Abstract
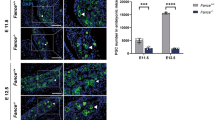
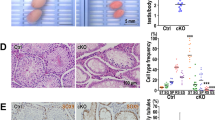
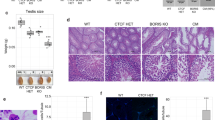
Fancj, the gene associated with Fanconi anemia (FA) Complementation Group J, encodes a DNA helicase involved in homologous recombination repair and the cellular response to replication stress. FANCJ functions in part through its interaction with key DNA repair proteins, including MutL homolog-1 (MLH1), Breast Cancer Associated gene-1 (BRCA1), and Bloom syndrome helicase (BLM). All three of these proteins are involved in a variety of events that ensure genome stability, including the events of DNA double strand break (DSB) repair during prophase I of meiosis. Meiotic DSBs are repaired through homologous recombination resulting in non-crossovers (NCO) or crossovers (CO). The frequency and placement of COs are stringently regulated to ensure that each chromosome receives at least one CO event, and that longer chromosomes receive at least one additional CO, thus facilitating the accurate segregation of homologous chromosomes at the first meiotic division. In the present study, we investigated the role of Fancj during prophase I using a gene trap mutant allele. Fancj GT/GT mutants are fertile, but their testes are very much smaller than wild-type littermates, predominantly as a result of impeded spermatogonial proliferation and mildly increased apoptosis during testis development in the fetus. This defect in spermatogonial proliferation is consistent with mutations in other FA genes. During prophase I, early events of synapsis and DSB induction/repair appear mostly normal in Fancj GT/GT males, and the FANCJ-interacting protein BRCA1 assembles normally on meiotic chromosome cores. However, MLH1 focus frequency is increased in Fancj GT/GT males, indicative of increased DSB repair via CO, and is concomitant with increased chiasmata at diakinesis. This increase in COs in the absence of FANCJ is associated with increased localization of BLM helicase protein, indicating that BLM may facilitate the increased rate of crossing over in Fancj GT/GT males. Taken together, these results demonstrate a critical role for FANCJ in spermatogenesis at two stages: firstly in the proliferative activity that gives rise to the full complement of testicular spermatogonia and secondly in the establishment of appropriate CO numbers during prophase I.







Similar content being viewed by others
References
Agoulnik AI et al (2002) A novel gene, Pog, is necessary for primordial germ cell proliferation in the mouse and underlies the germ cell deficient mutation, gcd. Hum Mol Genet 11:3047–3053
Andreassen PR, Ren K (2009) Fanconi anemia proteins, DNA interstrand crosslink repair pathways, and cancer therapy. Curr Cancer Drug Targets 9:101–117
Baker SM et al (1996) Involvement of mouse Mlh1 in DNA mismatch repair and meiotic crossing over. Nat Genet 13:336–342
Baudat F, Imai Y, de Massy B (2013) Meiotic recombination in mammals: localization and regulation. Nat Rev Genet 14:794–806. doi:10.1038/nrg3573
Berchowitz LE, Francis KE, Bey AL, Copenhaver GP (2007) The role of AtMUS81 in interference-insensitive crossovers in A. thaliana. PLoS Genet 3:e132
Bolcun-Filas E, Schimenti JC (2012) Genetics of meiosis and recombination in mice. Int Rev Cell Mol Biol 298:179–227. doi:10.1016/B978-0-12-394309-5.00005-5
Cantor SB, Xie J (2010) Assessing the link between BACH1/FANCJ and MLH1 in DNA crosslink repair. Environ Mol Mutagen 51:500–507. doi:10.1002/em.20568
Cantor SB et al (2001) BACH1, a novel helicase-like protein, interacts directly with BRCA1 and contributes to its DNA repair function. Cell 105:149–160
