Abstract
Background
Since the declaration of COVID-19 pandemic, several case reports of demyelination of both peripheral and central nervous systems have been published. The association between CNS demyelination and viral infection has long been documented, and this link was recently reported following SARS-CoV-2 infection as well.
Objectives
In this systematic review, we aim to investigate the existing literature on CNS demyelination associated with SARS-CoV-2, and the proposed pathophysiological mechanisms.
Methods
We conducted a systematic review of articles in PubMed, SCOPUS, EMBASE, Cochrane, Google Scholar and Ovid databases, from 1 January 2020 until June 15, 2021. The following keywords were used: “COVID-19”, “SARS-CoV-2”, “demyelination”, “demyelinating disease”, “multiple sclerosis”, “neuromyelitis optica”, and “transverse myelitis”.
Results
A total of 60 articles were included in the final analysis of this systematic review and included 102 patients: 52 (51%) men and 50 (49%) women, with a median age of 46.5 years. The demyelination mimicked a variety of conditions with a picture of encephalitis/encephalomyelitis being the most common. At the same time other patterns were less frequently reported such as MS, NMOSD and even MOGAD. Longitudinally extensive transverse myelitis (LETM) was the most frequently reported pattern of spinal cord involvement.
Conclusion
A growing body of literature has shown an association between SARS‐CoV‐2 infection and the development of different types of CNS demyelination. Although causality cannot readily be inferred, this review may suggest a probable causal relationship, through a para-infectious or post-infectious immune-mediated etiology in COVID-19 patients. This relationship needs to be clarified in future research.
Similar content being viewed by others
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) emerged in Wuhan, China, in December 2019, and coronavirus disease 2019 (COVID-19) was declared a pandemic on March 11, 2020. Since then, several neurological manifestations have been reported, including both peripheral and central nervous systems (CNS) demyelinating diseases [1, 2]. Several articles reported the occurrence of acute demyelinating encephalomyelitis (ADEM), transverse myelitis (TM), multiple sclerosis (MS), and even neuromyelitis optica spectrum disorder (NMOSD), in association with COVID-19 infection [3,4,5].
There is an ongoing debate whether this association is merely related to the neurotropic features of SARS-CoV-2, or secondary to an acute or delayed immune-mediated response [6]. Coronavirus family showed clear evidence of neurotropic properties, as CNS demyelination has been reported with Middle East respiratory syndrome coronavirus (MERS-COV) and SARS-COV-1 [7,8,9].
When it comes to SARS-CoV-2, data are still limited in terms of cases of para-infectious and post-infectious CNS demyelinating/inflammatory diseases. Recent evidence has shown that SARS-CoV-2 can cross the blood–brain barrier and induce acute or delayed CNS demyelination [10]. Various mechanisms have been suggested including virus-induced hypercoagulable or proinflammatory states, direct viral invasion of the CNS, and post-infectious immune-mediated processes [11].
In this systematic review, we aim to investigate the available evidence regarding the different types of CNS demyelination in association with SARS-CoV-2 infection, and the proposed pathophysiological mechanisms in these cases.
Methods
Design
This systematic review collected data from PubMed, SCOPUS, EMBASE, Cochrane library, Google Scholar and Ovid databases, in accordance with Preferred Reporting Items for Systematic Reviews and Meta‐analysis (PRISMA) guidelines. We aimed to identify relevant articles that discussed CNS demyelination in association with SARS-CoV-2 infection from 1 January 2020 until June 15, 2021.
Search strategy
A pre-specified searching strategy consisted of a variation of keywords of relevant medical subject headings (MeSH) and keywords, including: “COVID-19”, “SARS-CoV-2”, “demyelination”, “demyelinating disease”, “multiple sclerosis”, “neuromyelitis optica”, and “transverse myelitis”. Furthermore, we hand-searched additional relevant COVID-19 articles that were referenced in the selected studies.
Inclusion criteria
We included all peer-reviewed publications that reported any form of CNS demyelination in association with COVID-19 infection, including but not limited to case reports and case series that met the following criteria: (i) studies reporting early or delayed acute CNS demyelination after COVID-19 infection; (ii) studies reporting possible association of cases fulfilling the diagnostic criteria of multiple sclerosis (MS), transverse myelitis (TM), neuromyelitis optica spectrum disorder (NMOSD), or myelin oligodendrocyte glycoprotein antibody disease (MOGAD), and COVID-19 infection; and (iii) studies published in English.
Exclusion criteria
The review was restricted to studies published in English. Publications that were not peer-reviewed were excluded from this study. We also excluded review papers, viewpoints, commentaries, unless reporting a case of demyelination, and studies where demyelination affected peripheral rather than central nervous systems. We also excluded cases not supported by positive imaging findings, laboratory or clinical evidence of COVID-19 infection.
