Abstract
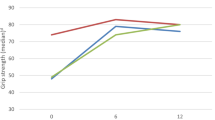
This multi-center Italian prospective observational study reports the 4 months follow-up data of 87 patients affected by chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) and multifocal motor neuropathy (MMN) shifted from intravenous to subcutaneous immunoglobulin treatment. A therapeutic shift from intravenous to subcutaneous immunoglobulin was performed in 87 patients (66 CIDP; 21 MMN) affected by immune-mediated peripheral neuropathies with evidence of a sustained clinical response to intravenous immunoglobulin. Patients were evaluated by means of the Overall Neuropathy Limitation Scale, Medical Research Council Scale and Life Quality Index questionnaire, both at the time of therapeutic shift and after 4 months of subcutaneous immunoglobulin treatment. A sustained clinical efficacy was observed after the switch to subcutaneous immunoglobulin: the Overall Neuropathy Limitation Scale score improved in the group of 66 CIDP patients (P = 0.018), with only one subject reporting a worsening of 1 point, and remained stable in the group of 21 MMN patients (P = 0.841), with one subject reporting a worsening of two points. An improvement in the patient’s perception of therapeutic setting was reported in both groups. This large multi-center study confirms the short-term clinical equivalence of subcutaneous versus intravenous immunoglobulin and a possible improvement in the patient’s perception of therapeutic setting with the subcutaneous administration. However, further studies are required to extend the results to a longer observational period.
Similar content being viewed by others
References
Van den Bergh PY, Hadden RD, Bouche P, Cornblath DR, Hahn A, Illa I et al (2010) European Federation of Neurological Societies; Peripheral Nerve Society. European Federation of Neurological Societies/Peripheral Nerve Society guideline on management of chronic inflammatory demyelinating polyradiculoneuropathy: report of a joint task force of the European Federation of Neurological Societies and the Peripheral Nerve Society—first revision. Eur J Neurol 17:356–363
Joint Task Force of the EFNS and the PNS (2010) European Federation of Neurological Societies/Peripheral Nerve Society guideline on management of multifocal motor neuropathy. Report of a joint task force of the European Federation of Neurological Societies and the Peripheral Nerve Society—first revision. J Peripher Nerv Syst 15:295–301
Gardulf A, Hammarstrom L, Smith CL (1991) Home treatment of hypogammaglobulinemia with subcutaneous gammaglobulin by rapid infusion. Lancet 338:162–166
Markvardsen LH, Debost JC, Harbo T, Sindrup SH, Andersen H, Christiansen I et al (2013) Subcutaneous immunoglobulin in responders to intravenous therapy with chronic inflammatory demyelinating polyradiculoneuropathy. Eur J Neurol 20:836–842
Harbo T, Andersen H, Hess A, Hansen K, Sindrup SH, Jakobsen J (2009) Subcutaneous versus intravenous immunoglobulin in multifocal motor neuropathy: a randomized, single-blinded cross-over trial. Eur J Neurol 16:631–638
Köller H, Schroeter M, Feischen H, Hartung HP, Kieseier BC (2006) Subcutaneous self-infusions of immunoglobulins as a potential therapeutic regimen in immune-mediated neuropathies. J Neurol 253:1505–1506
Lee DH, Linker RA, Paulus W, Schneider-Gold C, Chan A, Gold R (2008) Subcutaneous immunoglobulin infusion: a new therapeutic option in chronic inflammatory demyelinating polyneuropathy. Muscle Nerve 37:406–409
Eftimov F, Vermeulen M, de Haan RJ, van den Berg LH, van Schaik IN (2009) Subcutaneous immunoglobulin therapy for multifocal motor neuropathy. J Peripher Nerv Syst 14:93–100
Harbo T, Andersen H, Jakobsen J (2010) Long-term therapy with high doses of subcutaneous imunoglobulin in multifocal motor neuropathy. Neurology 75:1377–1380
Cocito D, Serra G, Falcone Y, Paolasso I (2011) The efficacy of subcutaneous immunoglobulin administration in chronic inflammatory demyelinating polyneuropathy responders to intravenous immunoglobulin. J Peripher Nerv Syst 16:150–152
Misbah SA, Baumann A, Fazio R, Dacci P, Schmidt DS, Burton J et al (2011) A smooth transition protocol for patients with multifocal motor neuropathy going from intravenous to subcutaneous immunoglobulin therapy: an open-label proof-of-concept study. J Peripher Nerv Syst 16:92–97
Nicolay U, Kiessling P, Berger M, Gupta S, Yel L, Roifman CM et al (2006) Health-related quality of life and treatment satisfaction in North American patients with primary immunedeficiency diseases receiving subcutaneous IgG self-infusions at home. J Clin Immunol 26:65–72
Boërio D, Créange A, Hogrel JY, Guéguen A, Bertrand D, Lefaucheur JP (2010) Nerve excitability changes after intravenous immunoglobulin infusions in multifocal motor neuropathy and chronic inflammatory demyelinating neuropathy. J Neurol Sci 292:63–71
Rajabally YA (2013) Subcutaneous immunoglobulin therapy for inflammatory neuropathy: current evidence base and future prospects. J Neurol Neurosurg Psychiatry. doi:10.1136/jnnp-2013-305644
