Abstract
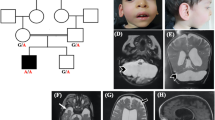
Pontocerebellar hypoplasia (PCH) type 1 is characterized by the co-occurrence of spinal anterior horn involvement and hypoplasia of the cerebellum and pons. EXOSC3 has been recently defined as a major cause of PCH type 1. Three different phenotypes showing variable severity have been reported. We identified a homozygous mutation [c.395A > C/p.D132A] in EXOSC3 in four patients with muscle hypotonia, developmental delay, spinal anterior horn involvement, and prolonged survival, consistent with the “mild PCH1 phenotype”. Interestingly, isolated cerebellar hypoplasia limited to the hemispheres or involving both hemispheres and vermis was the main neuroradiologic finding, whereas the pontine volume was in the normal range for age. These findings strongly suggest that analysis of the EXOSC3 gene should be recommended also in patients with spinal anterior horn involvement and isolated cerebellar hypoplasia.



Similar content being viewed by others
References
Namavar Y, Barth PG, Poll-The BT, Baas F (2011) Classification, diagnosis and potential mechanisms in pontocerebellar hypoplasia. Orphanet J Rare Dis 6:50
Barth PG (1993) Pontocerebellar hypoplasias: an overview of a group of inherited neurodegenerative disorders with fetal onset. Brain Dev 15:411–422
Rudnik-Schöneborn S, Sztriha L, Aithala GR et al (2003) Extended phenotype of pontocerebellar hypoplasia with infantile spinal muscular atrophy. Am J Med Genet A 117A:10–17
Lev D, Michelson-Kerman M, Vinkler C, Blumkin L, Shalev SA, Lerman-Sagie T (2008) Infantile onset progressive cerebellar atrophy and anterior horn cell degeneration—a late-onset variant of PCH-1? Eur J Paediatr Neurol 12:97–101
Renbaum P, Kellerman E, Jaron R et al (2009) Spinal muscular atrophy with pontocerebellar hypoplasia is caused by a mutation in the VRK1 gene. Am J Hum Genet 85:281–289
Namavar Y, Barth PG, Kasher PR et al (2011) Clinical, neuroradiological and genetic findings in pontocerebellar hypoplasia. Brain 134(Pt 1):143–156
Simonati A, Cassandrini D, Bazan D, Santorelli FM (2011) TSEN54 mutation in a child with pontocerebellar hypoplasia type 1. Acta Neuropathol 121:671–673
Wan J, Yourshaw M, Mamsa H et al (2012) Mutations in the RNA exosome component gene EXOSC3 cause pontocerebellar hypoplasia and spinal motor neuron degeneration. Nat Genet 44:704–708
Rudnik-Schöneborn S, Senderek J, Jen JC et al (2013) Pontocerebellar hypoplasia type 1. Clinical spectrum and relevance of EXOSC3 mutations. Neurology 80:1–9
Liu Q, Greimann JC, Lima CD (2006) Reconstitution, activities, and structure of the eukaryotic RNA exosome. Cell 127:1223–1237
Lorentzen E, Basquin J, Conti E (2008) Structural organization of the RNA-degrading exosome. Curr Opin Struct Biol 18:709–713
Lehner B, Sanderson CM (2004) A protein interaction framework for human mRNA degradation. Genome Res 14:1315–1323
Raijmakers R, Egberts WV, van Venrooij WJ, Pruijn GJ (2002) Protein-protein interactions between human exosome components support the assembly of RNase PH-type subunits into a six-membered PNPase-like ring. J Mol Biol 323:653–663
Sloan KE, Schneider C, Watkins NJ (2012) Comparison of the yeast and human nuclear exosome complexes. Biochem Soc Trans 40:850–855
Biancheri R, Bruno C, Cassandrini D, Bertini E, Santorelli FM, Rossi A (2011) Cerebellar hypoplasia and brainstem thinning associated with severe white matter and basal ganglia abnormalities in a child with an mtDNA deletion. J Inherit Metab Dis 34:1225–1227
Cassandrini D, Biancheri R, Tessa A et al (2010) Pontocerebellar hypoplasia: clinical, pathologic, and genetic studies. Neurology 75:1459–1464
Raininko R, Autti T, Vanhanen SL et al (1994) The normal brainstem from infancy to old age. Neuroradiology 36:364–368
Callahan KP, Butler JS (2008) Evidence for core exosome independent function of the nuclear exoribonuclease Rrp6p. Nucleic Acids Res 36:6645–6655
Callahan KP, Butler JS (2010) TRAMP complex enhances RNA degradation by the nuclear exosome component Rrp6. J Biol Chem 285:3540–3547
Kiss DL, Andrulis ED (2011) The exozyme model: a continuum of functionally distinct complexes. RNA 17:1–13
Kolb SJ, Sutton S, Schoenberg DR (2010) RNA processing defects associated with diseases of the motor neuron. Muscle Nerve 41:5–17
Tomecki R, Kristiansen MS, Lykke-Andersen S et al (2010) The human core exosome interacts with differentially localized processive RNases: hDIS3 and hDIS3L. EMBO J 29:2342–2357
Kiss DL, Hou D, Gross RH, Andrulis ED (2012) Dis3- and exosome subunit-responsive 3′ mRNA instability elements. Biochem Biophys Res Commun 423:461–466
Acknowledgments
We thank P. Broda for technical assistance. The patients samples were obtained from the “Cell Line and DNA Biobank from patients affected by genetic diseases” (G. Gaslini Institute)—Telethon Genetic Biobank Network (Project No. GTB12001).
Conflicts of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Carlo Minetti and Filippo Maria Santorelli contributed equally to this work.
Rights and permissions
About this article
Cite this article
Biancheri, R., Cassandrini, D., Pinto, F. et al. EXOSC3 mutations in isolated cerebellar hypoplasia and spinal anterior horn involvement. J Neurol 260, 1866–1870 (2013). https://doi.org/10.1007/s00415-013-6896-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-013-6896-0