Abstract
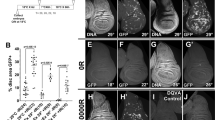
Homologous recombination (HR) is one of the key pathways to repair double-strand breaks (DSBs). Rad51 serves an important function of catalysing strand exchange between two homologous sequences in the HR pathway. In higher organisms, rad51 function is indispensable with its absence leading to early embryonic lethality, thus precluding any mechanistic probing of the system. In contrast, the absence of Drosophila rad51 (spn-A/rad51) has been associated with defects in the germline, without any reported detrimental consequences to Drosophila somatic tissues. In this study, we have performed a systematic analysis of developmental defects in somatic tissues of spn-A mutant flies by using genetic complementation between multiple spn-A alleles. Our current study, for the first time, uncovers a requirement for spn-A in somatic tissue maintenance during both larval and pupal stages. Also, we show that spn-A mutant exhibits patterning defects in abdominal cuticle in the stripes and bristles, while there appear to be only subtle defects in the adult wing and eye. Interestingly, spn-A mutant shows a discernible phenotype of low temperature sensitivity, suggesting a role of spn-A in temperature sensitive cellular processes. In summary, our study describes the important role played by spn-A/rad51 in Drosophila somatic tissues.





Similar content being viewed by others
References
Abdu U, Brodsky M, Schüpbach T (2002) Activation of a meiotic checkpoint during Drosophila oogenesis regulates the translation of gurken through Chk2/Mnk. Curr Biol 12:1645–1651. https://doi.org/10.1016/S0960-9822(02)01165-X
Abdu U, Gonza A, Ghabrial A, Schu T (2003) The Drosophila Spn-D gene encodes a RAD51C-like protein that is required exclusively during meiosis. Genetics 165:197–204
Awano N, Xu C, Ke H, Inoue K, Inouye M, Phadtare S (2007) Complementation analysis of the cold-sensitive phenotype of the Escherichia Coli CsdA deletion strain. J Bacteriol 189(16):5808–5815. https://doi.org/10.1128/JB.00655-07
Baliga C, Majhi S, Mondal K, Bhattacharjee A, Vijayraghavan K (2016) Rational elicitation of cold-sensitive phenotypes. PNAS 113(18):E2506–E2515. https://doi.org/10.1073/pnas.1604190113
Bartek J, Bartkova J, Lukas J (2007) DNA damage signalling guards against activated oncogenes and tumour progression. Oncogene 26:7773–7779. https://doi.org/10.1038/sj.onc.1210881
Baumann P, West SC (1998) Role of the human RAD51 protein in homologous recombination and double-stranded-break repair. Trends Biochem Sci 23(7):247–251. https://doi.org/10.1016/S0968-0004(98)01232-8
Bergantiños C, Corominas M, Serras F (2010) Cell death-induced regeneration in wing imaginal discs requires JNK signalling. Development (Cambridge, England) 137:1169–1179. https://doi.org/10.1242/dev.045559
Cannan WJ, Pederson DS (2016) Mechanisms and consequences of double-strand DNA break formation in chromatin. J Cell Ph J Cell Physiol 231(1):3–14. https://doi.org/10.1002/jcp.25048.Mechanisms
Cappelli E, Townsend S, Griffin C, Thacker J (2011) Homologous recombination proteins are associated with centrosomes and are required for mitotic stability. Exp Cell Res 317(8):1203–1213. https://doi.org/10.1016/j.yexcr.2011.01.021
Clemente-Ruiz M, Murillo-Maldonado JM, Benhra N, Quiroga G, Nebreda AR, Marco MN (2016) Gene dosage imbalance contributes to chromosomal instability-induced tumorigenesis. 290–302. https://doi.org/10.1016/j.devcel.2016.01.008
Do AT, Brooks JT, Le Neveu MK, LaRocque JR (2014) Double-strand break repair assays determine pathway choice and structure of gene conversion events in Drosophila melanogaster. G3: Genes Genomes Genet 4(3):425–432. https://doi.org/10.1534/g3.113.010074
Fasullo M, Giallanza P, Zheng D, Cera C, Bennett T (2001) Saccharomyces cerevisiae Rad51 mutants are defective in DNA damage-associated sister chromatid exchanges but exhibit increased rates of homology-directed translocations. Genetics 158(3):959–972
Fregoso M, Lainé J-P, Aguilar-Fuentes J, Mocquet V, Reynaud E, Coin F, Egly J-M, Zurita M (2007) DNA repair and transcriptional deficiencies caused by mutations in the Drosophila P52 subunit of TFIIH generate developmental defects and chromosome fragility. Mol Cell Biol 27(10):3640–3650. https://doi.org/10.1128/MCB.00030-07
