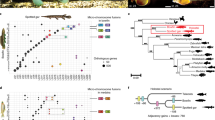
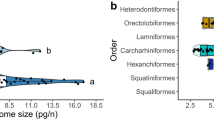
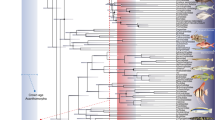
Abstract
Acipenseriformes is an order of ray-finned fishes, comprising 27 extant species of sturgeons and paddlefishes inhabiting waters of the Northern Hemisphere. The order has a basal position within Actinopteri (ray-finned fish minus polypterids) and is characterized by many specific morphological and genomic features, including high diploid chromosome numbers, various levels of ploidy between species, unclear sex determination, and propensity to interspecific hybridization. Recent advances in molecular genetics, genomics, and comparative cytogenetics produced novel data on different aspects of acipenseriform biology, including improved phylogenetic reconstructions and better understanding of genome structure. Here, we discuss the cytogenetic and genomic traits of acipenseriforms and their connection with polyploidization and tolerance to interspecific hybridization.



Similar content being viewed by others
References
Arefjev VA (1989) Karyotype variability in successive generations after hybridization between the great sturgeon, Huso huso (L.), and sterlet, Acipenser ruthenus L. J Fish Biol 35:819–828
Bemis WE, Findeis EK, Grande L (1997) An overview of Acipenseriformes. Environ Biol Fish 48:25–71. doi:10.1023/A:1007370213924
Birstein VJ, DeSalle R (1998) Molecular phylogeny of Acipenserinae. Mol Phylogenet Evol 9:141–155. doi:10.1006/mpev.1997.0443
Birstein VJ, Hanner R, DeSalle R (1997) Phylogeny of the Acipenseriformes: cytogenetic and molecular approaches. Environ Biol Fish 48:127–155. doi:10.1023/A:1007366100353
Braasch I, Gehrke AR, Smith JJ et al (2016) The spotted gar genome illuminates vertebrate evolution and facilitates human-teleost comparisons. Nat Genet 48:427–437. doi:10.1038/ng.3526
Burtzev I (1969) Obtaining offspring from intergeneric hybrid between beluga, Huso huso (L.), and the sterlet, Acipenser ruthenus L. In: Genetics selection and hybridization of fishes. Moscow, Nauka, pp 232–242
Chicca M, Suciu R, Ene C et al (2002) Karyotype characterization of the stellate sturgeon, Acipenser stellatus by chromosome banding and fluorescent in situ hybridization. J Appl Ichthyol 18:298–300. doi:10.1046/j.1439-0426.2002.00378.x
Comai L (2005) The advantages and disadvantages of being polyploid. Nat Rev Genet 6:836–846. doi:10.1038/nrg1711
Dehal P, Boore JL (2005) Two rounds of whole genome duplication in the ancestral vertebrate. PLoS Biol 3, e314. doi:10.1371/journal.pbio.0030314
Devlin RH, Nagahama Y (2002) Sex determination and sex differentiation in fish: an overview of genetic, physiological, and environmental influences. Aquaculture 208:191–364. doi:10.1016/S0044-8486(02)00057-1
Drauch Schreier A, Gille D, Mahardja B, May B (2011) Neutral markers confirm the octoploid origin and reveal spontaneous autopolyploidy in white sturgeon, Acipenser transmontanus. J Appl Ichthyol 27:24–33. doi:10.1111/j.1439-0426.2011.01873.x
Flynn SR, Matsuoka M, Reith M et al (2006) Gynogenesis and sex determination in shortnose sturgeon, Acipenser brevirostrum Lesuere. Aquaculture 253:721–727. doi:10.1016/j.aquaculture.2005.09.016
Fopp-Bayat D (2010) Meiotic gynogenesis revealed not homogametic female sex determination system in Siberian sturgeon (Acipenser baeri Brandt). Aquaculture 305:174–177. doi:10.1016/j.aquaculture.2010.04.011
Gamble T (2016) Using RAD-seq to recognize sex-specific markers and sex chromosome systems. Mol Ecol 25:2114–2116. doi:10.1111/mec.13648