Cheng NC et al (2000) Mice with a targeted disruption of the Fanconi anemia homolog Fanca. Hum Mol Genet 9:1805–1811
de Vries SS, Baart EB, Dekker M, Siezen A, de Rooij DG, de Boer P, te Riele H (1999) Mouse MutS-like protein Msh5 is required for proper chromosome synapsis in male and female meiosis. Genes Dev 13:523–531
Edelmann W et al (1996) Meiotic pachytene arrest in MLH1-deficient mice. Cell 85:1125–1134
Edelmann W et al (1999) Mammalian MutS homologue 5 is required for chromosome pairing in meiosis. Nat Genet 21:123–127
Evans EP, Breckon G, Ford CE (1964) An air-drying method for meiotic preparations from mammalian testes. Cytogenet Cell Genet 3:289–294
Ewen KA, Koopman P (2010) Mouse germ cell development: from specification to sex determination. Mol Cell Endocrinol 323:76–93. doi:10.1016/j.mce.2009.12.013
Franklin FC, Higgins JD, Sanchez-Moran E, Armstrong SJ, Osman KE, Jackson N, Jones GH (2006) Control of meiotic recombination in Arabidopsis: role of the MutL and MutS homologues. Biochem Soc Trans 34:542–544. doi:10.1042/BST0340542
Gong Z, Kim JE, Leung CC, Glover JN, Chen J (2010) BACH1/FANCJ acts with TopBP1 and participates early in DNA replication checkpoint control. Mol Cell 37:438–446. doi:10.1016/j.molcel.2010.01.002
Higgins JD, Buckling EF, Franklin FC, Jones GH (2008) Expression and functional analysis of AtMUS81 in Arabidopsis meiosis reveals a role in the second pathway of crossing-over. Plant J 54:152–162
Hiom K (2010) FANCJ: solving problems in DNA replication. DNA Repair 9:250–256. doi:10.1016/j.dnarep.2010.01.005
Hoffmann ER, Borts RH (2004) Meiotic recombination intermediates and mismatch repair proteins. Cytogenet Genome Res 107:232–248
Hollingsworth NM, Ponte L, Halsey C (1995) MSH5, a novel MutS homolog, facilitates meiotic reciprocal recombination between homologs in Saccharomyces cerevisiae but not mismatch repair. Genes Dev 9:1728–1739
Holloway JK, Booth J, Edelmann W, McGowan CH, Cohen PE (2008) MUS81 generates a subset of MLH1-MLH3-independent crossovers in mammalian meiosis. PLoS Genet 4:e1000186
Holloway JK, Morelli MA, Borst PL, Cohen PE (2010) Mammalian BLM helicase is critical for integrating multiple pathways of meiotic recombination. J Cell Biol 188:779–789. doi:10.1083/jcb.200909048
Holloway JK et al. (2011) Mammalian BTBD12 (SLX4) protects against genomic instability during mammalian spermatogenesis. PLoS Genetics 7:e1002094. doi:10.1371/journal.pgen.1002094
Holloway JK, Sun X, Yokoo R, Villeneuve AM, Cohen PE (2014) Mammalian CNTD1 is critical for meiotic crossover maturation and de-selection of excess pre-crossover sites. J Cell Biol 205:633–641
Houghtaling S, Timmers C, Noll M, Finegold MJ, Jones SN, Meyn MS, Grompe M (2003) Epithelial cancer in Fanconi anemia complementation group D2 (Fancd2) knockout mice. Genes Dev 17:2021–2035. doi:10.1101/gad.11034031103403
Jessop L, Lichten M (2008) Mus81/Mms4 endonuclease and Sgs1 helicase collaborate to ensure proper recombination intermediate metabolism during meiosis. Mol Cell 31:313–323
Kneitz B et al (2000) MutS homolog 4 localization to meiotic chromosomes is required for chromosome pairing during meiosis in male and female mice. Genes Dev 14:1085–1097
Knipscheer P et al (2009) The Fanconi anemia pathway promotes replication-dependent DNA interstrand cross-link repair. Science 326:1698–1701. doi:10.1126/science.1182372
Kolas NK et al (2005) Localization of MMR proteins on meiotic chromosomes in mice indicates distinct functions during prophase I. J Cell Biol 171:447–458
Koomen M et al (2002) Reduced fertility and hypersensitivity to mitomycin C characterize Fancg/Xrcc9 null mice. Hum Mol Genet 11:273–281