Data extraction
Titles and abstracts of all identified studies were independently screened for relevance by the two reviewers, followed by full-text screening of the deemed eligible papers. The same reviewers then extracted data on the following parameters: article title, authors, publication year, age and gender of the patients, COVID-19-related information, onset of neurological symptoms, findings of neurological examination, MRI findings, laboratory work-up, CSF analysis, treatment and clinical outcome. Severity of COVID-19 infection was categorized into mild; asymptomatic infections or infections with mild symptoms not requiring hospitalization, moderate; requiring hospitalization but not ICU admission, and severe; requiring ICU admission and mechanical ventilation.
Statistical analysis
Qualitative data were described in percentages and numbers. Quantitative data were described using range (minimum and maximum), mean, standard deviation, and median. Significance of the obtained results was judged at the 5% level, but it could not be calculated due to insufficient data. A meta-analysis was planned to evaluate the association of the demographic findings, clinical, radiological and laboratory findings and outcomes, but it could not be performed due to lack of sufficient data.
Results
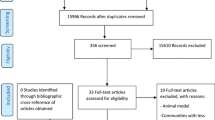
As illustrated in Fig. 1, our systematic search resulted in an initial number of 1,399 of potentially relevant articles, of which 966 were screened by title and abstract, following duplicates removal. Applying the inclusion/exclusion criteria to the full-text documents, 83 articles were deemed eligible, of which, 60 studies with a total of 102 patients, were included in the systematic review.
Brain demyelination
We identified 78 cases of brain demyelination from 38 articles. There were 40 males and 38 females. The median age of patients was 45.5 (0.17–77) years. The most common presenting neurological symptoms were lethargy, altered sensorium with or without seizures. In some cases, the symptoms followed COVID-19 infection by few weeks, while in others, they started at initial presentation and overlapped with COVID-19 symptoms. The demyelination mimicked a variety of conditions with the picture of encephalitis/encephalomyelitis being the most common (91%). At the same time, other patterns were less frequently reported such as MS, NMOSD and even MOGAD (10%).
It still remains an unanswered question whether demyelination represents a manifestation of direct viral invasion to the CNS, or it is an immune-mediated process triggering other well-known conditions, or in some cases a mere sequel for hypoxia affecting the CNS as a direct result of respiratory affection.
We summarized the results based on the most likely clinical diagnosis, in view of symptoms, laboratory and imaging findings in Tables 1 and 2.
Encephalitis/encephalomyelitis-like
A total of 71/78 patients (90%) presented by an encephalopathic clinical picture, lethargy, loss of consciousness and/or seizures which was supported by imaging findings. The median age of patients was 47 years. Hemorrhage and/or necrosis were reported in 26/71 (36.6%) of those patients. Of whom, 21/26 suffered severe COVID-19 infection which required mechanical ventilation. This observation raises the concern of a hypoxic ischemic theory and the need for anticoagulation in severely ill cases. A summary of the clinical characteristics is presented in Table 1.
MS-like demyelination
Three cases of MS-like demyelination: two females and one male have been reported. Their ages were 24, 28 and 29 years. The presentation was localizing to brain stem in two of them. On the other hand, unilateral optic neuritis was the main complaint in the third case.
In all cases the COVID-19 symptoms preceded the neurological symptoms by 2–4 weeks and the infection was mild in severity.
Oligoclonal bands were positive in two patients and not tested in the third.
Although the patient reported by Moore et al. fulfilled the 2017 McDonald criteria [12], we believe the MRI lesions were quite atypical being round in shape and larger than typical MS plaques. Follow-up is mandatory to securely establish the diagnosis.
The same atypical pattern of demyelination applies to the patient reported by Yavari et al. [5], where the lesions were large and located mainly in deep white matter and subcortical areas.
Despite the fact that the third case presented with optic neuritis, the associated field defect rendered the presentation atypical. Moreover, only two supratentorial lesions were detected in addition to the optic nerve lesion which does not fulfill the 2017 McDonald criteria.
That raises the question of whether these cases represent true MS or just a post-viral demyelinating syndrome. Findings of clinical characteristics are summarized in Table 2.
NMOSD and MOGAD-like demyelination
Longitudinally extensive transverse myelitis (LETM) and symptomatic cerebral syndrome are two of the well-known core features of NMOSD. In addition, ADEM could herald the first presentation of the disorder [13]. The patient reported by Zhogi et al. [14] was a 21-year-old male who presented with lethargy, vomiting, weakness of both lower limbs and urine retention, few weeks after suspicious COVID-19 symptoms.
Brain MRI revealed affection of bilateral corticospinal tracts up to cerebral peduncle and pons, marbled hyperintensity in the splenium of corpus callosum. Moreover, LETM was reported on his spinal MRI and testing for AQP4 antibody was positive. This patient received a probable diagnosis of COVID-19 based on his previous symptoms, chest imaging and positive antibody testing.
Additionally, Correa et al. [4] reported another female patient with LETM and brain hyperintensities in the fornix and subfornical area. The serology was positive for AQP4 after receiving a definite diagnosis of COVID-19 few weeks earlier.