Grunina AS, Rekubratskiĭ AV, Tsvetkova LI et al (2011) Dispermic androgenesis in sturgeons with the help of cryopreserved sperm: production of androgenetic hybrids between Siberian and Russian sturgeons. Ontogenez 42:133–145
Hale MC, McCormick CR, Jackson JR, Dewoody JA (2009) Next-generation pyrosequencing of gonad transcriptomes in the polyploid lake sturgeon (Acipenser fulvescens): the relative merits of normalization and rarefaction in gene discovery. BMC Genomics 10:203. doi:10.1186/1471-2164-10-203
Havelka M, Kaspar V, Hulak M, Flajshans M (2011) Sturgeon genetics and cytogenetics: a review related to ploidy levels and interspecific hybridization. Folia Zool Czech Repub 60:93
Havelka M, Hulák M, Rodina M, Flajšhans M (2013) First evidence of autotriploidization in sterlet (Acipenser ruthenus). J Appl Genet 54:201–207. doi:10.1007/s13353-013-0143-3
Havelka M, Hulák M, Ráb P et al (2014) Fertility of a spontaneous hexaploid male Siberian sturgeon, Acipenser baerii. BMC Genet 15:5. doi:10.1186/1471-2156-15-5
Havelka M, Bytyutskyy D, Symonová R et al (2016) The second highest chromosome count among vertebrates is observed in cultured sturgeon and is associated with genome plasticity. Genet Sel Evol GSE. doi:10.1186/s12711-016-0194-0
Kim DS, Nam YK, Noh JK et al (2005) Karyotype of North American shortnose sturgeon Acipenser brevirostrum with the highest chromosome number in the Acipenseriformes. Ichthyol Res 52:94–97. doi:10.1007/s10228-004-0257-z
Kitano J, Peichel CL (2012) Turnover of sex chromosomes and speciation in fishes. Environ Biol Fish 94:549–558. doi:10.1007/s10641-011-9853-8
Krieger J, Fuerst PA (2002) Evidence for a slowed rate of molecular evolution in the order acipenseriformes. Mol Biol Evol 19:891–897
Li XC, Barringer BC, Barbash DA (2009) The pachytene checkpoint and its relationship to evolutionary patterns of polyploidization and hybrid sterility. Heredity 102:24–30. doi:10.1038/hdy.2008.84
Ludwig A, Belfiore NM, Pitra C et al (2001) Genome duplication events and functional reduction of ploidy levels in sturgeon (Acipenser, Huso and Scaphirhynchus). Genetics 158:1203–1215
Ludwig A, Lippold S, Debus L, Reinartz R (2009) First evidence of hybridization between endangered sterlets (Acipenser ruthenus) and exotic Siberian sturgeons (Acipenser baerii) in the Danube River. Biol Invasions 11:753–760
Lynch M (2002) Gene duplication and evolution. Science 297:945–947. doi:10.1126/science.1075472
Mable BK, Alexandrou MA, Taylor MI (2011) Genome duplication in amphibians and fish: an extended synthesis. J Zool 284:151–182. doi:10.1111/j.1469-7998.2011.00829.x
Nakatani Y, Takeda H, Kohara Y, Morishita S (2007) Reconstruction of the vertebrate ancestral genome reveals dynamic genome reorganization in early vertebrates. Genome Res 17:1254–1265. doi:10.1101/gr.6316407
Ogden R, Gharbi K, Mugue N et al (2013) Sturgeon conservation genomics: SNP discovery and validation using RAD sequencing. Mol Ecol 22:3112–3123. doi:10.1111/mec.12234
Ohno S (1970) Evolution by gene duplication. Springer-Verlag
Ohno S, Muramoto J, Stenius C et al (1969) Microchromosomes in holocephalian, chondrostean and holostean fishes. Chromosoma 26:35–40
Omoto N, Maebayashi M, Adachi S et al (2005) Sex ratios of triploids and gynogenetic diploids induced in the hybrid sturgeon, the bester (Huso huso female×Acipenser ruthenus male). Aquaculture 245:39–47. doi:10.1016/j.aquaculture.2004.12.004
Otto SP (2007) The evolutionary consequences of polyploidy. Cell 131:452–462. doi:10.1016/j.cell.2007.10.022
