Abstract
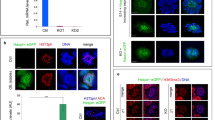
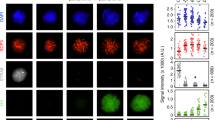
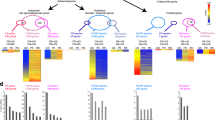
During meiotic prophase I, interactions between maternal and paternal chromosomes, under checkpoint surveillance, establish connections between homologs that promote their accurate distribution to meiotic progeny. In human, faulty meiosis causes aneuploidy resulting in miscarriages and genetic diseases. Meiotic processes occur in the context of chromatin; therefore, histone post-translational modifications are expected to play important roles. Here, we report the cytological distribution of the evolutionarily conserved DOT1L methyltransferase and the different H3K79 methylation states resulting from its activity (mono-, di- and tri-methylation; H3K79me1, me2 and me3, respectively) during meiotic prophase I in mouse spermatocytes. In the wild type, whereas low amounts of H3K79me1 are rather uniformly present throughout prophase I, levels of DOT1L, H3K79me2 and H3K79me3 exhibit a notable increase from pachynema onwards, but with differential subnuclear distribution patterns. The heterochromatic centromeric regions and the sex body are enriched for H3K79me3. In contrast, H3K79me2 is present all over the chromatin, but is largely excluded from the sex body despite the accumulation of DOT1L. In meiosis-defective mouse mutants, the increase of DOT1L and H3K79me is blocked at the same stage where meiosis is arrested. H3K79me patterns, combined with the cytological analysis of the H3.3, γH2AX, macroH2A and H2A.Z histone variants, are consistent with a differential role for these epigenetic marks in male mouse meiotic prophase I. We propose that H3K79me2 is related to transcriptional reactivation on autosomes during pachynema, whereas H3K79me3 may contribute to the maintenance of repressive chromatin at centromeric regions and the sex body.








Similar content being viewed by others
References
Acquaviva L, Székvölgyi L, Dichtl B, Dichtl BS, de La Roche Saint André C, Nicolas A, Géli V (2013) The COMPASS subunit Spp1 links histone methylation to initiation of meiotic recombination. Science 339:215–218. doi:10.1126/science.1225739
Anglin JL, Deng L, Yao Y, Cai G, Liu Z, Jiang H, Cheng G, Chen P et al (2012) Synthesis and structure–activity relationship investigation of adenosine-containing inhibitors of histone methyltransferase DOT1L. J Med Chem 55:8066–8074. doi:10.1021/jm300917h
Barchi M, Mahadevaiah S, Di Giacomo M, Baudat F, de Rooij DG, Burgoyne PS, Jasin M, Keeney S (2005) Surveillance of different recombination defects in mouse spermatocytes yields distinct responses despite elimination at an identical developmental stage. Mol Cell Biol 25:7203–7215. doi:10.1128/MCB.25.16.7203-7215.2005
Barchi M, Roig I, Di Giacomo M, de Rooij DG, Keeney S, Jasin M (2008) ATM promotes the obligate XY crossover and both crossover control and chromosome axis integrity on autosomes. PLoS Genet 4:e1000076. doi:10.1371/journal.pgen.1000076
Barry ER, Krueger W, Jakuba CM, Veilleux E, Ambrosi DJ, Nelson CE, Rasmussen TP (2009) ES cell cycle progression and differentiation require the action of the histone methyltransferase Dot1L. Stem Cells 27:1538–1547. doi:10.1002/stem.86
Barski A, Cuddapah S, Cui K, Roh T-Y, Schones DE, Wang Z, Wei G, Chepelev I et al (2007) High-resolution profiling of histone methylations in the human genome. Cell 129:823–837. doi:10.1016/j.cell.2007.05.009
Baudat F, Manova K, Yuen JP, Jasin M, Keeney S (2000) Chromosome synapsis defects and sexually dimorphic meiotic progression in mice lacking Spo11. Mol Cell 6:989–998