MOGAD was reported in three post-COVID-19 patients based on positive antibody testing; the first was a 15-year-old patient who suffered subacute bilateral optic neuritis. COVID-19 infection was suspected based on his previous symptoms and two family members developing the infection a few weeks later. It was confirmed with positive nasopharyngeal PCR testing.
The second case was a 47-year-old male who presented with bilateral optic neuritis following asymptomatic infection, and was suspected based on his contact with positive cases. The infection was never confirmed though. Findings of clinical characteristics are shown in Table 2
The third case was a 4-year-old child presenting with ADEM. He had a mild infection with marked improvement after high-dose steroid treatment (Table 1).
Transverse myelitis
A total of 40 cases of TM have been reported in relation to SARS-CoV-2, of whom, 24 were isolated TM (Table 3), and 16 as a part of diffuse demyelinating process (Tables 1, 2). The cases included 19 females and 21 males.
LETM was the most frequently reported pattern of spinal involvement reported in 72.5% of cases of myelitis (19/24 of cases of isolated TM and 10/16 of cases of diffuse demyelination). Conus medullaris involvement was reported in 3 cases (3/40).
There are many mechanisms by which SARS-CoV-2 can induce myelitis: acute viral myelitis, post-COVID-19 immune-mediated myelitis, ischemic myelitis, part of an inflammatory demyelinating syndrome triggered by COVID-19 infection (ADEM, MS, NMOSD, and MOGAD). Table 3 summarizes the reported cases of isolated myelitis with COVID-19 infection.
Demyelination in special populations (children, pregnancy and puerperium)
Twenty pediatric, one pregnant and one postpartum cases of COVID-19-related brain demyelination have been reported. Although the pregnant and postpartum females had severe COVID-19 infection that required mechanical ventilation, they recovered almost completely with high doses of steroids. The presentation was of typical ADEM in one and diffuse leukoencephalitis with microbleeds in the other.
Among the pediatric cohort, there were 12 males and 8 females, with a median age of 9 years. Of them, 60% (12/20) suffered from severe COVID-19 infection. All patients presented with a picture of ADEM with (5/20) or without associated myelitis. In the five patients with myelitis, four showed LETM. The outcome was favorable (marked to complete recovery) in 13/20 patients. COVID-19-related isolated TM was reported in five pediatric patients (Table 3). Their median age was 11 years. LETM was the presenting imaging feature in all of them. Unfortunately, the outcome ranged between no to partial improvement.
Discussion
Several recent studies have evaluated the possible mechanisms of COVID-19-associated demyelination. Viral infection has demonstrated the ability to induce an inflammatory response, activating myelin-specific T cells, which can accelerate the development of early or delayed virus-induced demyelination [15]. Historically, SARS-CoV-1 and MERS-CoV, which are genetically similar to SARS-CoV-2, has been associated with central demyelination in literature [7].
Several experimental studies [16] revealed that murine coronavirus infection of susceptible mice has led to an inflammatory demyelination similar to MS, with coronavirus RNA sequences and its antigen detected in the demyelinating lesions. Furthermore, in one study [17], HCoV-229E viral RNA and HCoV-myelin cross-reactive T cell lines were predominantly detectable in the CNS of 36% of patients with MS, compared to none in patients with other neurological diseases and normal controls.
Although the exact mechanism of virus spread in the CNS has not been established, the two possible explanations are either hematogenous spread from systemic circulation to CNS or trans-neuronal spread through the olfactory pathway. In addition, the CNS can be potentially compromised through an ischemic–hypoxic insult resulting from severe respiratory affection or by latent immune-mediated mechanisms.
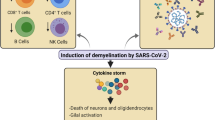
SARS-CoV-2 exhibits neurotropic and neuro-invasive properties and can cause direct neurological damage, through binding to angiotensin-converting enzyme-2 (ACE-2) receptors, whose expression is ubiquitous, including the CNS, or via blood circulation through Virchow Robin spaces [11]. Moreover, delayed CNS damage appears to be mediated by an undesired immune reaction following acute infection, leading to CNS demyelination [18].
Accumulated evidence showed that SARS-CoV-2 and several proinflammatory cytokines, including IL-1β, IL-2, IL-4, IL-6, IL-8, IL-10, TNF-α, and IFN-γ, can cross the blood–brain barrier, affecting macrophages, microglia, and astrocytes, which are the principal cells that mediate innate immunity in the CNS, thus creating a perfect cytokine storm for a proinflammatory state [10, 19]. IL-6 is an important proinflammatory mediator that can induce an immune response in the nervous system, and plays a crucial role in regulating the immune response in MS. In experimental autoimmune encephalomyelitis (EAE) model of MS, IL-6 aggravates clinical manifestations, neuroinflammation, and demyelination, principally by promoting pathogenic T helper (Th) 17 cell generation in the peripheral lymphoid organs [20, 21]. The levels of IL-6 were found to be correlated with the severity of COVID-19 symptoms, and this dysregulation can affect both innate and acquired immunity [1]. Furthermore, most COVID-19 patients exhibit increased circulating levels of IL-17 [22], which has a documented role in MS pathogenesis, mainly based on the data obtained from EAE model [23].
