Abstract
Objective
In this prospective population-based registry study on ALS survival, we investigated the role of riluzole treatment, together with other clinical factors, on the prognosis in incident ALS cases in Emilia Romagna Region (ERR), Italy.
Methods
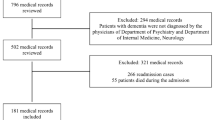
A registry for ALS has been collecting all incident cases in ERR since 2009. Detailed clinical data from all patients diagnosed with ALS between 1.1.2009 and 31.12.2014 have been analyzed for this study, with last follow up date set at 31.12.2015.
Results
During the 6 years of the study, there were 681 incident cases with a median tracheostomy-free survival of 40 months (95% CI 36–44) from onset and of 26 months (95% CI 24–30) from diagnosis; 573 patients (84.14%) were treated with riluzole, 207 (30.39%) patients underwent gastrostomy, 246 (36.12%) non invasive ventilation, and 103 (15.15%) invasive ventilation. Patients who took treatment for ≥ 75% of disease duration from diagnosis had a median survival of 29 months compared to 18 months in patients with < 75% treatment duration. In multivariable analysis, factors independently influencing survival were age at onset (HR 1.04, 95% CI 1.02–1.05, p < 0.001), dementia (HR 1.56, 95% CI 1.05–2.32, p = 0.027), degree of diagnostic certainty (HR 0.88, 95% CI 0.78–0.98, p = 0.021), gastrostomy (HR 1.46, 95% CI 1.14–1.88, p = 0.003), NIV (HR 1.43, 95% CI 1.12–1.82, p = 0.004), and weight loss at diagnosis (HR 1.05, 95% CI 1.03–1.07, p < 0.001), diagnostic delay (HR 0.98, 95% CI 0.97–0.99, p = 0.004), and % treatment duration (HR 0.98, 95% CI 0.98–0.99, p < 0.001).
Conclusions
Independently from other prognostic factors, patients who received riluzole for a longer period of time survived longer, but further population based studies are needed to verify if long-tem use of riluzole prolongs survival.

Similar content being viewed by others
References
Chiò A, Calvo A, Moglia C, Mazzini L, Mora G, PARALS study group (2011) Phenotypic heterogeneity of amyotrophic lateral sclerosis: a population based study. J Neurol Neurosurg Psychiatry 82:740–746
Al-Chalabi A, Hardiman O, Kiernan MC et al (2016) Amyotrophic lateral sclerosis: moving towards a new classification system. Lancet Neurol 15:1182–1194. https://doi.org/10.1016/S1474-4422(16)30199-5
Calvo A, Moglia C, Lunetta C et al (2017) Factors predicting survival in ALS: a multicenter Italian study. J Neurol 264:54–63. https://doi.org/10.1007/s00415-016-8313-y
Rooney J, Byrne S, Heverin M, Tobin K, Dick A, Donaghy C, Hardiman O (2015) A multidisciplinary clinic approach improves survival in ALS: a comparative study of ALS in Ireland and Northern Ireland. J Neurol Neurosurg Psychiatry 86:406–501
Georgoulopoulou E, Fini N, Vinceti M et al (2013) The impact of clinical factors, riluzole and therapeutic interventions on ALS survival: a population based study in Modena, Italy. Amyotroph Lateral Scler Frontotemporal Degener 8421:1–8. https://doi.org/10.3109/21678421.2013.763281
Mitsumoto H, Brooks BR, Silani V (2014) Clinical trials in amyotrophic lateral sclerosis: why so many negative trials and how can trials be improved? Lancet Neurol 13:1127–1138. https://doi.org/10.1016/S1474-4422(14)70129-2
Hardiman O, van den Berg LH, Kiernan MC (2011) Clinical diagnosis and management of amyotrophic lateral sclerosis. Nat Rev Neurol 7:639–649
Vucic S, Lin CS, Cheah BC, Murray J, Menon P, Krishnan AVKM (2013) Riluzole exerts central and peripheral modulating effects in amyotrophic lateral sclerosis. Brain 136:1361–1370