Bernt KM, Zhu N, Sinha AU, Vempati S, Faber J, Krivtsov AV, Feng Z, Punt N et al (2011) MLL-rearranged leukemia is dependent on aberrant H3K79 methylation by DOT1L. Cancer Cell 20:66–78. doi:10.1016/j.ccr.2011.06.010
Bhalla N, Dernburg AF (2005) A conserved checkpoint monitors meiotic chromosome synapsis in Caenorhabditis elegans. Science 310:1683–1686. doi:10.1126/science.1117468
Bolcun-Filas E, Schimenti JC (2012) Genetics of meiosis and recombination in mice. Int Rev Cell Mol Biol 298:179–227. doi:10.1016/B978-0-12-394309-5.00005-5
Borde V, Robine N, Lin W, Bonfils S, Géli V, Nicolas A (2009) Histone H3 lysine 4 trimethylation marks meiotic recombination initiation sites. EMBO J 28:99–111. doi:10.1038/emboj.2008.257
Brachet E, Sommermeyer V, Borde V (2012) Interplay between modifications of chromatin and meiotic recombination hotspots. Biol Cell 104:51–69. doi:10.1111/boc.201100113
Burgoyne PS, Mahadevaiah SK, Turner JMA (2009) The consequences of asynapsis for mammalian meiosis. Nat Rev Genet 10:207–216. doi:10.1038/nrg2505
Castaño Betancourt MC, Cailotto F, Kerkhof HJ, Cornelis FMF, Doherty SA, Hart DJ, Hofman A, Luyten FP et al (2012) Genome-wide association and functional studies identify the DOT1L gene to be involved in cartilage thickness and hip osteoarthritis. Proc Natl Acad Sci U S A 109:8218–8223. doi:10.1073/pnas.1119899109
Cohen PE, Pollack SE, Pollard JW (2006) Genetic analysis of chromosome pairing, recombination, and cell cycle control during first meiotic prophase in mammals. Endocr Rev 27:398–426. doi:10.1210/er.2005-0017
Cowell I, Aucott R, Mahadevaiah S, Huskisson N, Bongiorni S, Prantera G, Fanti L, Pimpinelli S et al (2002) Heterochromatin, HP1 and methylation at lysine 9 of histone H3 in animals. Chromosoma 111:22–36. doi:10.1007/s00412-002-0182-8
Daigle SR, Olhava EJ, Therkelsen CA, Majer CR, Sneeringer CJ, Song J, Johnston LD, Scott MP et al (2011) Selective killing of mixed lineage leukemia cells by a potent small-molecule DOT1L inhibitor. Cancer Cell 20:53–65. doi:10.1016/j.ccr.2011.06.009
de la Fuente R, Parra MT, Viera A, Calvente A, Gómez R, Suja JA, Rufas JS, Page J (2007) Meiotic pairing and segregation of achiasmate sex chromosomes in eutherian mammals: the role of SYCP3 protein. PLoS Genet 3:e198. doi:10.1371/journal.pgen.0030198
de la Fuente R, Sánchez A, Marchal JA, Viera A, Parra MT, Rufas JS, Page J (2012) A synaptonemal complex-derived mechanism for meiotic segregation precedes the evolutionary loss of homology between sex chromosomes in arvicolid mammals. Chromosoma 121:433–446. doi:10.1007/s00412-012-0374-9
De Vos D, Frederiks F, Terweij M, van Welsem T, Verzijlbergen KF, Iachina E, de Graaf EL, Maarten Altelaar AF et al (2011) Progressive methylation of ageing histones by Dot1 functions as a timer. EMBO Rep 12:956–962. doi:10.1038/embor.2011.131
Feng Q, Wang H, Ng HH, Erdjument-Bromage H, Tempst P, Struhl K, Zhang Y (2002) Methylation of H3-lysine 79 is mediated by a new family of HMTases without a SET domain. Curr Biol 12:1052–1058
Feng Y, Yang Y, Ortega MM, Copeland JN, Zhang M, Jacob JB, Fields TA, Vivian JL et al (2010) Early mammalian erythropoiesis requires the Dot1L methyltransferase. Blood 116:4483–4491. doi:10.1182/blood-2010-03-276501
Fernandez-Capetillo O, Mahadevaiah SK, Celeste A, Romanienko PJ, Camerini-Otero RD, Bonner WM, Manova K, Burgoyne P et al (2003) H2AX is required for chromatin remodeling and inactivation of sex chromosomes in male mouse meiosis. Dev Cell 4:497–508
Frederiks F, Tzouros M, Oudgenoeg G, van Welsem T, Fornerod M, Krijgsveld J, van Leeuwen F (2008) Nonprocessive methylation by Dot1 leads to functional redundancy of histone H3K79 methylation states. Nat Struct Mol Biol 15:550–557. doi:10.1038/nsmb.1432
