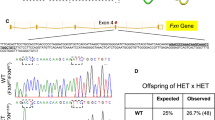
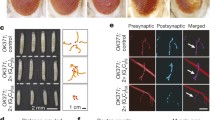
Abstract
Patients with Friedreich ataxia (FRDA) have severely reduced levels of the mitochondrial protein frataxin, which results from a large GAA triplet-repeat expansion within the frataxin gene (FXN). High evolutionary conservation of frataxin across species has enabled the development of disease models of FRDA in various unicellular and multicellular organisms. Mouse models include classical knockout models, in which the Fxn gene is constitutively inactivated, and knock-in models, in which a GAA repeat mutation or the conditional allele is inserted into the genome. Recently, “humanised” GAA repeat expansion mouse models were obtained by combining the constitutive knockout with the transgenic expression of a yeast artificial chromosome carrying the human FRDA locus. In lower organisms such as Caenorhabditis elegans and Drosophila, straight-forward and conditional RNA interference technology has provided an easy way to knock down frataxin expression. Conditional mouse models have been used for pre-clinical trials of potential therapeutic agents, including idebenone, MnTBAP (a superoxide dismutase mimetic), and iron chelators. Various models of FRDA have shown that different, even opposite, phenotypes can be observed, depending on the level of frataxin expression. Additional studies with animal models will be essential for an enhanced understanding of the disease pathophysiology and for the development of better therapies.
Similar content being viewed by others
References
Pandolfo M (2006) Friedreich ataxia.In: Wells RD, Ashizawa T (eds) GeneticInstabilities and Neurological Diseases,Boston, Elsevier Academic Press, pp277–298
Puccio H, Koenig M (2002) Friedreichataxia: a paradigm for mitochondrialdiseases. Curr Opin Genet Dev 12:272–277
Cossee M, Puccio H, Gansmuller A,Koutnikova H, Dierich A, LeMeur M,Fischbeck K, Dollé P, Koenig M (2000)Inactivation of the Friedreich ataxiamouse gene leads to early embryoniclethality without iron accumulation.Hum Mol Genet 9:1219–1226
Puccio H, Simon D, Cossée M, Criqui-Filipe P, Tiziano F, Melki J, HindelangC, Matyas R, Rustin P, Koenig M (2001)Mouse models for Friedreich ataxiaexhibit cardiomyopathy, sensory nervedefect and Fe-S enzyme deficiencyfollowed by intramitochondrial irondeposits. Nat Genet 27:181–186
Seznec H, Simon D, Monassier L,Criqui-Filipe P, Gansmuller A, Rustin P,Koenig M, Puccio H (2004) Idebenonedelays the onset of cardiac functionalalteration without correction of Fe-Senzymes deficit in a mouse model forFriedreich ataxia. Hum Mol Genet 13:1017–1024
Duby G, Foury F, Ramazzotti A,Herr mann J, Lutz T (2002) A nonessentialfunction for yeast frataxin iniron- sulfur cluster assembly. Hum MolGenet 11:2635–2643
Muhlenhoff U, Richhardt N, Ristow M,Kispal G, Lill R (2002) The yeastfrataxin homolog Yfh1p plays a specificrole in the maturation of cellularFe/S proteins. Hum Mol Genet 11:2025–2036
Martelli A, Wattenhofer-Donze M,Schmucker S, Bouvet S, Reutenauer L,Puccio H (2007) Frataxin is essentialfor extramitochondrial Fe-S clusterproteins in mammalian tissues. HumMol Genet 16:2651–2658
Michael S, Petrocine SV, Qian J,Lamarche JB, Knutson MD, GarrickMD, Koeppen AH (2006) Iron andiron-responsive proteins in the cardiomyopathyof Friedreich’s ataxia.Cerebellum 5:257–267