Gutierrez J, Federici T, Peterson B, Bartus R, Betourne A, Boulis NM (2016) Development of intrathecal riluzole: a new route of administration for the treatment of amyotrophic lateral sclerosis patients. Neurosurgery 63:193
Dyer AM (2017) Riluzole 5 mg/mL oral suspension : for optimized drug delivery in amyotrophic lateral sclerosis. Drug Design Dev Ther. https://doi.org/10.2147/dddt.s123776
Miller RG, Mitchell JD, Moore DH (2012) Riluzole for amyotrophic lateral sclerosis (ALS)/motor neuron disease (MND)(Review). Forbes 3:1–34. https://doi.org/10.1002/14651858.CD001447
Zoccolella S, Beghi E, Palagano G et al (2007) Riluzole and amyotrophic lateral sclerosis survival: a population-based study in southern Italy. Eur J Neurol 14:262–268. https://doi.org/10.1111/j.1468-1331.2006.01575.x
Mitchell JD, O’Brien MR, Joshi M (2006) Audit of outcomes in motor neuron disease (MND) patients treated with riluzole. Amyotroph Lateral Scler Other Mot Neuron Disord 7:67–71
Turner MR, Bakker M, Sham P, Shaw CE, Leigh PN, Al-Chalabi A (2001) The Kings’ Database 1999–2000: an analysis of the effect on survival of interventions in amyotrophic lateral sclerosis. Amyotroph Lateral Scler Other Mot Neuron Disord 2:43
Traynor BJ, Alexander M, Corr B, Frost E, Hardiman O (2003) An outcome study of riluzole in amyotrophic lateral sclerosis—a population-based study in Ireland, 1996–2000. J Neurol 250:473–479
Cetin H, Rath J, Füzi J et al (2015) Epidemiology of amyotrophic lateral sclerosis and effect of riluzole on disease course. Neuroepidemiology 44:6–15. https://doi.org/10.1159/000369813
Chen L, Liu X, Tang L et al (2016) Long-term use of riluzole could improve the prognosis of sporadic amyotrophic lateral sclerosis patients: a real-world cohort study in China. Front Aging Neurosci 8:1–8. https://doi.org/10.3389/fnagi.2016.00246
Mandrioli J, Biguzzi S, Guidi C et al (2014) Epidemiology of amyotrophic lateral sclerosis in Emilia Romagna Region (Italy): a population based study. Amyotrop Later Scler Frontotemp Degener 8421:262–268. https://doi.org/10.3109/21678421.2013.865752
Brooks BR, Miller RG, Swash M, Munsat TL (2000) El Escorial revisited: revised criteria for the diagnosis of amyotrophic lateral sclerosis. Amyotroph Lateral Scler Other Motor Neuron Disord 1:293–299. https://doi.org/10.1080/146608200300079536
Franchignoni F, Mora G, Giordano A et al (2013) Evidence of multidimensionality in the ALSFRS-R Scale: a critical appraisal on its measurement properties using Rasch analysis. J Neurol Neurosurg Psychiatry 84:1340–1345. https://doi.org/10.1136/jnnp-2012-304701
Franchignoni F, Mandrioli J, Giordano A, Ferro S (2015) A further Rasch study confirms that ALSFRS-R does not conform to fundamental measurement requirements. Amyotroph Lateral Scler Front Degener 8421:1–7. https://doi.org/10.3109/21678421.2015.1026829
Fasano A, Fini N, Ferraro D et al (2017) Percutaneous endoscopic gastrostomy, body weight loss and survival in amyotrophic lateral sclerosis: a population-based registry study. Amyotroph Lateral Scler Front Degener. https://doi.org/10.1080/21678421.2016.1270325
Andersen PM, Abrahams S, Borasio GD et al (2012) EFNS guidelines on the Clinical Management of Amyotrophic Lateral Sclerosis (MALS)—revised report of an EFNS task force. Eur J Neurol 19:360–375. https://doi.org/10.1111/j.1468-1331.2011.03501.x
