Abstract
Purpose
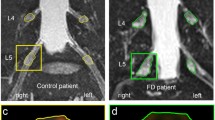
To examine dorsal root ganglia and proximal nerve segments in patients carrying the Fabry-related GLA-gene variant p.D313Y in comparison to patients with classical Fabry mutations and healthy controls by morphometric and functional magnetic resonance neurography.
Methods
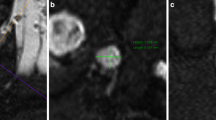
This prospective multicenter study examines the lumbosacral dorsal root ganglia and sciatic nerve in 11 female p.D313Y patients by a standardized magnetic resonance neurography protocol at 3 T. Volumes of dorsal root ganglia L3 to S2, permeability of dorsal root ganglia L5 and S1, and spinal nerve L5 as well as cross-sectional area of the sciatic nerve were assessed and compared to 10 females carrying a classical Fabry mutation and 16 healthy female controls.
Results
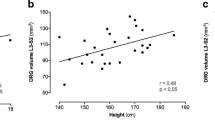
Compared to healthy controls, dorsal root ganglia volumes of p.D313Y females were enlarged by 53% (L3), 48% (L4), 43% (L5), 57% (S1) (p < 0.001), and 55% (S2) (p < 0.05), but less pronounced compared to females carrying a classical Fabry mutation. Compared to healthy controls, p.D313Y patients showed no changes of dorsal root ganglia vascular permeability, while patients with a classical Fabry mutation showed a distinct decrease (p < 0.05). Sciatic nerve cross-sectional area was mildly increased by 6% in p.D313Y as well as in classical Fabry patients (p < 0.05).
Conclusions
Patients carrying the GLA-gene variant p.D313Y show distinctly enlarged dorsal root ganglia, while vascular permeability remains within normal limits. Overall, these alterations partially share characteristics commonly seen in patients with a mutation causing classical FD. This suggests that p.D313Y causes a potentially treatable condition resembling an early stage of Fabry disease.



Similar content being viewed by others
References
Yasuda M, Shabbeer J, Benson SD, Maire I, Burnett RM, Desnick RJ (2003) Fabry disease: characterization of alpha-galactosidase A double mutations and the D313Y plasma enzyme pseudodeficiency allele. Hum Mutat 22(6):486–492. https://doi.org/10.1002/humu.10275
Eng CM, Resnick-Silverman LA, Niehaus DJ, Astrin KH, Desnick RJ (1993) Nature and frequency of mutations in the alpha-galactosidase A gene that cause Fabry disease. Am J Hum Genet 53(6):1186–1197
Hasholt L, Ballegaard M, Bundgaard H, Christiansen M, Law I, Lund AM, Norremolle A, Krogh Rasmussen A, Ravn K, Tumer Z, Wibrand F, Feldt-Rasmussen U (2017) The D313Y variant in the GLA gene - no evidence of a pathogenic role in Fabry disease. Scand J Clin Lab Invest 77(8):617–621. https://doi.org/10.1080/00365513.2017.1390782
Oder D, Uceyler N, Liu D, Hu K, Petritsch B, Sommer C, Ertl G, Wanner C, Nordbeck P (2016) Organ manifestations and long-term outcome of Fabry disease in patients with the GLA haplotype D313Y. BMJ Open 6(4):e010422. https://doi.org/10.1136/bmjopen-2015-010422
Niemann M, Rolfs A, Giese A, Mascher H, Breunig F, Ertl G, Wanner C, Weidemann F (2013) Lyso-Gb3 indicates that the alpha-galactosidase a mutation D313Y is not clinically relevant for fabry disease. JIMD Rep 7:99–102. https://doi.org/10.1007/8904_2012_154
Eng CM, Germain DP, Banikazemi M, Warnock DG, Wanner C, Hopkin RJ, Bultas J, Lee P, Sims K, Brodie SE, Pastores GM, Strotmann JM, Wilcox WR (2006) Fabry disease: guidelines for the evaluation and management of multi-organ system involvement. Genet Med 8(9):539–548. https://doi.org/10.1097/01.gim.0000237866.70357.c6
