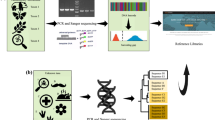
Abstract
Among the repetitive elements, satellite DNA (SatDNA) emerges as extensive arrays of highly similar tandemly repeated units, spanning megabases in length. Given that the satDNA PboSat01-176, previously characterized in P. boiei, prompted our interest for having a high abundance in P. boiei and potential for centromeric satellite, here, we employed various approaches, including low coverage genome sequencing, followed by computational analysis and chromosomal localization techniques in four Proceratophrys species and, investigating the genomic presence and sharing, as well as its potential for chromosomal centromere marker in Proceratophrys frog species. Our findings demonstrate that PboSat01-176 exhibits high abundance across all four Proceratophrys species, displaying distinct characteristics that establish it as the predominant repetitive DNA element in these species. The satellite DNA is prominently clustered in the peri/centromeric region of the chromosomes, particularly in the heterochromatic regions. The widespread presence of PboSat01-176 in closely related Proceratophrys species reinforces the validity of the library hypothesis for repetitive sequences. Thus, this study highlighted the utility of the satDNA family PboSat01-176 as a reliable centromeric marker in Proceratophrys species, with potential to be applied in other species of anuran amphibians. The observed sharing and maintenance of this sequence within the genus suggest possibilities for future research, particularly through expanded sampling to elucidate parameters that underlie the library hypothesis and the evolutionary dynamics of satDNA sequences.



Similar content being viewed by others
Data availability
Not applicable.
References
Amaro RC, Carnaval ACQ, Yonenaga-Yassuda Y, Rodrigues MT (2012) Demographic process in montane Atlantic rainforest: Molecular and cytogenetic evidence from the endemic frog Proceratophrys boiei. Mol Phylogenet Evol 62:880–888. https://doi.org/10.1016/j.ympev.2011.11.004
Ananias F, Modesto ADS, Mendes SC, Napoli MF (2007) Unusual primitive heteromorphic ZZ/ZW sex chromosomes in Proceratophrys boiei (Anura, Cycloramphidae, Alsodinae), with description of C-Band interpopulational polymorphism. Hereditas 144:206–212. https://doi.org/10.1111/j.2007.0018-0661.02026.x
Bardella VB, Milani D, Cabral-de-Mello DC (2020) Analysis of Holhymenia histrio genome provides insight into the satDNA evolution in an insect with holocentric chromosomes. Chromosom Res 28:369–380. https://doi.org/10.1007/s10577-020-09642-1
Cabral-de-Mello DC, Marec F (2021) Universal fluorescence in situ hybridization (FISH) protocol for mapping repetitive DNAs in insects and other arthropods. Mol Genet Genomics 296:513–526. https://doi.org/10.1007/s00438-021-01765-2
Catania S, Pidoux AL, Allshire RC (2015) Sequence features and transcriptional stalling within centromere DNA promote establishment of CENP-A chromatin. PLoS Genet 11:e1004986. https://doi.org/10.1371/journal.pgen.1004986
Da Silva MJ, Destro RF, Gazoni T, Narimatsu H, Pereira Dos Santos PS, Haddad CFB, Parise-Maltempi PP (2020) Great abundance of satellite DNA in Proceratophrys (Anura, Odontophrynidae) revealed by genome sequencing. Cytogenet Genome Res 160(3):141–147. https://doi.org/10.1159/000506531
Drummond A, Ashton B, Cheung M, Heled J, Kearse M, Moir R et al (2015) Geneious v4. 8. 2009. [Online]. Available: http://www.geneious.com (Accessed 01, 2024)
Frost DR (2023) Amphibian Species of the World: an Online Reference. Version 6.0 (American Museum of Natural History, New York 2023). http://research.amnh.org/herpetology/amphibia/index.html
Fry K, Salser W (1977) Nucleotide sequences of HS-Satellite DNA from Kangaroo Rat Dipodomys Ordii and characterization of similar sequences in other rodents. Cell 12:1069–1084. https://doi.org/10.1016/0092-8674(77)90170-2
Garrido-Ramos MA (2017) Satellite DNA: an evolving topic. Genes 8:230. https://doi.org/10.3390/genes8090230
Gatto KP, Seger KR, Garcia PCA, Lourenco LB (2019) Satellite DNA mapping in Pseudis fusca (Hylidae, Pseudinae) provides new insights into sex chromosome evolution in paradoxical frogs. Genes 10:160. https://doi.org/10.3390/genes10020160
Guzmán K, Roco ÁS, Stöck M, Ruiz-García A, García-Muñoz E, Bullejos M (2022) Identification and characterization of a new family of long satellite DNA, specific of true toads (Anura, Amphibia, Bufonidae). Sci Rep 17:13960. https://doi.org/10.1038/s41598-022-18051-9
Henikoff S, Ahmad K, Malik HS (2001) The centromere paradox: stable inheritance with rapidly evolving DNA. Science 293:1098–1102. https://doi.org/10.1126/science.1062939
João Da Silva M, Gazoni T, Haddad CFB, Parise-Maltempi PP (2023) Analysis in Proceratophrys boiei genome illuminates the satellite DNA content in a frog from the Brazilian Atlantic forest. Front Genet 14:1101397. https://doi.org/10.3389/fgene.2023.1101397
Junier T, Pagni M (2000) Dotlet: diagonal plots in a web browser. Bioinformatics 16:178–179
Louzada S, Lopes M, Ferreira D, Adega F, Escudeiro A, Gama-Carvalho M, Chaves R (2020) Decoding the role of Satellite DNA in Genome Architecture and plasticity - an Evolutionary and Clinical Affair. Genes 11:72. https://doi.org/10.3390/genes11010072