Peng Z, Ludwig A, Wang D et al (2007) Age and biogeography of major clades in sturgeons and paddlefishes (Pisces: Acipenseriformes). Mol Phylogenet Evol 42:854–862. doi:10.1016/j.ympev.2006.09.008
Phillips RB (2013) Evolution of the sex chromosomes in salmonid fishes. Cytogenet Genome Res 141:177–185. doi:10.1159/000355149
Romanenko SA, Biltueva LS, Serdyukova NA et al (2015) Segmental paleotetraploidy revealed in sterlet (Acipenser ruthenus) genome by chromosome painting. Mol Cytogenet 8:90. doi:10.1186/s13039-015-0194-8
Saber HM, Hallajian A (2014) Study of sex determination system in ship sturgeon, Acipenser nudiventris using meiotic gynogenesis. Aquac Int 22:273–279. doi:10.1007/s10499-013-9676-z
Shelton WL, Mims SD (2012) Evidence for female heterogametic sex determination in paddlefish Polyodon spathula based on gynogenesis. Aquaculture 356–357:116–118. doi:10.1016/j.aquaculture.2012.05.029
Taylor JS, Raes J (2004) Duplication and divergence: the evolution of new genes and old ideas. Annu Rev Genet 38:615–643. doi:10.1146/annurev.genet.38.072902.092831
Van Eenennaam A, Eenennaam JV, Medrano JF, Doroshov SI (1999a) Brief communication. Evidence of female heterogametic genetic sex determination in white sturgeon. J Hered 90:231–233. doi:10.1093/jhered/90.1.231
Van Eenennaam AL, Murray JD, Medrano JF (1999b) Synaptonemal complex analysis in spermatocytes of white sturgeon (Acipenser transmontanus Richardson). J Appl Ichthyol 15:284. doi:10.1111/j.1439-0426.1999.tb00263.x
Vasil’ev VP (2009) Mechanisms of polyploid evolution in fish: polyploidy in sturgeons. In: Carmona R, Domezain A, García-Gallego M, et al. (eds) Biology, conservation and sustainable development of sturgeons. Springer Netherlands, pp 97–117
Vidotto M, Grapputo A, Boscari E et al (2013) Transcriptome sequencing and de novo annotation of the critically endangered Adriatic sturgeon. BMC Genomics 14:407. doi:10.1186/1471-2164-14-407
Wertheim B, Beukeboom LW, van de Zande L (2013) Polyploidy in animals: effects of gene expression on sex determination, evolution and ecology. Cytogenet Genome Res 140:256–269. doi:10.1159/000351998
Wuertz S, Gaillard S, Barbisan F et al (2006) Extensive screening of sturgeon genomes by random screening techniques revealed no sex-specific marker. Aquaculture 258:685–688. doi:10.1016/j.aquaculture.2006.03.042
Yue H, Li C, Du H et al (2015) Sequencing and de novo assembly of the gonadal transcriptome of the endangered Chinese sturgeon (Acipenser sinensis). PLoS One 10, e0127332. doi:10.1371/journal.pone.0127332
Zhou H, Fujimoto T, Adachi S et al (2011) Genome size variation estimated by flow cytometry in Acipenser mikadoi, Huso dauricus in relation to other species of Acipenseriformes. J Appl Ichthyol 27:484–491. doi:10.1111/j.1439-0426.2010.01648.x
Acknowledgments
The work was supported by RSF grant No. 14-14-00275.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval and consent to participate
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
This article does not contain any studies with human participants performed by any of the authors.
Additional information
This article is related to the 21st International Chromosome Conference (Foz do Iguaçu, Brazil, July 10-13, 2016).
Rights and permissions
About this article
Cite this article
Trifonov, V.A., Romanenko, S.S., Beklemisheva, V.R. et al. Evolutionary plasticity of acipenseriform genomes. Chromosoma 125, 661–668 (2016). https://doi.org/10.1007/s00412-016-0609-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00412-016-0609-2