Moreover, autopsy data showed activation of astrocytes and microglia with infiltration of cytotoxic T cells, particularly in the brainstem, in COVID-19 patients [24]. In addition, Toll-like receptors (TLR), the main pattern recognition receptors expressed by CNS cells, have played a significant role in the pathogenesis of MS, and EAE model [25]. TLR are also believed to play a significant role in the pathogenesis of COVID‐19, mainly through recognition of viral particles, activation of the innate immune system, and secretion of pro‐inflammatory cytokines [26].
Another possible explanation could be the production of antibodies against myelin triggered by the virus. This para-infectious or post-infectious etiology is reported in several cases of post-SARS-CoV-2 Guillain–Barre syndrome. SARS-CoV-2 may play a role in triggering MS, similar to the documented role of Epstein–Barr virus [27]. These key aspects represent a maladaptive immune response to SARS-CoV-2 characterized by hyperactivity of innate immunity followed by immune dysregulation.
In view of this data, the reported cases in this review support a demyelinating aspect to SARS-CoV-2 infection. In the majority of cases, COVID-19 infection was confirmed by RT-PCR testing and in the remaining by adequate clinical, radiological and serological testing. Although causality cannot be adequately established, there is enough evidence to warrant further large-scale studies.
Brain demyelination was reported in a good number of cases; 78 patients, with encephalitis/encephalomyelitis presentation being the most common. The development of hemorrhage is potentially related to the severity of the respiratory involvement. Hypoxia as a result of such severe infection is a very likely contributing mechanism.
Although MS and other demyelinating diseases were reported, caution is a must when interpreting these findings, as in some cases the clinical presentation and MRI lesions are atypical. Further supporting laboratory investigations such as OCBs, VEP and spinal cord imaging will be needed to support the diagnosis. Despite the presence of MOG antibodies in some cases, MOGAD diagnosis should be made with caution, as acute bilateral optic neuritis and ADEM could be triggered by COVID-19 infection. A close follow-up is recommended to establish a solid diagnosis.
The fact the cytokines are involved to a great extent in COVID-19 pathogenesis could explain the trigger of MS and other common demyelinating disorders as they share the same pathogenic mechanisms. This might also explain the beneficial effects of steroids, plasma exchange (PLEX) and intravenous immunoglobulins (IVIg) in many of the reported cases.
It is noteworthy to mention the high frequency of LETM in the cases presenting with myelitis. Although it is shared by many diseases like NMOSD, idiopathic TM, and ADEM, SARS-CoV-2 myelitis should be added to the list of differential diagnosis of LETM.
With regard to the pediatric age group, and contrary to adult population, CNS demyelination appears to be less common, and usually associated with the development of multisystem inflammatory syndrome (MIS-C) [28]. Demyelinating disease etiologies can be difficult to stratify in children, as many of the initial presentations overlap among disease and syndromes; however, the most frequent type was post-infectious, immune-mediated ADEM-like presentation, followed by TM, and isolated splenial lesions [29]. Furthermore, the time of presentation, and the constantly negative PCR in CSF samples from affected patients, strongly suggests a post-infectious mechanism for the pathogenesis of CNS demyelination [30]. However, rare cases of acute, rather than post-infectious cerebellar ataxia have been recently reported in children with COVID-19 [31].
There are some limitations to the current systematic review. The main limitation is that it was based on small number of case reports and case series, despite extensive search of available literature, which hindered the ability to perform a meta-analysis. Moreover, although the selected reports provided relevant information, there was great heterogeneity regarding several aspects of the collected data. Furthermore, in few cases, nasopharyngeal RT-PCR testing was not performed, and in the majority of cases, CSF-PCR was negative, probably due to delayed presentation. Despite these shortcomings, the current review provides preliminary data on SARS-CoV-2-associated demyelinating diseases that can guide neurologists in dealing with such cases, and help future research.
Conclusion
This systematic review has shown an association between SARS‐CoV‐2 infection and the development of different types of CNS demyelination in literature, although causality cannot be made with absolute certainty. A probable para-infectious or post-infectious immune-mediated etiology might be implicated in patients with COVID-19. We are currently facing a dilemma of diagnosing common neurological disorders in the setting of this viral infection, raising the question of whether there is causality in this association, or just coincidence. The long-term prognosis of such cases is not clear, which may have implications regarding the use of disease-modifying therapies, or symptomatic treatments, in these patients. This relationship needs to be clarified in future research.