Ghabrial A, Ray RP, Schüpbach T (1998) Okra and spindle-B encode components of the RAD52 DNA repair pathway and affect meiosis and patterning in Drosophila oogenesis. Genes Dev 12(17):2711–2723. https://doi.org/10.1101/gad.12.17.2711
González-Reyes A, Elliott H, St Johnston D (1997) Oocyte determination and the origin of polarity in Drosophila: the role of the spindle genes. Development 124:4927–4937
Haynie JL, Bryant PJ (1977) The effects of X-rays on the proliferation dynamics of cells in the imaginal wing disc of Drosophila melanogaster. Wilhelm Roux’s Arch Dev Biol 183(2):85–100. https://doi.org/10.1007/BF00848779
Helleday T, Eshtad S, Nik-zainal S (2014) Mechanisms underlying mutational. Nat Publ Group 15(9):585–598. https://doi.org/10.1038/nrg3729
Huang X, Li J, Bao F, Zhang X, Yang S (2010) A gain-of-function mutation in the Arabidopsis disease resistance gene RPP4 confers sensitivity to low temperature. Plant Physiol 154(October):796–809. https://doi.org/10.1104/pp.110.157610
Jeggo PA, Lo M (2007) DNA double-strand breaks: their cellular and clinical impact? Oncogene 26(56):7717–7719. https://doi.org/10.1038/sj.onc.1210868
Johnson-Schlitz DM, Flores C, Engels WR (2007) Multiple-pathway analysis of double-strand break repair mutations in Drosophila. PLoS Genet 3(4):0544–0558. https://doi.org/10.1371/journal.pgen.0030050
Kelso AA, Goodson SD, Temesvari LA, Sehorn MG (2017) Data in brief data on Rad51 amino acid sequences from higher and lower eukaryotic model organisms and parasites. Data Brief 10:364–368. https://doi.org/10.1016/j.dib.2016.12.002
Khan C, Muliyil S, Ayyub C, Rao BJ (2017) The initiator caspase Dronc plays a non-apoptotic role in promoting DNA damage signalling in D. melanogaster. J Cell Sci 130(18). https://doi.org/10.1242/jcs.200782
Klovstad M, Abdu U, Schüpbach T (2008) Drosophila Brca2 is required for mitotic and meiotic DNA repair and efficient activation of the meiotic recombination checkpoint. PLoS Genet 4(2):e31. https://doi.org/10.1371/journal.pgen.0040031
Larson JR, Facemyer EM, Shen K-f, Ukil L, Osmani SA (2014) Insights into dynamic mitotic chromatin organization through the NIMA kinase. Genetics 196(January):177–195. https://doi.org/10.1534/genetics.113.156745
Lieber MR (1998) Pathological and physiological double-strand breaks. Am J Pathol 153(5):1323–1332. https://doi.org/10.1016/S0002-9440(10)65716-1
Madigan JP, Chotkowski HL, Glaser RL (2002) DNA double-strand break-induced phosphorylation of Drosophila histone variant H2Av helps prevent radiation-induced apoptosis. Nucleic Acids Res 30(17):3698–3705. https://doi.org/10.1093/nar/gkf496
McCaffrey R, St Johnston D, Gonzlez-Reyes A (2006) Drosophila mus301/spindle-C encodes a helicase with an essential role in double-strand DNA break repair and meiotic progression. Genetics 174:1273–1285. https://doi.org/10.1534/genetics.106.058289
McVey M, LaRocque JR, Adams MD, Sekelsky JJ (2004) Formation of deletions during double-strand break repair in Drosophila DmBlm mutants occurs after strand invasion. Proc Natl Acad Sci U S A 101(44):15694–15699. https://doi.org/10.1073/pnas.0406157101
Morata G, Shlevkov E, Pérez-Garijo A (2011) Mitogenic signaling from apoptotic cells in Drosophila. Develop Growth Differ 53(2):168–176. https://doi.org/10.1111/j.1440-169X.2010.01225.x
Nakazawa N, Arakawa O, Yanagida M (2019) Condensin locates at transcriptional termination sites in mitosis , possibly releasing mitotic transcripts. Open Biol 9(10):190125. https://doi.org/10.1098/rsob.190125
Ninov N, Chiarelli DA, Martín-Blanco E (2007) Extrinsic and intrinsic mechanisms directing epithelial cell sheet replacement during Drosophila metamorphosis. Development (Cambridge, England) 134(2):367–379. https://doi.org/10.1242/dev.02728
Petrini JH, Bressan DA, Yao MS (1997) The RAD52 epistasis group in mammalian double strand break repair. Semin Immunol 9(3):181–188. https://doi.org/10.1006/smim.1997.0067
Poulton JS, Cuningham JC, Peifer M (2014) Acentrosomal Drosophila epithelial cells exhibit abnormal cell division, leading to cell death and compensatory proliferation. Dev Cell 30(6):731–745. https://doi.org/10.1016/j.devcel.2014.08.007
Preston CR, Flores CC, Engels WR (2006) Differential usage of alternative pathways of double-Strand break repair in Drosophila. Genetics 172(2):1055–1068. https://doi.org/10.1534/genetics.105.050138