Mângia S, Oliveira EF, Santana DJ, Koroiva R, Paiva F, Garda AA (2020) Revising the taxonomy of Proceratophrys Miranda-Ribeiro, 1920 (Anura: Odontophrynidae) from the Brazilian semiarid Caatinga: morphology, calls and molecules support a single widespread species. J Zool Syst Evol Res 58(4):1151–1172. https://doi.org/10.1111/jzs.12365
Melters DP, Bradnam KR, Young HA, Telis N, May MR, Ruby JG, Sebra R, Peluso P, EidJ, Rank D, Garcia JF, DeRisi JL, Smith T, Tobias C, Ross-Ibarra J, Korf I, Chan SWL (2013) Comparative analysis of tandem repeats from hundreds of species reveals unique insights into centromere evolution. Genome Bio 14:R10. https://doi.org/10.1186/gb-2013-14-1-r10
Novák P, Neumann P, Pech J, Steinhaisl J, Macas J (2013) RepeatExplorer: a galaxy-based web server for genome-wide characterization of eukaryotic repetitive elements from next-generation sequence reads. Bioinformatics 29(6):792–793. https://doi.org/10.1093/bioinformatics/btt054
Novák P, Neumann P, Macas J (2020) Global analysis of repetitive DNA from unassembled sequence reads using RepeatExplorer2. Nat Protoc 15(11):3745–3776. https://doi.org/10.1038/s41596-020-0400-y
Palacios-Gimenez OM, Milani D, Song H, Marti DA, López-León MD, Ruiz-Ruano FJ, Camacho JPM, Cabral-De-Mello DC, O’Neill R (2020) Eight million years of Satellite DNA evolution in Grasshoppers of the Genus Schistocerca illuminate the Ins and outs of the Library hypothesis. Genome Biol Evol 12:88–102. https://doi.org/10.1093/gbe/evaa018
Pereira JA, Cabral-de-Mello DC, Lopes DM (2023) The Satellite DNAs populating the genome of Trigona hyalinata and the sharing of a highly abundant satDNA in Trigona Genus. Genes 14:418. https://doi.org/10.3390/genes14020418
Pinkel D, Straume T, Gray JW (1986) Cytogenetic analysis using quantitative, high-sensitivity, fluorescence hybridization. Proc Natl Acad Sci 83(9):2934–2938. https://doi.org/10.1073/pnas.83.9.2934
Plohl M, Meštrovi´c N, Mravinac B (2012) Satellite DNA evolution. Genome Dyn 7:126–152. https://doi.org/10.1159/000337122
Plohl M, Meštrovi´c N, Mravinac B (2014) Centromere identity from the DNA point of view. Chromosoma 123:313–325. https://doi.org/10.1007/s00412-014-0462-0
R Core Team (2021) R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing. URL: https://www.Rproject.org/
Rudd MK, Wray GA, Willard HF (2006) The evolutionary dynamics of alpha-satellite. Genome Res 16:88–96. https://doi.org/10.1101/gr.3810906
Šatovi´c-Vukši´c E, Plohl M (2023) Satellite DNAs—From localized to highly dispersed Genome Components. Genes 14:742. https://doi.org/10.3390/genes14030742
Silva MJ, Santos MD, Gazoni T, Cholak LR, Haddad CFB, Parise-Maltempi PP (2021) Cytogenetic approaches provide evidence of a conserved diploid number and cytological differences between Proceratophrys species (Anura: Odontophrynidae). Acad Bras Cienc 93:e20201650. https://doi.org/10.1590/0001-3765202120201650
Smit AFA, Hubley R, Green P (2013) Repeat masker. [Online]. Available: http://www.repeatmasker.org/ (Accessed 05, 2023)
Vittorazzi SE, Lourenço LB, Del-Grande ML, Recco-Pimentel SM (2011) Satellite DNA derived from 5S rDNA in Physalaemus cuvieri. Cytogenet Genome Res 134:101–107. https://doi.org/10.1159/000325540. Anura, Leiuperidae
Acknowledgements
We would like to thank members of the Animal Cytogenetics Laboratory (UNESP) for the valuable suggestions, constructive comments, and critical reading of an earlier version of the manuscript, especially Ms. Carolina Crepaldi. We would also like to thank Prof. Diogo Cavalcanti Cabral-de-Mello for the valuable suggestions, access to genome sequencing, and access to all computational resources. We would also like to thank Prof. Célio Fernando Baptista Haddad and Dra Luiza Rieder Cholak for their kindness in collecting part of the Proceratophrys specimens analyzed in this work.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) - Finance Code 001 and by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP #2017/00195_7).
Author information
Authors and Affiliations
Contributions
MJS contributed to the work’s conception, data acquisition, analysis and interpretation, and wrote the manuscript. MJS and TG contributed to the acquisition of specimens for biological and genomic material. MJS and RFD conducted cytogenetic analysis. PPM conceptualized, funded and coordinated the research. All authors contributed to manuscript revision, read and approved the submitted version.
Corresponding author
Ethics declarations
Ethical statement
The animal study was reviewed and approved by Animal Ethics Committee (Comitê de Ética no Uso de Animais - CEUA - permission 21/2019), Biosciences Institute, UNESP, Rio Claro, SP, Brazil.
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethics approval and consent to participate
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
da Silva, M.J., Destro, R.F., Gazoni, T. et al. Interspecific cytogenomic comparison reveals a potential chromosomal centromeric marker in Proceratophrys frog species. Chromosoma (2024). https://doi.org/10.1007/s00412-024-00819-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00412-024-00819-9