Seznec H, Simon D, Bouton C,Reutenauer L, Hertzog A, Golik P,Procaccio V, Patel M, Drapier JC,Koenig M, Puccio H (2005) Friedreichataxia: the oxidative stress paradox.Hum Mol Genet 14:463–474
Pandolfo M (2008) Drug insight: antioxidanttherapy in inherited ataxias.Nat Clin Pract Neurol 4:86–96
Chantrel-Groussard K, Geromel V,Puccio H, Koenig M, Munnich A, RötiqA, Rustin P (2001) Disabled earlyrecruitment of antioxidant defenses inFriedreich’s ataxia. Hum Mol Genet10:2061–2067
Anderson PR, Kirby K, Orr WC,Hilliker AJ, Phillips JP (2008) Hydrogenperoxide scavenging rescuesfrataxin deficiency in a Drosophilamodel of Friedreich’s ataxia. Proc NatlAcad Sci USA 105:611–616
Ristow M, Mulder H, Pomplun D,Schulz TJ, Müller-Schmehl K, KrauseA, Fex M, Puccio H, Müller J, Isken F,Spranger J, Müller-Wieland D,Magnuson MA, Möhlig M, Koenig M,Pfeiffer AF (2003) Frataxin deficiencyin pancreatic islets causes diabetes dueto loss of beta cell mass. J Clin Invest112:527–534
Finocchiaro G, Baio G, Micossi P, PozzaG, di Donato S (1988) Glucose metabolismalterations in Friedreich’s ataxia.Neurology 38:1292-1296
Simon D, Seznec H, Gansmuller A,Carelle N, Weber P, Metzger D, RustinP, Koenig M, Puccio H (2004) Friedreichataxia mouse models with progressivecerebellar and sensory ataxiareveal autophagic neurodegenerationin dorsal root ganglia. J Neurosci 24:1987–1995
Santos MM, Ohshima K, Pandolfo M(2001) Frataxin deficiency enhancesapoptosis in cells differentiating intoneuroectoderm. Hum Mol Genet 10:1935–1944
Wong A, Yang J, Cavadini P, Gellera C,Lonnerdal B, Taroni F, Cortopassi G(1999) The Friedreich’s ataxia mutationconfers cellular sensitivity tooxidant stress which is rescued bychelators of iron and calcium and inhibitorsof apoptosis. Hum Mol Genet8:425–430
Brunk UT, Terman A (2002) Lipofuscin:mechanisms of age-related accumulationand influence on cell function.Free Radic Biol Med 33:611–619
Lamarche J, Luneau C, Lemieux B(1982) Ultrastructural observations onspinal ganglion biopsy in Friedreich’sataxia: a preliminary report. Can JNeurol Sci 9:137–139
Lamarche JB, Cote M, Lemieux B(1980) The cardiomyopathy of Friedreich’sataxia morphological observationsin 3 cases. Can J Neurol Sci 7:389–396
Larnaout A, Belal S, Zouari M, Fki M,Ben Hamida C, Goebel HH, BenHamida M, Hentati F (1997) Friedreich’sataxia with isolated vitamin Edeficiency: a neuropathological studyof a Tunisian patient. Acta Neuropathol(Berl) 93:633–637
Lemasters JJ, Nieminen AL, Qian T,Trost LC, Elmore SP, Nishimura Y,Crowe RA, Cascio WE, Bradham CA,Brenner DA, Herman B (1998) Themitochondrial permeability transitionin cell death: a common mechanism innecrosis, apoptosis and autophagy.Biochim Biophys Acta 1366:177–196
Miranda CJ, Santos MM, Ohshima K,Smith J, Li L, Bunting M, Cossée M,Koenig M, Sequeiros J, Kaplan J,Pandolfo M (2002) Frataxin knockinmouse. FEBS Lett 512:291–297
Rai M, Soragni E, Jenssen K, Burnett R,Herman D, Coppola G, Geschwind DH,Gottesfeld JM, Pandolfo M (2008)HDAC inhibitors correct frataxin deficiencyin a Friedreich ataxia mousemodel. PLoS ONE 3:e1958
Festenstein R (2006) Breaking thesilence in Friedreich’s ataxia. Nat ChemBiol 2:512–513
Herman D, Jenssen K, Burnett R,Soragni E, Perlman SL, Gottesfeld JM(2006) Histone deacetylase inhibitorsreverse gene silencing in Friedreich’sataxia. Nat Chem Biol 2:551–558
