Abstract
Background
The aim of the present study was to assess the regional variation in cerebral perfusion, vasomotor reactivity (VMR) and the role of cerebral collaterals in patients with symptomatic internal carotid artery (ICA).
Methods
Seventeen functionally independent patients (60±9 years, mean±SD) with a unilateral symptomatic internal carotid artery occlusion and a <30% contralateral ICA stenosis were investigated. (99 m) Tc-hexamethyl propyleneamine oxime (HMPAO) single photon emission computed tomography (SPECT) was performed to study cerebral blood flow in rest and during a CO2 challenge in the cerebellum, temporal lobe, occipital lobe, basal ganglia, frontal lobe and parietal lobe. Time of flight and phase contrast MRA were used to study collateral flow via circle of Willis.
Results
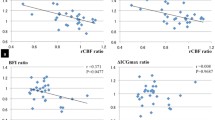
In rest, cerebral perfusion on the side ipsilateral to the ICA occlusion was decreased compared with the contralateral side in the basal ganglia (p<0.05), frontal lobe (p<0.01) and parietal lobe (p<0.01). During a CO2 challenge only the ipsilateral frontal lobe demonstrated a perfusion decrease compared with the contralateral frontal lobe (p<0.05). Furthermore, in patients without collateral flow via the anterior circle of Willis the perfusion of the ipsilateral frontal lobe was significantly decreased (p<0.01) during the CO2 challenge and crossed cerebellar diaschisis with a decreased perfusion on the contralateral cerebellar hemisphere was detected (p<0.05). No cerebral blood flow (CBF) differences were found for present/absent collateral flow via the posterior communicating artery.
Conclusion
Regional assessment of cerebral perfusion and VMR with SPECT demonstrated the heterogeneity of cerebral hemodynamics and the importance of collateral flow via the anterior circle of Willis.




Similar content being viewed by others
References
Adams HP Jr., Powers WJ, Grubb RL Jr., Clarke WR, Woolson RF (2001) Preview of a new trial of extracranial-to-intracranial arterial anastomosis: the carotid occlusion surgery study. Neurosurg Clin N Am 12:613–624
Arakawa S, Minematsu K, Hirano T, Tanaka Y, Hasegawa Y, Hayashida K, Yamaguchi T (2003) Topographic distribution of misery perfusion in relation to internal and superficial borderzones. AJNR Am J Neuroradiol 24:427–435
Bamford JM, Sandercock PAG, Warlow CP, Slattery J (1989) Interobserver agreement for the assessment of handicap in stroke patients. Stroke 20:828
Barnett HJ, Meldrum HE (2001) Endarterectomy for carotid stenosis: new approaches in patient selection. Cerebrovasc Dis 11:105–11
Barrett KM, Ackerman RH, Gahn G, Romero JM, Candia M (2001) Basilar and middle cerebral artery reserve: a comparative study using transcranial Doppler and breath-holding techniques. Stroke 32:2793–2796
Brozici M, van der Zwan A, Hillen B (2003) Anatomy and functionality of leptomeningeal anastomoses: a review. Stroke 34:2750–2762
Derdeyn CP, Grubb RL Jr., Powers WJ (1999) Cerebral hemodynamic impairment: methods of measurement and association with stroke risk. Neurology 53:251–259
Gordon IL, Stemmer EA, Wilson SE (1995) Redistribution of blood flow after carotid endarterectomy. J Vasc Surg 22:349–358
Gur AY, Bornstein NM (2003) Cerebral vasomotor reactivity of the posterior circulation in patients with carotid occlusive disease. Eur J Neurol 10:75–78
Henderson RD, Eliasziw M, Fox AJ, Rothwell PM, Barnett HJM (2000) Angiographically defined collateral circulation and risk of stroke in patients with severe carotid artery stenosis. Stroke 31:128–132
Hendrikse J, Hartkamp MJ, Hillen B, Mali WP, Van der Grond J (2001) Collateral ability of the circle of Willis in patients with unilateral internal carotid artery occlusion: border zone infarcts and clinical symptoms. Stroke 32:2768–2773
Imaizumi M, Kitagawa K, Hashikawa K, Oku N, Teratani T, Takasawa M, Yoshikawa T, Rishu P, Ohtsuki T, Hori M, Matsumoto M, Nishimura T (2002) Detection of misery perfusion with split-dose 123I-iodoamphetamine single-photon emission computed tomography in patients with carotid occlusive diseases. Stroke 33:2217–2223
Infeld B, Davis SM, Lichtenstein M, Mitchell PJ, Hopper JL (1995) Crossed cerebellar diaschisis and brain recovery after stroke. Stroke 26:90–95