Van Doorn PA, Kuitwaard K, Jacobs BC (2011) Serum IgG levels as biomarkers for optimizing IVIg therapy in CIDP. J Peripher Nerv Syst 16:38–40
Vlam L, Cats EA, Willemse E, Franssen H, Medic J, Piepers S et al (2013) Pharmacokinetics of intravenous immunoglobulin in multifocal motor neuropathy. J Neurol Neurosurg Psychiatry. doi:10.1136/jnnp-2013-306227
Kuitwaard K, van Doorn PA, Vermeulen M, van den Berg LH, Brusse E, van der Kooi AJ et al (2013) Serum IgG levels in IV immunoglobulin treated chronic inflammatory demyelinating polyneuropathy. J Neurol Neurosurg Psychiatry 84:859–861
Van Nes SI, Vanhoutte EK, van Doorn PA, Hermans M, Bakkers M, Kuitwaard K et al (2011) Rasch-built Overall Disability Scale (R-ODS) for immune-mediated peripheral neuropathies. Neurology 76:337–345
Cocito D, Serra G, Paolasso I, Barilà DA, Lopiano L, Cattel L (2012) Economic and quality of life evaluation of different modalities of immunoglobulin therapy in chronic dysimmune neuropathies. J Peripher Nerv Syst 17:426–428
Lazzaro C, Lopiano L, Cocito D (2014) Subcutaneous vs Intravenous administration of immunoglobulin in chronic inflammatory demyelinating polyneuropathy: an Italian cost-minimization analysis. Neurol Sci 35:1023–1034
Conflicts of interest
This research was funded by the “Associazione Neuropatie Croniche Piemonte ONLUS”. The sponsor had no role in the design of the study; the collection, analysis and interpretation of the data; the decision to approve publication of the finished manuscript. Dario Cocito received honoraria for lecturing from Baxter, CSL Behring, and Kedrion; he received personal compensation for serving in Advisory Board of CSL Behring, Kedrion and Lilly and travel grants to attend scientific meeting from Baxter, Grifols, Kedrion, and CSL Behring. Anna Mazzeo reports travel support for Scientific Meetings from CSL Behring and Kedrion. Raffaella Fazio received personal compensation for serving in Advisory Board of CSL Behring and Baxter, and travel grants from Baxter, Grifols, Kedrion and CLS Behring. Ada Francia received travel grants and honoraria from Biogenidec, Genzyme, Novartis, Almirall, Merck Serono, CSL Behring, Kedrion, Teva. Gabriele Siciliano received personal compensation for serving in Advisory Board from Grunenthal, Baxter and Grifols. Giovanni Antonini received honoraria for lecturing from Kedrion and travelgrant from Genzyme, Behring e Kedrion. Eduardo Nobile-Orazio reports personal compensation for serving in the Advisory Board of CSL Behring, Baxter, Kedrion and Novartis; he received honoraria for lecturing travel grants to attend scientific meetings from Baxter, CSL Behring, Grifols and Kedrion. Erdita Peci, Girolama Alessandra Marfia received travel grant to attend scientific meetings from CSL Behring. Aristide Merola, Paola Valentino, Rocco Liguori, Massimiliano Filosto, Angelo Maurizio Clerici, Stefania Lelli, Ilaria Cecconi, and Leonardo Lopiano declare no conflicts of interest.
Ethical standards
The authors declare that they acted in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
A list of contributors can be found in “Appendix 1”.
Appendix 1
Appendix 1
Contributors from the “SCIg and Chronic Dysimmune Neuropathies Italian Network”.
Dario Cocito, Erdita Peci, Aristide Merola, Alberto Romagnolo, Department of Neuroscience, University of Turin; Antonio Toscano, Anna Mazzeo, Luca Gentile, Massimo Russo, Department of Neuroscience, Psychiatry and Anesthesiology, A.O.U. Policlinico “G. Martino” Messina; Raffaella Fazio, Daniela Velardo, Department of Neurology, IRCCS San Raffaele Milano; Ada Francia, Simona Pontecorvo III Neurological Clinic, Policlinico Umberto I Roma; Paola Valentino, Rita Nisticò, Neurological Clinic, Department of Medical and Surgical Sciences, University Magna Grecia of Catanzaro; Rocco Liguori, Vincenzo Angelo Donadio, Neurological Clinic IRCCS, Institute of Neurological Sciences of Bologna; Massimiliano Filosto, Neurological Clinic—Neuromuscular Diseases and Neuropathies Section, University and A.O “Spedali Civili” of Brescia; Gabriele Siciliano, Erica Schirinzi, Department of Clinical and Experimental Medicine, University of Pisa; Giorgio Bono, Maurizio Clerici, Sc Neurology and Stroke Unit, A.O. Hospital Varese—Circolo and Fondazione Macchi, University of Insubria; Stefania Lelli, Tiziana Rosso, Operative Unit of Neurology, Hospital San Giacomo Apostolo, Castelfranco Veneto; Girolama Alessandra Marfia, Giorgia Mataluni, Dipartment of Neuroscience, Policlinico tor Vergata; Giovanni Antonini, Alessandro Clemenzi, Neurological Sciences, University “La Sapienza” of Rome; Ilaria Cecconi, Michele Suprani, U.O Child Neuropsychiatry, Hospital Sant’Orsola Malpighi Bologna; Eduardo Nobile-Orazio, Neurology 2, Department of Translational Medicine, University of Milano, IRCCS Istituto Clinico Humanitas, Rozzano.
Rights and permissions
About this article
Cite this article
Cocito, D., Merola, A., Peci, E. et al. Subcutaneous immunoglobulin in CIDP and MMN: a short-term nationwide study. J Neurol 261, 2159–2164 (2014). https://doi.org/10.1007/s00415-014-7444-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-014-7444-2