Fini N, Georgoulopoulou E, Vinceti M et al (2014) Noninvasive and invasive ventilation and enteral nutrition for ALS in Italy. Muscle Nerve 50:508–516
Suissa S (2008) Immortal time bias in pharmaco-epidemiology. Am J Epidemiol 167:492–499
Labra J, Menon P, Byth K et al (2015) Rate of disease progression: a prognostic biomarker in ALS. J Neurol Neurosurg Psychiatry. https://doi.org/10.1136/jnnp-2015-310998
Thygesen SK, Christiansen CF, Christensen S, Lash TL, Sørensen HT (2011) The predictive value of ICD-10 diagnostic coding used to assess Charlson comorbidity index conditions in the population-based Danish National Registry of Patients. BMC Med Res Methodol 11:83. https://doi.org/10.1186/1471-2288-11-83
Bensimon G, Lacomblez L, Meiningier V (1994) A controlled trial of riluzole in amyotrophic lateral sclerosis. ALS/Riluzole Study Group 330:585–591
Miller RG, Jackson CE, Kasarskis EJ et al (2009) Practice parameter update: the care of the patient with amyotrophic lateral sclerosis: multidisciplinary care, symptom management, and cognitive/behavioral impairment (an evidence-based review): report of the Quality Standards Subcommittee of the American. Neurology 73:1227–1233
Cetin H, Klickovic U, Rath J et al (2015) Associations between co-medications and survival in ALS—a cohort study from Austria. J Neurol. https://doi.org/10.1007/s00415-015-7767-7
Gordon PH (2013) Amyotrophic Lateral Sclerosis: an update for 2013 Clinical Features, Pathophysiology, Management and Therapeutic Trials. Aging Dis 4:295–310. https://doi.org/10.14336/AD.2013.0400295
Sívori M, Rodríguez GE, Pascansky D, Sáenz C, Sica RE (2007) Outcome of sporadic amyotrophic lateral sclerosis treated with non-invasive ventilation and riluzole. Med (B Aires) 67:326–330
Tavakoli M (2002) Disease progression in amyotrophic lateral sclerosis. Identifying the cost-utility of riluzole by disease stage. Eur J Heal Econ 3:156–165
Chio A, Logroscino G, Hardiman O et al (2009) Prognostic factors in ALS: a critical review. Amyotroph Lateral Scler 10:310–323. https://doi.org/10.3109/17482960802566824
Chio A, Mora G, Leone M et al (2002) Early symptom progression rate is related to ALS outcome: a prospective population-based study. Neurology 59:99–103. https://doi.org/10.1212/WNL.59.12.2012-a
Kiernan MC, Vucic S, Cheah BC et al (2011) Amyotrophic lateral sclerosis. Lancet 377:942–955. https://doi.org/10.1016/S0140-6736(10)61156-7
Lee CT-C, Chiu Y-W, Wang K-C et al (2013) Riluzole and prognostic factors in amyotrophic lateral sclerosis long-term and short-term survival: a population-based study of 1149 cases in Taiwan. J Epidemiol 23:35–40. https://doi.org/10.2188/jea.JE20120119
Knibb JA, Keren N, Kulka A et al (2016) A clinical tool for predicting survival in ALS. J Neurol Neurosurg Psychiatry 87:1361–1367. https://doi.org/10.1136/jnnp-2015-312908
Chiò A, Canosa A, Gallo S et al (2011) ALS clinical trials: do enrolled patients accurately represent the ALS population? Neurology 77:1432–1437. https://doi.org/10.1212/WNL.0b013e318232ab9b
Lacomblez L, Bensimon G, Leigh PN, Guillet P, Meininger V (1996) Dose-ranging study of riluzole in amyotrophic lateral sclerosis. Amyotrophic Lateral Sclerosis/Riluzole Study Group II. Lancet 347:1425–1431
Bensimon G, Lacomblez L, Delumeau JC, Bejuit R, Truffinet P, Meininger V (2002) A study of riluzole in the treatment of advanced stage or elderly patients with amyotrophic lateral sclerosis. J Neurol 249:609–615
Rooney J, Fogh I, Westeneng HJ et al (2017) C9orf72 expansion differentially affects males with spinal onset amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry 88:281