References
Andalib S, Biller J, Di Napoli M, Moghimi N, McCullough LD, Rubinos CA et al (2021) Peripheral nervous system manifestations associated with COVID-19. Curr Neurol Neurosci Rep 21(3):9
Montalvan V, Lee J, Bueso T, De Toledo J, Rivas K (2020) Neurological manifestations of COVID-19 and other coronavirus infections: a systematic review. Clin Neurol Neurosurg 194:105921
Paterson RW, Brown RL, Benjamin L, Nortley R, Wiethoff S, Bharucha T et al (2020) The emerging spectrum of COVID-19 neurology: clinical, radiological and laboratory findings. Brain 143(10):3104–3120
Corrêa DG, de Souza Lima FC, da Cruz Bezerra D, Coutinho AC, Hygino da Cruz LC (2021) COVID-19 associated with encephalomyeloradiculitis and positive anti-aquaporin-4 antibodies: cause or coincidence? Mult Scler 27(6):973–976
Yavari F, Raji S, Moradi F, Saeidi M (2020) Demyelinating changes alike to multiple sclerosis: a case report of rare manifestations of COVID-19. In: Banerjee TK (ed) Case reports in neurological medicine. Hindawi, London, pp 1–4
Mahalakshmi AM, Ray B, Tuladhar S, Bhat A, Paneyala S, Patteswari D et al (2021) Does COVID-19 contribute to development of neurological disease? Immun Inflamm Dis 9(1):48–58
Gu J, Gong E, Zhang B, Zheng J, Gao Z, Zhong Y et al (2005) Multiple organ infection and the pathogenesis of SARS. J Exp Med 202(3):415–424
Ann Yeh E, Collins A, Cohen ME, Duffner PK, Faden H (2004) Detection of coronavirus in the central nervous system of a child with acute disseminated encephalomyelitis. Pediatrics 113(1):e73–e76
Verstrepen K, Baisier L, De Cauwer H (2020) Neurological manifestations of COVID-19, SARS and MERS. Acta Neurol Belg 120(5):1051–1060
Desforges M, Le Coupanec A, Dubeau P, Bourgouin A, Lajoie L, Dubé M et al (2019) Human coronaviruses and other respiratory viruses: underestimated opportunistic pathogens of the central nervous system? Viruses 12(1):14
Wu Y, Xu X, Chen Z, Duan J, Hashimoto K, Yang L et al (2020) Nervous system involvement after infection with COVID-19 and other coronaviruses. Brain Behav Immun 87:18–22
Moore L, Ghannam M, Manousakis G (2021) A first presentation of multiple sclerosis with concurrent COVID-19 infection. eNeurologicalSci 22:100299
Wingerchuk DM, Banwell B, Bennett JL, Cabre P, Carroll W, Chitnis T et al (2015) International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology 85(2):177–189
Zoghi A, Ramezani M, Roozbeh M, Darazam IA, Sahraian MA (2020) A case of possible atypical demyelinating event of the central nervous system following COVID-19. Mult Scler Relat Disord. 44:102324
Perlman S, Zhao J (2017) Roles of regulatory T cells and IL-10 in virus-induced demyelination. J Neuroimmunol 15(308):6–11
Murray RS, Cai GY, Hoel K, Zhang JY, Soike KF, Cabirac GF (1992) Coronavirus infects and causes demyelination in primate central nervous system. Virology 188(1):274–284
Talbot PJ, Boucher A, Duquette P, Gruslin E (2005) Coronaviruses and neuroantigens: myelin proteins, myelin genes. In: Lavi E, Constantinescu CS (eds) Experimental models of multiple sclerosis [Internet]. Springer US, Boston [cited 2021 Jul 1], pp 781–791. https://doi.org/10.1007/0-387-25518-4_43
Lima M, Siokas V, Aloizou A-M, Liampas I, Mentis A-FA, Tsouris Z et al (2020) Unraveling the possible routes of SARS-COV-2 invasion into the central nervous system. Curr Treat Options Neurol 22(11):37
Han H, Ma Q, Li C, Liu R, Zhao L, Wang W et al (2020) Profiling serum cytokines in COVID-19 patients reveals IL-6 and IL-10 are disease severity predictors. Emerg Microbes Infect 9(1):1123–1130
Petkovic F, Castellano B (2016) The role of interleukin-6 in central nervous system demyelination. Neural Regen Res 11(12):1922
Janssens K, Slaets H, Hellings N (2015) Immunomodulatory properties of the IL-6 cytokine family in multiple sclerosis. Ann N Y Acad Sci 1351:52–60
Cao X (2020) COVID-19: immunopathology and its implications for therapy. Nat Rev Immunol 20(5):269–270
Kostic M, Dzopalic T, Zivanovic S, Zivkovic N, Cvetanovic A, Stojanovic I et al (2014) IL-17 and glutamate excitotoxicity in the pathogenesis of multiple sclerosis. Scand J Immunol 79(3):181–186
