosenberger J, Szilasiova J et al (2015) The mediating effect of coping on the association between fatigue and quality of life in patients with multiple sclerosis. Psychol Health Med 20(6):653–661
Visser MM, Goodin P, Parsons MW, Lillicrap T, Spratt NJ, Levi CR et al (2019) Modafinil treatment modulates functional connectivity in stroke survivors with severe fatigue. Sci Rep 9(1):9660
Lexell J, Jonasson SB, Brogardh C (2018) Psychometric properties of three fatigue rating scales in individuals with late effects of polio. Ann Rehabil Med 42(5):702–712
Bech P, Rasmussen NA, Olsen LR, Noerholm V, Abildgaard W (2001) The sensitivity and specificity of the major depression inventory, using the present state examination as the index of diagnostic validity. J Affect Disord 66(2–3):159–164
Olsen LR, Jensen DV, Noerholm V, Martiny K, Bech P (2003) The internal and external validity of the major depression inventory in measuring severity of depressive states. Psychol Med 33(2):351–356
Rahbek MA, Mikkelsen EE, Overgaard K, Vinge L, Andersen H, Dalgas U (2017) Exercise in myasthenia gravis: A feasibility study of aerobic and resistance training. Muscle Nerve 56(4):700–709
Paul RH, Cohen RA, Goldstein JM, Gilchrist JM (2000) Fatigue and its impact on patients with myasthenia gravis. Muscle Nerve 23(9):1402–1406
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40(5):373–383
Mouri H, Jo T, Matsui H, Fushimi K, Yasunaga H (2020) Effect of sugammadex on postoperative myasthenic crisis in myasthenia gravis patients: propensity score analysis of a japanese nationwide database. Anesth Analg 130(2):367–373
EQ-5D [Internet]. [cited 2020 Sep 28]. Available from: https://euroqol.org/
Wittrup-Jensen KU, Lauridsen J, Gudex C, Pedersen KM (2009) Generation of a danish TTO value set for EQ-5D health states. Scand J Public Health 37(5):459–466
Gavrilov YV, Alekseeva TM, Kreis OA, Valko PO, Weber KP, Valko Y (2020) Depression in myasthenia gravis: a heterogeneous and intriguing entity. J Neurol 267(6):1802–1811
Alekseeva TM, Gavrilov YV, Kreis OA, Valko PO, Weber KP, Valko Y (2018) Fatigue in patients with myasthenia gravis. J Neurol 265(10):2312–2321
Hoffmann S, Ramm J, Grittner U, Kohler S, Siedler J, Meisel A (2016) Fatigue in myasthenia gravis: risk factors and impact on quality of life. Brain Behav 6(10):e00538
Kittiwatanapaisan W, Gauthier DK, Williams AM, Oh SJ (2003) Fatigue in myasthenia gravis patients. J Neurosci Nurs J Am Assoc Neurosci Nurs 35(2):87–93
Matcham F, Rayner L, Steer S, Hotopf M (2013) The prevalence of depression in rheumatoid arthritis: a systematic review and meta-analysis. Rheumatol Oxf Engl 52(12):2136–2148
Boeschoten RE, Braamse AMJ, Beekman ATF, Cuijpers P, van Oppen P, Dekker J et al (2017) Prevalence of depression and anxiety in multiple sclerosis: A systematic review and meta-analysis. J Neurol Sci 372:331–341
Carstensen B, Rønn PF, Jørgensen ME (2020) Prevalence, incidence and mortality of type 1 and type 2 diabetes in Denmark 1996–2016. BMJ Open Diabetes Res Care. https://doi.org/10.1136/bmjdrc-2019-001071
Ibsen H, Jørgensen T, Jensen GB, Jacobsen IA (2009) Hypertension–prevalence and treatment. Ugeskr Laeger 171(24):1998–2000
Cutter G, Xin H, Aban I, Burns TM, Allman PH, Farzaneh-Far R et al (2019) Cross-sectional analysis of the myasthenia gravis patient registry: disability and treatment. Muscle Nerve 60(6):707–715
Funding
N/A.
Author information
Authors and Affiliations
Contributions
LKA and AJ have full access to all data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: Vissing. Acquisition, analysis, or interpretation of data: All authors. Drafting of the manuscript: All authors. Critical revision of the manuscript for important intellectual content: All authors. Statistical analysis: Andersen. Obtained funding: Vissing. Administrative, technical, or material support: Andersen, Jakobsson. Supervision: Vissing.
Corresponding author
Ethics declarations
Conflicts of interest
Vissing received an unrestricted grant from Argenx to cover salary and patient transport expenses. The company had no influence on study design or in the writing of the manuscript. Andersen, Jakobsson, and Revsbech report no disclosures.
Ethics approval
The study was approved by the ethics committee of the Capital Region of Denmark.
Consent to participate
Informed consent was obtained from all included patients.
Consent for publication
N/A.
Rights and permissions
About this article
Cite this article
Andersen, L.K., Jakobsson, A.S., Revsbech, K.L. et al. Causes of symptom dissatisfaction in patients with generalized myasthenia gravis. J Neurol 269, 3086–3093 (2022). https://doi.org/10.1007/s00415-021-10902-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-021-10902-1