Acknowledgements
The authors acknowledge ERRALS group: Project coordinator: J. Mandrioli. Collaborating centers: Department of Neuroscience, St. Agostino Estense Hospital, Azienda Ospedaliero Universitaria di Modena, Italy (J. Mandrioli, N. Fini, A. Fasano, E. Zucchi, A. Gessani, L. Ferri, S. Malerba, P. Nichelli). Department of Neurology, Bufalini Hospital, Cesena, Italy (Y. Handouk, S. Biguzzi, W. Neri). Department of Neurology, Forlì Hospital, Forlì, Italy (C. Guidi, W. Neri). Department of Neurosciences and Rehabilitation, St Anna Hospital, Ferrara (E. Sette, V. Tugnoli and M. R. Tola). Department of Neurology, G. Da Saliceto Hospital, Piacenza, Italy (E. Terlizzi, D. Guidetti). Department of Neurology, Infermi Hospital, Rimini, Italy (M. Currò Dossi, M. Pasquinelli, J. Andruccioli, A. Ravasio). Department of Neurology, Faenza and Ravenna Hospital, Ravenna, Italy (M. Casmiro,F. Rasi). Department of Neurology and IRCCS Istituto delle Scienze Neurologiche di Bologna, Bellaria Hospital, Bologna, Italy (F. Salvi, I. Bartolomei, R. Michelucci). Department of Biomedical and Neuromotor Science and IRCCS Istituto delle Scienze Neurologiche di Bologna, University of Bologna, Bologna, Italy (P. Avoni, S. De Pasqua, R. Liguori). Department of Neurology, IRCCS Arcispedale Santa Maria Nuova, Reggio Emilia, Italy (R. Rizzi, E. Canali, L. Codeluppi, F. Valzania). Department of Neuroscience, University of Parma, Parma, Italy (A. Taga, V. Pietrini, M. C. Trapasso, and L. Zinno). Department of Clinical and Experimental Medicine, Respiratory Disease and Lung Function Unit, University of Parma, Parma, Italy (M. Aiello). Department of Neurology, Fidenza Hospital, Parma, Italy (D. Medici, E. Chierici, E. Montanari). Department of Neurology, Carpi Hospital, Modena, Italy (M. Santangelo, S. Amidei). Department of Biomedical and Specialty Surgical Sciences, University of Ferrara, Ferrara, Italy (I. Casetta, M. Pugliatti, E. Della Coletta, V. Govoni, E. Granieri). Department of Neurology, Imola Hospital, Bologna, Italy (P. De Massis, V. Mussuto). Department of Neurology and IRCCS Istituto delle Scienze Neurologiche di Bologna, Maggiore Hospital, Bologna, Italy (A. Borghi, A. Gabellini, R. Michelucci). Neurology Unit, St. Orsola-Malpighi University Hospital, AOU di Bologna, Italy (R. Rinaldi, F. Cirignotta). Department of Hospital Services, Emilia Romagna Regional Health Authority, Bologna, Italy (S. Ferro). Neuroepidemiology Unit, IRCCS Istituto delle Scienze Neurologiche di Bologna, Italy (R. D’Alessandro). CREAGEN Research Center for Environmental, Genetic and Nutritional Epidemiology, University of Modena and Reggio Emilia, Modena, Italy.
Funding
Emilia Romagna Registry for ALS is supported by a Grant from the Emilia Romagna Regional Health Authority.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflicts of interest.
Ethical standards
The study has been approved by the ethics committees of ERR and has therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki.
Additional information
Members of ERRALS Group are listed in the Acknowledgements.
Rights and permissions
About this article
Cite this article
Mandrioli, J., Malerba, S.A., Beghi, E. et al. Riluzole and other prognostic factors in ALS: a population-based registry study in Italy. J Neurol 265, 817–827 (2018). https://doi.org/10.1007/s00415-018-8778-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-018-8778-y