Kruisselbrink E, Guryev V, Brouwer K, Pontier DB, Cuppen E, Tijsterman M (2008) Mutagenic capacity of endogenous G4 DNA underlies genome instability in FANCJ-defective C. elegans. Curr Biol 18:900–905. doi:10.1016/j.cub.2008.05.013
Kumaraswamy E, Shiekhattar R (2007) Activation of BRCA1/BRCA2-associated helicase BACH1 is required for timely progression through S phase. Mol Cell Biol 27:6733–6741. doi:10.1128/MCB.00961-07
Lawson KA, Hage WJ (1994) Clonal analysis of the origin of primordial germ cells in the mouse. Ciba Found Symp 182:68–84, discussion 84–91
Leung CC, Gong Z, Chen J, Glover JN (2011) Molecular basis of BACH1/FANCJ recognition by TopBP1 in DNA replication checkpoint control. J Biol Chem 286:4292–4301. doi:10.1074/jbc.M110.189555
Levitus M et al (2005) The DNA helicase BRIP1 is defective in Fanconi anemia complementation group. J Nat Genet 37:934–935. doi:10.1038/ng1625
Lipkin SM et al (2002) Meiotic arrest and aneuploidy in MLH3-deficient mice. Nat Genet 31:385–390
Litman R et al (2005) BACH1 is critical for homologous recombination and appears to be the Fanconi anemia gene product FANC J. Cancer Cell 8:255–265. doi:10.1016/j.ccr.2005.08.004
Lu B, Bishop CE (2003) Mouse GGN1 and GGN3, two germ cell-specific proteins from the single gene Ggn, interact with mouse POG and play a role in spermatogenesis. J Biol Chem 278:16289–16296. doi:10.1074/jbc.M211023200
Luo Y, Hartford SA, Zeng R, Southard TL, Shima N, Schimenti JC (2014) Hypersensitivity of primordial germ cells to compromised replication-associated DNA repair involves ATM-p53-p21 signaling. PLoS Genet 10:e1004471. doi:10.1371/journal.pgen.1004471
Mahadevaiah SK et al (2001) Recombinational DNA double-strand breaks in mice precede synapsis. Nat Genet 27:271–276
Modzelewski AJ, Holmes RJ, Hilz S, Grimson A, Cohen PE (2012) AGO4 regulates entry into meiosis and influences silencing of sex chromosomes in the male mouse germline. Dev Cell 23:251–264. doi:10.1016/j.devcel.2012.07.003
Moens PB, Freire R, Tarsounas M, Spyropoulos B, Jackson SP (2000) Expression and nuclear localization of BLM, a chromosome stability protein mutated in Bloom’s syndrome, suggest a role in recombination during meiotic prophase. J Cell Sci 113:663–672
Muniandy PA, Liu J, Majumdar A, Liu ST, Seidman MM (2010) DNA interstrand crosslink repair in mammalian cells: step by step. Crit Rev Biochem Mol Biol 45:23–49. doi:10.3109/10409230903501819
Noll M et al (2002) Fanconi anemia group A and C double-mutant mice: functional evidence for a multi-protein Fanconi anemia complex. Exp Hematol 30:679–688
Oh SD, Lao JP, Taylor AF, Smith GR, Hunter N (2008) RecQ helicase, Sgs1, and XPF family endonuclease, Mus81-Mms4, resolve aberrant joint molecules during meiotic recombination. Mol Cell 31:324–336
Paquis-Flucklinger V, Santucci-Darmanin S, Paul R, Saunieres A, Turc-Carel C, Desnuelle C (1997) Cloning and expression analysis of a meiosis-specific MutS homolog: the human MSH4 gene. Genomics 44:188–194
Peng M, Litman R, Xie J, Sharma S, Brosh RM Jr, Cantor SB (2007) The FANCJ/MutLalpha interaction is required for correction of the cross-link response in FA-J cells. EMBO J 26:3238–3249. doi:10.1038/sj.emboj.7601754
Peng M, Xie J, Ucher A, Stavnezer J, Cantor SB (2014) Crosstalk between BRCA-Fanconi anemia and mismatch repair pathways prevents MSH2-dependent aberrant DNA damage responses. EMBO J 33:1698–1712. doi:10.15252/embj.201387530
Phadnis N, Hyppa RW, Smith GR (2011) New and old ways to control meiotic recombination. Trends Genet 27:411–421. doi:10.1016/j.tig.2011.06.007
Qiao H et al (2014) Antagonistic roles of ubiquitin ligase HEI10 and SUMO ligase RNF212 regulate meiotic recombination. Nat Genet 46:194–199. doi:10.1038/ng.2858