Matschke J, Lütgehetmann M, Hagel C, Sperhake JP, Schröder AS, Edler C et al (2020) Neuropathology of patients with COVID-19 in Germany: a post-mortem case series. Lancet Neurol 19(11):919–929
Racke MK, Drew PD (2009) Toll-like receptors in multiple sclerosis. In: Kielian T (ed) Toll-like receptors: roles in infection and neuropathology [Internet]. Springer, Berlin [cited 2021 Jul 1], pp 155–168 (Current Topics in Microbiology and Immunology; vol 336). https://doi.org/10.1007/978-3-642-00549-7_9
Khanmohammadi S, Rezaei N (2021) Role of Toll-like receptors in the pathogenesis of COVID-19. J Med Virol 93(5):2735–2739
Kamel WA, Ibrahim Ismail I, Al-Hashel JY (2021) Guillain–Barre syndrome following COVID-19 infection: first case report from Kuwait and review of the literature. Dubai Med J 18:1–5
Feldstein LR, Rose EB, Horwitz SM, Collins JP, Newhams MM, Son MBF et al (2020) Multisystem inflammatory syndrome in U.S. children and adolescents. N Engl J Med 383(4):334–346
Lindan CE, Mankad K, Ram D, Kociolek LK, Silvera VM, Boddaert N et al (2021) Neuroimaging manifestations in children with SARS-CoV-2 infection: a multinational, multicentre collaborative study. Lancet Child Adolesc Health 5(3):167–177
Stafstrom CE, Jantzie LL (2020) COVID-19: neurological considerations in neonates and children. Children (Basel) 7(9):E133
Sotgiu S, Uzzau S, Pippia A, Carta A, Antonucci R (2021) Expanding the spectrum of acute cerebellitis due to SARS-Cov-2. Pediatr Neurol 121:1–2
Zanin L, Saraceno G, Panciani PP, Renisi G, Signorini L, Migliorati K et al (2020) SARS-CoV-2 can induce brain and spine demyelinating lesions. Acta Neurochir 162(7):1491–1494
Brun G, Hak J-F, Coze S, Kaphan E, Carvelli J, Girard N et al (2020) COVID-19—white matter and globus pallidum lesions: demyelination or small-vessel vasculitis? Neurol Neuroimmunol Neuroinflamm 7(4):e777
Alqwaifly M (2020) Demyelinating solitary lesion of the central nervous system associated with COVID-19: a case report and literature review. Int J Adv Med 7(12):1884
Poyiadji N, Shahin G, Noujaim D, Stone M, Patel S, Griffith B (2020) COVID-19-associated acute hemorrhagic necrotizing encephalopathy: imaging features. Radiology 296(2):E119–E120
Varadan B, Shankar A, Rajakumar A, Subramanian S, Sathya AC, Hakeem AR et al (2021) Acute hemorrhagic leukoencephalitis in a COVID-19 patient—a case report with literature review. Neuroradiology 63(5):653–661
Yong MH, Chan YFZ, Liu J, Sanamandra SK, Kheok SW, Lim KC et al (2020) A rare case of acute hemorrhagic leukoencephalitis in a COVID-19 patient. J Neurol Sci 416:117035
Chalil A, Baker CS, Johnston RB, Just C, Debicki DB, Mayich MS et al (2021) Acute hemorrhagic encephalitis related to COVID-19. Neurol Clin Pract 11(2):e147–e151
Karapanayiotides T, Geka E, Prassopoulos P, Koutroulou I, Kollaras P, Kiourtzieva E et al (2020) Concentric demyelination pattern in COVID-19-associated acute haemorrhagic leukoencephalitis: a lurking catastrophe? Brain 143(12):e100
Handa R, Nanda S, Prasad A, Anand R, Zutshi D, Dass SK et al (2020) Covid-19-associated acute haemorrhagic leukoencephalomyelitis. Neurol Sci 41(11):3023–3026
Ghosh R, Dubey S, Finsterer J, Chatterjee S, Ray BK (2020) SARS-CoV-2-associated acute hemorrhagic, necrotizing encephalitis (AHNE) presenting with cognitive impairment in a 44-year-old woman without comorbidities: a case report. Am J Case Rep [Internet] [cited 2021 Jun 30]. https://www.amjcaserep.com/abstract/index/idArt/925641
Haqiqi A, Samuels TL, Lamb FJ, Moharrum T, Myers AE (2021) Acute haemorrhagic leukoencephalitis (Hurst disease) in severe COVID-19 infection. Brain Behav Immun Health. 12:100208
Mullaguri N, Sivakumar S, Battineni A, Anand S, Vanderwerf J (2021) COVID-19 related acute hemorrhagic necrotizing encephalitis: a report of two cases and literature review. Cureus [Internet] [cited 2021 Jun 30]. https://www.cureus.com/articles/50440-covid-19-related-acute-hemorrhagic-necrotizing-encephalitis-a-report-of-two-cases-and-literature-review