du Moulin M, Koehn AF, Golsari A, Dulz S, Atiskova Y, Patten M, Munch J, Avanesov M, Ullrich K, Muschol N (2017) The mutation p.D313Y is associated with organ manifestation in Fabry disease. Clin Genet 92(5):528–533. https://doi.org/10.1111/cge.13007
Selvarajah M, Nicholls K, Hewitson TD, Becker GJ (2011) Targeted urine microscopy in Anderson-Fabry disease: a cheap, sensitive and specific diagnostic technique. Nephrol Dial Transpl 26(10):3195–3202. https://doi.org/10.1093/ndt/gfr084
Godel T, Baumer P, Pham M, Kohn A, Muschol N, Kronlage M, Kollmer J, Heiland S, Bendszus M, Mautner VF (2017) Human dorsal root ganglion in vivo morphometry and perfusion in Fabry painful neuropathy. Neurology 89(12):1274–1282. https://doi.org/10.1212/WNL.0000000000004396
Gadoth N, Sandbank U (1983) Involvement of dorsal root ganglia in Fabry’s disease. J Med Genet 20(4):309–312
Godel T, Kohn A, Muschol N, Kronlage M, Schwarz D, Kollmer J, Heiland S, Bendszus M, Mautner VF, Baumer P (2018) Dorsal root ganglia in vivo morphometry and perfusion in female patients with Fabry disease. J Neurol. https://doi.org/10.1007/s00415-018-9053-y
Whybra C, Kampmann C, Krummenauer F, Ries M, Mengel E, Miebach E, Baehner F, Kim K, Bajbouj M, Schwarting A, Gal A, Beck M (2004) The Mainz Severity Score Index: a new instrument for quantifying the Anderson-Fabry disease phenotype, and the response of patients to enzyme replacement therapy. Clin Genet 65(4):299–307. https://doi.org/10.1111/j.1399-0004.2004.00219.x
Godel T, Pham M, Heiland S, Bendszus M, Baumer P (2016) Human dorsal-root-ganglion perfusion measured in-vivo by MRI. Neuroimage 141:81–87. https://doi.org/10.1016/j.neuroimage.2016.07.030
Jimenez-Andrade JM, Herrera MB, Ghilardi JR, Vardanyan M, Melemedjian OK, Mantyh PW (2008) Vascularization of the dorsal root ganglia and peripheral nerve of the mouse: implications for chemical-induced peripheral sensory neuropathies. Mol Pain 4:10. https://doi.org/10.1186/1744-8069-4-10
Hirakawa H, Okajima S, Nagaoka T, Kubo T, Takamatsu T, Oyamada M (2004) Regional differences in blood-nerve barrier function and tight-junction protein expression within the rat dorsal root ganglion. Neuroreport 15(3):405–408
Aerts JM, Groener JE, Kuiper S, Donker-Koopman WE, Strijland A, Ottenhoff R, van Roomen C, Mirzaian M, Wijburg FA, Linthorst GE, Vedder AC, Rombach SM, Cox-Brinkman J, Somerharju P, Boot RG, Hollak CE, Brady RO, Poorthuis BJ (2008) Elevated globotriaosylsphingosine is a hallmark of Fabry disease. Proc Natl Acad Sci USA 105(8):2812–2817. https://doi.org/10.1073/pnas.0712309105
Rombach SM, Dekker N, Bouwman MG, Linthorst GE, Zwinderman AH, Wijburg FA, Kuiper S, Vd Bergh Weerman MA, Groener JE, Poorthuis BJ, Hollak CE, Aerts JM (2010) Plasma globotriaosylsphingosine: diagnostic value and relation to clinical manifestations of Fabry disease. Biochim Biophys Acta 1802(9):741–748. https://doi.org/10.1016/j.bbadis.2010.05.003
Niemann M, Rolfs A, Stork S, Bijnens B, Breunig F, Beer M, Ertl G, Wanner C, Weidemann F (2014) Gene mutations versus clinically relevant phenotypes: lyso-Gb3 defines Fabry disease. Circ Cardiovasc Genet 7(1):8–16. https://doi.org/10.1161/CIRCGENETICS.113.000249
Torra R (2008) Renal manifestations in Fabry disease and therapeutic options. Kidney Int Suppl 111:S29–S32. https://doi.org/10.1038/ki.2008.522