Rosselli F, Briot D, Pichierri (2003) The Fanconi anemia pathway and the DNA interstrand cross-links repair. Biochimie 85:1175–1184
Ross-Macdonald P, Roeder GS (1994) Mutation of a meiosis-specific MutS homolog decreases crossing over but not mismatch correction. Cell 79:1069–1080
Santucci-Darmanin S, Neyton S, Lespinasse F, Saunieres A, Gaudray P, Paquis-Flucklinger V (2002) The DNA mismatch-repair MLH3 protein interacts with MSH4 in meiotic cells, supporting a role for this MutL homolog in mammalian meiotic recombination. Hum Mol Genet 11:1697–1706
Seal S et al (2006) Truncating mutations in the Fanconi anemia J gene BRIP1 are low-penetrance breast cancer susceptibility alleles. Nat Genet 38:1239–1241. doi:10.1038/ng1902
Snowden T, Shim KS, Schmutte C, Acharya S, Fishel R (2008) hMSH4-hMSH5 adenosine nucleotide processing and interactions with homologous recombination machinery. J Biol Chem 283:145–154. doi:10.1074/jbc.M704060200
Suhasini AN, Brosh RM Jr (2011) Fanconi anemia and Bloom’s syndrome crosstalk through FANCJ-BLM helicase interaction. Trend Genet. doi:10.1016/j.tig.2011.09.003
Suhasini AN et al (2011) Interaction between the helicases genetically linked to Fanconi anemia group J and Bloom’s syndrome. EMBO J 30:692–705. doi:10.1038/emboj.2010.362
Tam PP, Snow MH (1981) Proliferation and migration of primordial germ cells during compensatory growth in mouse embryos. J Embryol Exp Morphol 64:133–147
Uroz L, Rajmil O, Templado C (2008) Premature separation of sister chromatids in human male meiosis. Hum Reprod 23:982–987. doi:10.1093/humrep/dem427
Walden H, Deans AJ (2014) The Fanconi anemia DNA repair pathway: structural and functional insights into a complex disorder. Annu Rev Biophys 43:257–278. doi:10.1146/annurev-biophys-051013-022737
Walpita D, Plug AW, Neff NF, German J, Ashley T (1999) Bloom’s syndrome protein, BLM, colocalizes with replication protein A in meiotic prophase nuclei of mammalian spermatocytes. Proc Natl Acad Sci 96:5622–5627
Wang TF, Kleckner N, Hunter N (1999) Functional specificity of MutL homologs in yeast: evidence for three Mlh1-based heterocomplexes with distinct roles during meiosis in recombination and mismatch correction. Proc Natl Acad Sci U S A 96:13914–13919
Wang Y, Cortez D, Yazdi P, Neff N, Elledge SJ, Qin J (2000) BASC, a super complex of BRCA1-associated proteins involved in the recognition and repair of aberrant DNA structures. Genes Dev 14:927–939, In Process Citation
Whitney MA et al (1996) Germ cell defects and hematopoietic hypersensitivity to gamma-interferon in mice with a targeted disruption of the Fanconi anemia C gene. Blood 88:49–58
Williams SA et al (2011) Functional and physical interaction between the mismatch repair and FA-BRCA pathways. Hum Mol Genet. doi:10.1093/hmg/ddr366
Wong JC, Alon N, McKerlie C, Huang JR, Meyn MS, Buchwald M (2003) Targeted disruption of exons 1 to 6 of the Fanconi Anemia group A gene leads to growth retardation, strain-specific microphthalmia, meiotic defects and primordial germ cell hypoplasia. Hum Mol Genet 12:2063–2076
Wu Y, Shin-ya K, Brosh RM Jr (2008) FANCJ helicase defective in Fanconia anemia and breast cancer unwinds G-quadruplex DNA to defend genomic stability. Mol Cell Biol 28:4116–4128. doi:10.1128/MCB.02210-07
Xu Y, Wu X, Her C (2015) hMSH5 facilitates the repair of camptothecin-induced double-strand breaks through an interaction with FANCJ. J Biol Chem. doi:10.1074/jbc.M115.642884
Yang Y et al (2001) Targeted disruption of the murine Fanconi anemia gene, Fancg/Xrcc9. Blood 98:3435–3440
Zou J, Zhang D, Qin G, Chen X, Wang H, Zhang D (2014) BRCA1 and FancJ cooperatively promote interstrand crosslinker induced centrosome amplification through the activation of polo-like kinase 1. Cell Cycle 13:3685–3697. doi:10.4161/15384101.2014.964973
