Abstract
Introduction
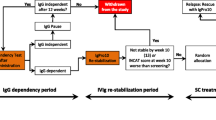
Individualized dosing is an established approach in intravenous immunoglobulin (IVIg) treatment for inflammatory neuropathies. There is less experience in effective dosing strategies for subcutaneous (SC) immunoglobulin.
Methods
We conducted a retrospective cohort study of patients with inflammatory neuropathies transferring from IVIg to SCIg in two UK peripheral nerve services. I-RODS and grip strength were used to measure outcome. Dose and clinical progress were documented at 1 year and at last review.
Results
44/56 patients remained on maintenance SCIg beyond 1 year (mean 3.3 years, range 1–9 years) with stable clinical outcomes. Clinical deteriorations were corrected by small increases in SCIg dose in 20 patients at 1 year, a further 9 requiring subsequent further up-titrations. Sixteen tolerated dose reduction. Mean dose change was + 2.4% from baseline. Two patients required IVIg bolus rescue (2 g/kg). Three patients successfully discontinued Ig therapy. Nine patients returned to IVIg due to clinical relapse or patient preference. Overall tolerance was good.
Discussion
Dose titration to clinical response is an effective approach in SCIg maintenance therapy.

Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
References
van Schaik IN et al (2018) Subcutaneous immunoglobulin for maintenance treatment in chronic inflammatory demyelinating polyneuropathy (PATH): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Neurol 17:35–46
Yoon M-S, Gold R, Kerasnoudis A (2015) Subcutaneous immunoglobulin in treating inflammatory neuromuscular disorders. Ther Adv Neurol Disord 8:153–159
Markvardsen LH et al (2014) Subcutaneous immunoglobulin preserves muscle strength in chronic inflammatory demyelinating polyneuropathy. Eur J Neurol 21:1465–1470
Chen Y, Wang C, Xu F, Ming F, Zhang H (2019) Efficacy and Tolerability of Intravenous Immunoglobulin and Subcutaneous Immunoglobulin in Neurologic Diseases. Clin Ther. 41(10):2112–2136. https://doi.org/10.1016/j.clinthera.2019.07.009
Cocito D et al (2014) Subcutaneous immunoglobulin in CIDP and MMN: a short-term nationwide study. J Neurol 261:2159–2164
Allen JA, Gelinas DF, Freimer M, Runken MC, Wolfe GI (2020) Immunoglobulin administration for the treatment of CIDP: IVIG or SCIG? J Neurol Sci. 15(408):116497. https://doi.org/10.1016/j.jns.2019.116497
MP L et al (2016) A proposed dosing algorithm for the individualized dosing of human immunoglobulin in chronic inflammatory neuropathies. J Peripher Nerv Syst 21:33–37
Berger M, Allen JA (2015) Optimizing IgG therapy in chronic autoimmune neuropathies: a hypothesis driven approach. Muscle Nerve 51:315–326
Eftimov F, Vermeulen M, de Haan RJ, van den Berg LH, van Schaik IN (2009) Subcutaneous immunoglobulin therapy for multifocal motor neuropathy. J Peripher Nerv Syst 14:93–100
Misbah SA et al (2011) A smooth transition protocol for patients with multifocal motor neuropathy going from intravenous to subcutaneous immunoglobulin therapy: an open-label proof-of-concept study. J Peripher Nerv Syst 16:92–97
Department of Health (2011). Clinical guidelines for immunoglobulin use: update to second edition
Van Den Bergh PYK et al (2010) European federation of neurological societies/peripheral nerve society guideline on management of chronic inflammatory demyelinating polyradiculoneuropathy: report of a joint task force of the European federation of neurological societies and the peripher. J Peripher Nerv Syst 15:1–9
Van Schaik INN et al (2010) European federation of neurological societies/peripheral nerve society guideline on management of multifocal motor neuropathy. Report of a joint task force of the European federation of neurological societies and the peripheral nerve society: first revis. J Periphe Nerv Syst 15:295–301
Draak THP et al (2016) Correlation of the patient’s reported outcome Inflammatory-RODS with an objective metric in immune-mediated neuropathies. Eur J Neurol 23:1248–1253
Draak THP et al (2015) Grip strength comparison in immune-mediated neuropathies: vigorimeter vs Jamar. J Peripher Nerv Syst 20:269–276
van Schaik IN et al (2016) Subcutaneous immunoglobulin for maintenance treatment in chronic inflammatory demyelinating polyneuropathy (The PATH Study): study protocol for a randomized controlled trial. Trials 17:1–15
Hadden RDM, Marreno F (2015) Switch from intravenous to subcutaneous immunoglobulin in CIDP and MMN: improved tolerability and patient satisfaction. Ther Adv Neurol Disord 8:14–19
Kuitwaard K et al (2017) Maintenance IV immunoglobulin treatment in chronic inflammatory demyelinating polyradiculoneuropathy. J Peripher Nerv Syst 22:425–432
Suleman A et al (2019) Evaluation of a personalized subcutaneous immunoglobulin treatment program for neurological patients. Can J Neurol Sci J Can Des Sci Neurol 46:38–43
Markvardsen LH, Harbo T (2017) Subcutaneous immunoglobulin treatment in CIDP and MMN. Efficacy, treatment satisfaction and costs. J Neurol Sci 378:19–25
Lazzaro C, Lopiano L, Cocito D (2014) Subcutaneous vs intravenous administration of immunoglobulin in chronic inflammatory demyelinating polyneuropathy: an Italian cost-minimization analysis. Neurol Sci 35:1023–1034
Cocito D et al (2016) Subcutaneous immunoglobulin in CIDP and MMN: a different long-term clinical response? J Neurol Neurosurg Psychiatry 87:791–793
Gentile L et al (2020) Long-term treatment with subcutaneous immunoglobulin in patients with chronic inflammatory demyelinating polyradiculoneuropathy: a follow-up period up to 7 years. Sci Rep 10:7910
Markvardsen LH et al (2013) Subcutaneous immunoglobulin in responders to intravenous therapy with chronic inflammatory demyelinating polyradiculoneuropathy. Eur J Neurol 20:836–842
Acknowledgements
MK gratefully received funding from a patient’s bequest. Professor Reilly is grateful to the Medical Research Council (MRC), MRC Centre grant (G0601943), and the National Institutes of Neurological Diseases and Stroke and office of Rare Diseases (U54NS065712) for their support. Dr Lunn and Dr Carr are also supported by the National Institute for Health Research University College London Hospitals Biomedical Research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Dr. Lunn was a Primary Investigator in studies for CSL Behring, UCB Pharma, Novartis, and Octapharma. He has also received ad hoc consulting fees from CSL Behring, UCB and an honorarium from Terumo BCT. Dr. Carr reports Grifols sponsorship for attendance at meeting and honorarium from CSL and Lupin for an advisory role. Dr Lavin reports sponsorship from CSL Behring and honorarium from CSL Behring and Grifols. Dr Kapoor reports Grifols sponsorship for attendance at meeting. The remaining authors have no conflicts of interest.
Ethical approval
We confirm that we have read the Journal’s position on issues involved in ethical publication and affirm that this report is consistent with those guidelines.
Rights and permissions
About this article
Cite this article
Kapoor, M., Keh, R., Compton, L. et al. Subcutaneous immunoglobulin dose titration to clinical response in inflammatory neuropathy. J Neurol 268, 1485–1490 (2021). https://doi.org/10.1007/s00415-020-10318-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-020-10318-3