Buechner S, Moretti M, Burlina AP, Cei G, Manara R, Ricci R, Mignani R, Parini R, Di Vito R, Giordano GP, Simonelli P, Siciliano G, Borsini W (2008) Central nervous system involvement in Anderson-Fabry disease: a clinical and MRI retrospective study. J Neurol Neurosurg Psychiatry 79(11):1249–1254. https://doi.org/10.1136/jnnp.2008.143693
Baptista MV, Ferreira S, Pinho EMT, Carvalho M, Cruz VT, Carmona C, Silva FA, Tuna A, Rodrigues M, Ferreira C, Pinto AA, Leitao A, Gabriel JP, Calado S, Oliveira JP, Ferro JM, Investigators POYS (2010) Mutations of the GLA gene in young patients with stroke: the PORTYSTROKE study-screening genetic conditions in Portuguese young stroke patients. Stroke 41(3):431–436. https://doi.org/10.1161/STROKEAHA.109.570499
Kolodny E, Fellgiebel A, Hilz MJ, Sims K, Caruso P, Phan TG, Politei J, Manara R, Burlina A (2015) Cerebrovascular involvement in Fabry disease: current status of knowledge. Stroke 46(1):302–313. https://doi.org/10.1161/STROKEAHA.114.006283
Sims K, Politei J, Banikazemi M, Lee P (2009) Stroke in Fabry disease frequently occurs before diagnosis and in the absence of other clinical events: natural history data from the Fabry Registry. Stroke 40(3):788–794. https://doi.org/10.1161/STROKEAHA.108.526293
Tondel C, Bostad L, Larsen KK, Hirth A, Vikse BE, Houge G, Svarstad E (2013) Agalsidase benefits renal histology in young patients with Fabry disease. J Am Soc Nephrol 24(1):137–148. https://doi.org/10.1681/ASN.2012030316
Hughes DA, Nicholls K, Shankar SP, Sunder-Plassmann G, Koeller D, Nedd K, Vockley G, Hamazaki T, Lachmann R, Ohashi T, Olivotto I, Sakai N, Deegan P, Dimmock D, Eyskens F, Germain DP, Goker-Alpan O, Hachulla E, Jovanovic A, Lourenco CM, Narita I, Thomas M, Wilcox WR, Bichet DG, Schiffmann R, Ludington E, Viereck C, Kirk J, Yu J, Johnson F, Boudes P, Benjamin ER, Lockhart DJ, Barlow C, Skuban N, Castelli JP, Barth J, Feldt-Rasmussen U (2017) Oral pharmacological chaperone migalastat compared with enzyme replacement therapy in Fabry disease: 18-month results from the randomised phase III ATTRACT study. J Med Genet 54(4):288–296. https://doi.org/10.1136/jmedgenet-2016-104178
Acknowledgements
We thank all patients for their valuable cooperation in this study. T.G. is supported by a postdoctoral fellowship from the Medical Faculty of the University of Heidelberg and received a research grant from Amicus Therapeutics. N.M. received travel grants, research grants and speaker honoraria from Shire, Sanofi Genzyme, and Amicus. J.K. received a research grant and personal fees from Alnylam Pharmaceuticals, the Olympia Morata stipend grant from the Medical Faculty of the University of Heidelberg, and lecture honoraria and financial support for conference attendance from Pfizer. She also advises for Akcea Therapeutics. M.B. received grants from the German Research Council (SFB 1158). S.H. was supported by a grant from the German Research Council (SFB 1118). V.M. was supported by a grant from the Märta and Erik Karberg Foundation for Medical Research.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Godel, T., Bäumer, P., Stumpfe, K. et al. Dorsal root ganglia volume is increased in patients with the Fabry-related GLA variant p.D313Y. J Neurol 266, 1332–1339 (2019). https://doi.org/10.1007/s00415-019-09262-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-019-09262-8