Radmanesh A, Derman A, Lui YW, Raz E, Loh JP, Hagiwara M et al (2020) COVID-19-associated diffuse leukoencephalopathy and microhemorrhages. Radiology 297(1):E223–E227
Sachs JR, Gibbs KW, Swor DE, Sweeney AP, Williams DW, Burdette JH et al (2020) COVID-19-associated leukoencephalopathy. Radiology 296(3):E184–E185
McLendon LA, Rao CK, Da Hora CC, Islamovic F, Galan FN (2021) Post-COVID-19 acute disseminated encephalomyelitis in a 17-month-old. Pediatrics 147(6):e2020049678
Scullen T, Keen J, Mathkour M, Dumont AS, Kahn L (2020) Coronavirus 2019 (COVID-19)-associated encephalopathies and cerebrovascular disease: the New Orleans Experience. World Neurosurg 141:e437–e446
Virhammar J, Kumlien E, Fällmar D, Frithiof R, Jackmann S, Sköld MK et al (2020) Acute necrotizing encephalopathy with SARS-CoV-2 RNA confirmed in cerebrospinal fluid. Neurology 95(10):445–449
Dixon L, Varley J, Gontsarova A, Mallon D, Tona F, Muir D et al (2020) COVID-19-related acute necrotizing encephalopathy with brain stem involvement in a patient with aplastic anemia. Neurol Neuroimmunol Neuroinflamm 7(5):e789
Montes-Ramirez J, Aquino-Lopez E (2020) COVID-19-associated diffuse leukoencephalopathy and cerebral microbleeds during puerperium. Int J Gynecol Obstet 151(3):466–467
McCuddy M, Kelkar P, Zhao Y, Wicklund D (2020) Acute demyelinating encephalomyelitis (ADEM) in COVID-19 infection: a case series. Neurol India 68(5):1192–1195
Assunção FB, Fragoso DC, Donoso Scoppetta TLP, Martins Maia AC (2021) COVID-19-associated acute disseminated encephalomyelitis-like disease. AJNR Am J Neuroradiol 42(4):E21–E23
Parsons T, Banks S, Bae C, Gelber J, Alahmadi H, Tichauer M (2020) COVID-19-associated acute disseminated encephalomyelitis (ADEM). J Neurol 267(10):2799–2802
Langley L, Zeicu C, Whitton L, Pauls M (2020) Acute disseminated encephalomyelitis (ADEM) associated with COVID-19. BMJ Case Rep 13(12):e239597
Wong PF, Craik S, Newman P, Makan A, Srinivasan K, Crawford E et al (2020) Lessons of the month 1: a case of rhombencephalitis as a rare complication of acute COVID-19 infection. Clin Med 20(3):293–294
Novi G, Rossi T, Pedemonte E, Saitta L, Rolla C, Roccatagliata L et al (2020) Acute disseminated encephalomyelitis after SARS-CoV-2 infection. Neurol Neuroimmunol Neuroinflamm 7(5):e797
Demirci Otluoglu G, Yener U, Demir MK, Yilmaz B (2020) Encephalomyelitis associated with Covid-19 infection: case report. Br J Neurosurg 7:1–3
Utukuri PS, Bautista A, Lignelli A, Moonis G (2020) Possible acute disseminated encephalomyelitis related to severe acute respiratory syndrome coronavirus 2 infection. AJNR Am J Neuroradiol 41:E82–E83
Lopes CCB, Brucki SMD, Passos Neto CEB, Corazza LA, Baima JPS, Fiorentino MD et al (2020) Acute disseminated encephalomyelitis in COVID-19: presentation of two cases and review of the literature. Arq Neuro-Psiquiatr 78(12):805–810
Kim HJ, Paul F, Lana-Peixoto MA, Tenembaum S, Asgari N, Palace J et al (2015) MRI characteristics of neuromyelitis optica spectrum disorder: an international update. Neurology 84(11):1165–1173
de Ruijter NS, Kramer G, Gons RAR, Hengstman GJD (2020) Neuromyelitis optica spectrum disorder after presumed coronavirus (COVID-19) infection: a case report. Mult Scler Relat Disord 46:102474
Zhou S, Jones-Lopez EC, Soneji DJ, Azevedo CJ, Patel VR (2020) Myelin oligodendrocyte glycoprotein antibody-associated optic neuritis and myelitis in COVID-19. J Neuroophthalmol 40(3):398–402
Kogure C, Kikushima W, Fukuda Y, Hasebe Y, Takahashi T, Shibuya T et al (2021) Myelin oligodendrocyte glycoprotein antibody-associated optic neuritis in a COVID-19 patient: a case report. Medicine 100(19):e25865
Valiuddin H, Skwirsk B, Paz-Arabo P (2020) Acute transverse myelitis associated with SARS-CoV-2: a case-report. Brain Behav Immun Health 5:100091
Moreno-Escobar MC, Kataria S, Khan E, Subedi R, Tandon M, Peshwe K et al (2021) Acute transverse myelitis with dysautonomia following SARS-CoV-2 infection: a case report and review of literature. J Neuroimmunol 353:577523
Munz M, Wessendorf S, Koretsis G, Tewald F, Baegi R, Krämer S et al (2020) Acute transverse myelitis after COVID-19 pneumonia. J Neurol 267(8):2196–2197