Radford SJ, Sekelsky JJ (2004) Taking Drosophila Rad51 for a SPiN. Nat Struct Mol Biol 11(January):9–10. https://doi.org/10.1038/nsmb0104-9
Repiso A, Bergantiños C, Corominas M, Serras F (2011) Tissue repair and regeneration in Drosophila imaginal discs. Develop Growth Differ 53(2):177–185. https://doi.org/10.1111/j.1440-169X.2010.01247.x
Ryoo HD, Gorenc T, Steller H (2004) Apoptotic cells can induce compensatory cell proliferation through the JNK and the wingless signaling pathways. Dev Cell 7(4):491–501. https://doi.org/10.1016/j.devcel.2004.08.019
Scott SP, Pandita TK (2006) The cellular control of DNA double-strand breaks. J Cell Biochem 99(6):1463–1475. https://doi.org/10.1002/jcb.21067.The
Shinohara A (1992) Rad51 protein involved in repair and recombination in S. cerevisiae is a reca-like protein. Cell 69:457–470
Shinohara A, Ogawa T (1999) Rad51/RecA protein families and the associated proteins in eukaryotes. Mutat Res DNA Repair 435(1):13–21. https://doi.org/10.1016/S0921-8777(99)00033-6
Sinha AK, Pavankumar TL, Kamisetty S, Mittal P, Ray MK (2013) Replication arrest is a major threat to growth at low temperature in Antarctic Pseudomonas syringae Lz4W. Mol Microbiol 89(July):792–810. https://doi.org/10.1111/mmi.12315
Staeva-Vieira E, Yoo S, Lehmann R (2003) An essential role of DmRad51/SpnA in DNA repair and meiotic checkpoint control. EMBO J 22(21):5863–5874. https://doi.org/10.1093/emboj/cdg564
Strauss EJ, Guthrie C (1991) A cold-sensitive MRNA splicing mutant is a member of the RNA helicase gene family. Genes Dev 5:629–641
Symington LS (2002) Role of RAD52 epistasis group genes in homologous recombination and double-strand break repair. Microbiol Mol Biol Rev 66(4):630–670. https://doi.org/10.1128/MMBR.66.4.630
Tearle RG, Nusslein-Volhard C (1987) Tubingen mutants and stock list. Drosophila Information Service 66:209–269
Tsuzuki T, Fujii Y, Sakumi K, Tominaga Y, Nakao K, Sekiguchi M, Matsushiro A, Yoshimura Y, Morita T (1996) Targeted disruption of the Rad51 gene leads to lethality in embryonic mice. Proc Natl Acad Sci U S A 93(June):6236–6240. https://doi.org/10.1073/pnas.93.13.6236
Vilenchik MM, Knudson AG (2003) Endogenous DNA double-strand breaks: production , fidelity of repair , and induction of cancer. Proc Natl Acad Sci 100(22):12871–12876
Wei DS, Rong YS (2007) A genetic screen for DNA double-strand break repair mutations in Drosophila. Genetics 177(1):63–77. https://doi.org/10.1534/genetics.107.077693
Wells BS, Yoshida E, Johnston LA (2006) Compensatory proliferation in Drosophila imaginal discs requires Dronc-dependent P53 activity. Curr Biol 16(16):1606–1615. https://doi.org/10.1016/j.cub.2006.07.046
Yamaguchi-shinozakit K (2000) Molecular responses to dehydration and low temperature: differences and cross-talk between two stress signaling pathways Kazuo ShinozakP. Curr Opin Plant Biol 3(3):217–223
Yoo S (2006) Characterization of Drosophila Rad51/SpnA protein in DNA binding and embryonic development. Biochem Biophys Res Commun 348(4):1310–1318. https://doi.org/10.1016/j.bbrc.2006.07.211
Yoo S, McKee BD (2005) Functional analysis of the Drosophila Rad51 gene (Spn-a) in repair of DNA damage and meiotic chromosome segregation. DNA Repair 4(2):231–242. https://doi.org/10.1016/j.dnarep.2004.09.009
Acknowledgments
We acknowledge Trudi Schupbach (Princeton University) for spn-A/rad51 mutant stocks; Bhagyashree Chalke and Rudheer D Bapat from TIFR-SEM facility; Maithreyi Narasimha and Ullas Kolthur from TIFR, Trudi Schupbach (Princeton University) and Baena-Lopez LA (University of Oxford, UK) for critical inputs; Bloomington Stock Centre, Developmental Studies Hybridoma Bank, and Vienna Drosophila Resource Centre for reagents; and funding support to BJR from DAE-TIFR and JC Bose award grant (DST).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 62914 kb)
Rights and permissions
About this article
Cite this article
Khan, C., Muliyil, S., Ayyub, C. et al. spn-A/rad51 mutant exhibits enhanced genomic damage, cell death and low temperature sensitivity in somatic tissues. Chromosoma 130, 3–14 (2021). https://doi.org/10.1007/s00412-020-00746-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00412-020-00746-5