Acknowledgments
We thank Peter Borst for his assistance with mouse handling, genotyping, and maintenance. We are indebted to Dr. Satoshi Namekawa (Cincinnati Children’s Hospital, OH) and Dr. Raimundo Freire (University of Tenerife, Spain) for generously providing antibodies critical for these studies. We thank Simon Boulton (Francis Crick Institute, London) for providing advice and primer sequences for genotyping. We are grateful to Robert Munroe and Christian Abratte of the Cornell Stem Cell and Transgenic Core Facility for performing blastocyst injections. The Cornell Stem Cell and Transgenic Core is supported by NYSDOH contract C024174. This work was supported through funding from the NICHD to J.K.H. (HD065870) and from the NIGMS to P.E.C (GM097263). A.C. was supported by an award from the Dextra Undergraduate Research Endowment fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Laboratory animals were maintained and utilized in accordance with Federal guidelines for the care and use of animals, and under a protocol approved by the Cornell Institutional Animal Care and Use Committee (IACUC). All authors declare that they have no conflicts of interest relating to the work described herein. This work was supported through funding from the NICHD to J.K.H. (HD065870) and from the NIGMS to P.E.C (GM097263). A.C. was supported by an award from the Dextra Undergraduate Research Endowment fund.
Additional information
This article is part of a Special Issue on “Recent advances in meiotic chromosome structure, recombination and segregation” edited by Marco Barchi, Paula Cohen and Scott Keeney.
Xianfei Sun, Miguel A. Brieño-Enriquez and Alyssa Cornelius contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Figure 1
Protein and gene structure for FANCJ and western blot showing loss of the FANCJ protein in Fancj GT/GT testis. (a) Protein structure of mouse FANCJ showing MLH1 interaction domain, helicase domain, and BRCA1 interaction domain. (b) Gene trap allele showing position of the gene trap cassette after exon 5 of the gene. The cassette contains an FRT-flanked βGeo gene. Genotyping primers are indicated. c Western blot showing the loss of 140 kDa FANCJ protein in testis lysates from homozygous mutant genetrap males. A non-specific band of approximately 96 kDa appears in all samples. The same gel was probed with an antibody against β-Tubulin. (GIF 25 kb)
Figure S2
Growth of wildtype (E14 parental) and Fancj GT/GT ES cells over seven days. At least three ES cell clones were grown for each cell line, beginning with equivalent numbers of cells being seeded onto 35 mm petri dishes. Cells were counted daily and replated in fresh medium as needed. (A) shows percentage of cells over the initial seed number, (B) shows raw cell counts for each clone. (GIF 106 kb)
Supplementary Figure 3
Examples of aberrant spermatocyte chromosome spreads from Fancj GT/GT testis. (a) Three cells stained for SYCP3 (green), the centromere (CREST, magenta) and DNA (DAPI, blue), showing many altered synapsis figures (exemplified by the white arrows). (b,c) Examples of γH2AX staining (red) on the SC (SYCP3, green) during mid-pachynema, showing persistent γH2AX staining. (GIF 49 kb)
Rights and permissions
About this article
Cite this article
Sun, X., Brieño-Enríquez, M.A., Cornelius, A. et al. FancJ (Brip1) loss-of-function allele results in spermatogonial cell depletion during embryogenesis and altered processing of crossover sites during meiotic prophase I in mice. Chromosoma 125, 237–252 (2016). https://doi.org/10.1007/s00412-015-0549-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00412-015-0549-2