Sarma D, Bilello L (2020) A case report of acute transverse myelitis following novel coronavirus infection. Clin Pract Cases Emerg Med 4(3):321–323
Sotoca J, Rodríguez-Álvarez Y (2020) COVID-19-associated acute necrotizing myelitis. Neurol Neuroimmunol Neuroinflamm 7(5):e803
Domingues RB, Mendes-Correa MC, de Moura Leite FBV, Sabino EC, Salarini DZ, Claro I et al (2020) First case of SARS-COV-2 sequencing in cerebrospinal fluid of a patient with suspected demyelinating disease. J Neurol 267(11):3154–3156
AlKetbi R, AlNuaimi D, AlMulla M, AlTalai N, Samir M, Kumar N et al (2020) Acute myelitis as a neurological complication of Covid-19: a case report and MRI findings. Radiol Case Rep 15(9):1591–1595
Durrani M, Kucharski K, Smith Z, Fien S (2020) Acute transverse myelitis secondary to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): a case report. Clin Pract Cases Emerg Med 4(3):344–348
Chow CCN, Magnussen J, Ip J, Su Y (2020) Acute transverse myelitis in COVID-19 infection. BMJ Case Rep 13(8):e236720
Kaur H, Mason JA, Bajracharya M, McGee J, Gunderson MD, Hart BL et al (2020) Transverse myelitis in a child with COVID-19. Pediatr Neurol 112:5–6
Masuccio FG, Barra M, Claudio G, Claudio S (2021) A rare case of acute motor axonal neuropathy and myelitis related to SARS-CoV-2 infection. J Neurol 268(7):2327–2330
Shahali H, Ghasemi A, Farahani RH, Nezami Asl A, Hazrati E (2021) Acute transverse myelitis after SARS-CoV-2 infection: a rare complicated case of rapid onset paraplegia. J Neurovirol 27(2):354–358
Chakraborty U, Chandra A, Ray AK, Biswas P (2020) COVID-19-associated acute transverse myelitis: a rare entity. BMJ Case Rep 13(8):e238668
Baghbanian SM, Namazi F (2020) Post COVID-19 longitudinally extensive transverse myelitis (LETM)–a case report. Acta Neurol Belg [Internet] [cited 2021 Jul 1]. https://doi.org/10.1007/s13760-020-01497-x
Guler MA, Keskin F, Tan H (2020) Acute myelitis secondary to COVID-19 in an adolescent: causality or coincidence? New Trend Med Sci 1(2):132–136
Fumery T, Baudar C, Ossemann M, London F (2021) Longitudinally extensive transverse myelitis following acute COVID-19 infection. Mult Scler Relat Disord 48:102723
Nejad Biglari H, Sinaei R, Pezeshki S, Khajeh HF (2021) Acute transverse myelitis of childhood due to novel coronavirus disease 2019: The first pediatric case report and review of literature. Iran J Child Neurol 15(1):107–112
Ali L, Khan A, Elalamy O, Canibano B, Al hatou M, Adeli G, Ahmed I, Iqrar A, Abdussalam A, Ibrahim AS, Sardar S (2020) A rare presentation of acute flaccid myelitis in COVID-19 patient: a case report. Pak J Neurol Sci (PJNS) 15(3), Article 5
Román GC, Gracia F, Torres A, Palacios A, Gracia K, Harris D (2021) Acute transverse myelitis (ATM): clinical review of 43 patients with COVID-19-associated ATM and 3 post-vaccination ATM serious adverse events with the ChAdOx1 nCoV-19 vaccine (AZD1222). Front Immunol 12:653786
Saberi A, Ghayeghran A, Hatamian H et al (2020) COVID-19-associated myelitis, para/post infectious or infectious myelitis: a case report from the north of Iran. Caspian J Neurol Sci. 6(2):132–138
Funding
No funding received.
Author information
Authors and Affiliations
Contributions
III: conceptualization (equal); data curation (equal); investigation (equal); methodology (equal); project administration (equal); validation (equal); supervision (equal); writing—original draft (equal); writing—review and editing (equal). SS: conceptualization (equal); data curation (equal); investigation (equal); methodology (equal); project administration (equal); validation (equal); supervision (equal); writing—original draft (equal); writing—review and editing (equal).
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Rights and permissions
About this article
Cite this article
Ismail, I.I., Salama, S. Association of CNS demyelination and COVID-19 infection: an updated systematic review. J Neurol 269, 541–576 (2022). https://doi.org/10.1007/s00415-021-10752-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-021-10752-x