Kaminogo M, Ochi M, Onizuka M, Takahata H, Shibata S (1999) An additional monitoring of regional cerebral oxygen saturation to HMPAO SPECT study during balloon test occlusion. Stroke 30:407–413
Kim GE, Cho YP, Lim SM (2002) The anatomy of the circle of Willis as a predictive factor for intra-operative cerebral ischemia (shunt need) during carotid endarterectomy. Neurol Res 24:237–240
Klijn CJM, Kappelle LJ, Tulleken CAF, van Gijn J (1997) Symptomatic carotid artery occlusion. A reappraisal of hemodynamic factors. Stroke 28:2084–2093
Leblanc R, Yamamoto YL, Tyler JL, Diksic M, Hakim A (1987) Borderzone ischemia. Ann Neurol 22:707–713
Liebeskind DS (2003) Collateral Circulation. Stroke 34:2279–2284
Markus H, Cullinane M (2001) Severely impaired cerebrovascular reactivity predicts stroke and TIA risk in patients with carotid artery stenosis and occlusion. Brain 124:457–467
Miralles M, Dolz JL, Cotillas J, Aldoma J, Santiso MA, Gimenez A, Capdevila A, Cairols MA (1995) The role of the circle of Willis in carotid occlusion: assessment with phase contrast MR angiography and transcranial duplex. Eur J Vasc Endovasc Surg 10:424–430
Momjian-Mayor I, Baron JC (2005) The pathophysiology of watershed infarction in internal carotid artery disease: review of cerebral perfusion studies. Stroke 36:567–577
Norrving B, Nilsson B, Risbert J (1982) rCBF in patients with carotid occlusion. Resting and hypercapnic flow related to collateral pattern. Stroke 13:155–162
Ogasawara K, Ogawa A, Yoshimoto T (2002) Cerebrovascular reactivity to acetazolamide and outcome in patients with symptomatic internal carotid or middle cerebral artery occlusion: a xenon-133 single-photon emission computed tomography study. Stroke 33:1857–1862
Ozgur HT, Kent WT, Masaryk A, Seeger JF, Williams W, Krupinski E, Melgar M, Labadie E (2001) Correlation of cerebrovascular reserve as measured by acetazolamide-challenged SPECT with angiographic flow patterns and intra- or extracranial arterial stenosis. AJNR Am J Neuroradiol 22:928–936
Park CW, Sturzenegger M, Douville CM, Aaslid R, Newell DW (2003) Autoregulatory response and CO2 reactivity of the basilar artery. Stroke 34:34–39
Schomer DF, Marks MP, Steinberg GK, Johnstone IM, Boothroyd DB, Ross MR, Pelc NJ, Enzmann DR (1994) The anatomy of the posterior communicating artery as a risk factor for ischemic cerebral infarction. N Engl J Med 330:1565–1570
Srinivasan A, Miller W, Stys P, Goyal M (2004) Crossed cerebellar diaschisis in stroke. Neurology 62:2130
Van Everdingen KJ, Visser GH, Klijn CJM, Kappelle LJ, Van der Grond J (1998) Role of collateral flow on cerebral hemodynamics in patients with unilateral internal carotid artery occlusion. Ann Neurol 44:167–176
Vanninen R, Koivisto K, Tulla H, Manninen H, Partanen K (1995) Hemodynamic effects of carotid endarterectomy by magnetic resonance flow quantification. Stroke 26:84–89
Vernieri F, Pasqualetti P, Matteis M, Passarelli F, Troisi E, Rossini PM, Caltagirone C, Silvestrini M (2001) Effect of collateral blood flow and cerebral vasomotor reactivity on the outcome of carotid artery occlusion. Stroke 32:1552–1558
Vernieri F, Pasqualetti P, Passarelli F, Rossini PM, Silvestrini M (1999) Outcome of carotid artery occlusion is predicted by cerebrovascular reactivity. Stroke 30:593–598
Zachrisson H, Berthelsen B, Blomstrand C, Holm J, Volkmann R (2001) Influence of poststenotic collateral pressure on blood flow velocities within high-grade carotid artery stenosis: differences between morphologic and functional measurements. J Vasc Surg 34:263–268
Zbornikova V, Lassvik C (1996) Relationship between the flow pattern and vasomotor reactivity in the ophthalmic artery, siphon and vessels within the circle of Willis in the unilateral internal carotid artery occlusion. Neurol Res 18:521–527
Author information
Authors and Affiliations
Corresponding author
Additional information
Received in revised form: 22 November 2005
Rights and permissions
About this article
Cite this article
de Boorder, M.J., van der Grond, J., van Dongen, A.J. et al. Spect measurements of regional cerebral perfusion and carbondioxide reactivity: Correlation with cerebral collaterals in internal carotid artery occlusive disease. J Neurol 253, 1285–1291 (2006). https://doi.org/10.1007/s00415-006-0192-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-006-0192-1