Pook MA, Al-Mahdawi S, Carroll CJ,Cossée M, Puccio H, Lawrence L, ClarkP, Lowrie MB, Bradley JL, Cooper JM,Koenig M, Chamberlain S (2001) Rescueof the Friedreich’s ataxia knockoutmouse by human YAC transgenesis.Neurogenetics 3:185–193
Al-Mahdawi S, Pinto RM, Ruddle P,Carroll C, Webster Z, Pook M (2004)GAA repeat instability in Friedreichataxia YAC transgenic mice. Genomics84:301–310
De Biase I, Rasmussen A, Monticelli A,Al-Mahdawi S, Pook M, Cocozza S,Bidichandani SI (2007) Somatic instabilityof the expanded GAA tripletrepeatsequence in Friedreich ataxiaprogresses throughout life. Genomics90:1–5
Al-Mahdawi S, Pinto RM, Varshney D,Lawrence L, Lowrie MB, Hughes S,Webster Z, Blake J, Cooper JM, King R,Pook MA (2006) GAA repeat expansionmutation mouse models of Friedreichataxia exhibit oxidative stressleading to progressive neuronal andcardiac pathology. Genomics 88:580–590
Al-Mahdawi S, Pinto RM, Ismail O,Varshney D, Lymperi S, Sandi C,Trabzuni D, Pook M (2008) The Friedreichataxia GAA repeat expansionmutation induces comparable epigeneticchanges in human and transgenicmouse brain and heart tissues.Hum Mol Genet 17:735–746
Ventura N, Rea S, Henderson ST,Condo I, Johnson TE, Testi R (2005)Reduced expression of frataxin extendsthe lifespan of Caenorhabditis elegans. Aging Cell 4:109–112
Vazquez-Manrique RP, Gonzalez-CaboP, Ros S, Aziz H, Baylis HA, Palau F(2006) Reduction of Caenorhabditis elegans frataxin increases sensitivity tooxidative stress, reduces lifespan, andcauses lethality in a mitochondrialcomplex II mutant. FASEB J 20:172–174
Zarse K, Schulz TJ, Birringer M, RistowM (2007) Impaired respiration is positivelycorrelated with decreased lifespan in Caenorhabditis elegans modelsof Friedreich ataxia. FASEB J 21:1271–1275
Rea SL, Ventura N, Johnson TE (2007)Relationship between mitochondrialelectron transport chain dysfunction,development, and life extension inCaenorhabditis elegans. PLoS Biol5:e259
Anderson PR, Kirby K, Hilliker AJ,Phillips JP (2005) RNAi-mediatedsuppression of the mitochondrial ironchaperone, frataxin, in Drosophila.Hum Mol Genet 14:3397–3405
Llorens JV, Navarro JA, Martínez-Sebastián MJ, Baylies MK, SchneuwlyS, Botella JA, Moltó MD (2007) Causativerole of oxidative stress in aDrosophila model of Friedreich ataxia.FASEB J 21:333–344
Bulteau AL, O’Neill HA, Kennedy MC,Ikeda-Saito M, Isaya G, Szweda LI(2004) Frataxin acts as an iron chaperoneprotein to modulate mitochondrialaconitase activity. Science 305:242–245
Gakh O, Park S, Liu G, Macomber L,Imlay JA, Ferreira GC, Isaya G (2006)Mitochondrial iron detoxification is aprimary function of frataxin thatlimits oxidative damage and preservescell longevity. Hum Mol Genet 15:467–479
Foury F, Cazzalini O (1997) Deletion ofthe yeast homologue of the humangene associated with Friedreich’sataxia elicits iron accumulation inmitochondria. FEBS Lett 411:373–377
Di Prospero NA, Fischbeck KH (2005)Therapeutics development for tripletrepeat expansion diseases. Nat RevGenet 6:756–765
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Puccio, H. Multicellular models of Friedreich ataxia. J Neurol 256 (Suppl 1), 18–24 (2009). https://doi.org/10.1007/s00415-009-1004-1
Issue Date:
DOI: https://doi.org/10.1007/s00415-009-1004-1