Greaves IK, Rangasamy D, Devoy M, Marshall Graves JA, Tremethick DJ (2006) The X and Y chromosomes assemble into H2A.Z, containing facultative heterochromatin, following meiosis. Mol Cell Biol 26:5394–5405. doi:10.1128/MCB.00519-06
Grey C, Barthès P, Chauveau-Le Friec G, Langa F, Baudat F, de Massy B (2011) Mouse PRDM9 DNA-binding specificity determines sites of histone H3 lysine 4 trimethylation for initiation of meiotic recombination. PLoS Biol 9:e1001176. doi:10.1371/journal.pbio.1001176
Hake SB, Garcia BA, Duncan EM, Kauer M, Dellaire G, Shabanowitz J, Bazett-Jones DP, Allis CD et al (2006) Expression patterns and post-translational modifications associated with mammalian histone H3 variants. J Biol Chem 281:559–568. doi:10.1074/jbc.M509266200
Handel MA (2004) The XY body: a specialized meiotic chromatin domain. Exp Cell Res 296:57–63. doi:10.1016/j.yexcr.2004.03.008
Handel MA, Schimenti JC (2010) Genetics of mammalian meiosis: regulation, dynamics and impact on fertility. Nat Rev Genet 11:124–136. doi:10.1038/nrg2723
Hochwagen A, Amon A (2006) Checking your breaks: surveillance mechanisms of meiotic recombination. Curr Biol 16:R217–228. doi:10.1016/j.cub.2006.03.009
Hoyer-Fender S, Costanzi C, Pehrson JR (2000) Histone macroH2A1.2 is concentrated in the XY-body by the early pachytene stage of spermatogenesis. Exp Cell Res 258:254–260. doi:10.1006/excr.2000.4951
Huyen Y, Zgheib O, Ditullio RA, Gorgoulis VG, Zacharatos P, Petty TJ, Sheston EA, Mellert HS et al (2004) Methylated lysine 79 of histone H3 targets 53BP1 to DNA double-strand breaks. Nature 432:406–411. doi:10.1038/nature03114
Jo SY, Granowicz EM, Maillard I, Thomas D, Hess JL (2011) Requirement for Dot1l in murine postnatal hematopoiesis and leukemogenesis by MLL translocation. Blood 117:4759–4768. doi:10.1182/blood-2010-12-327668
Jones B, Su H, Bhat A, Lei H, Bajko J, Hevi S, Baltus GA, Kadam S et al (2008) The histone H3K79 methyltransferase Dot1L is essential for mammalian development and heterochromatin structure. PLoS Genet 4:e1000190. doi:10.1371/journal.pgen.1000190
Joyce EF, McKim KS (2009) Drosophila PCH2 is required for a pachytene checkpoint that monitors double-strand-break-independent events leading to meiotic crossover formation. Genetics 181:39–51. doi:10.1534/genetics.108.093112
Kalitsis P, Griffiths B, Choo KHA (2006) Mouse telocentric sequences reveal a high rate of homogenization and possible role in Robertsonian translocation. Proc Natl Acad Sci U S A 103:8786–8791. doi:10.1073/pnas.0600250103
Kauppi L, Barchi M, Baudat F, Romanienko PJ, Keeney S, Jasin M (2011) Distinct properties of the XY pseudoautosomal region crucial for male meiosis. Science 331:916–920. doi:10.1126/science.1195774
Keeney S (2008) Spo11 and the formation of DNA double-strand breaks in meiosis. Genome Dyn Stab 2:81–123. doi:10.1007/7050_2007_026
Khalil AM, Driscoll DJ (2010) Epigenetic regulation of pericentromeric heterochromatin during mammalian meiosis. Cytogenet Genome Res 129:280–289. doi:10.1159/000315903
Kim S-K, Jung I, Lee H, Kang K, Kim M, Jeong K, Kwon CS, Han Y-M et al (2012a) Human histone H3K79 methyltransferase DOT1L binds actively transcribing RNA polymerase II to regulate gene expression. J Biol Chem 287:39698–39709. doi:10.1074/jbc.M112.384057
Kim W, Kim R, Park G, Park J-W, Kim J-E (2012b) Deficiency of H3K79 histone methyltransferase Dot1-like protein (DOT1L) inhibits cell proliferation. J Biol Chem 287:5588–5599. doi:10.1074/jbc.M111.328138
Kouskouti A, Talianidis I (2005) Histone modifications defining active genes persist after transcriptional and mitotic inactivation. EMBO J 24:347–357. doi:10.1038/sj.emboj.7600516
Krivtsov AV, Feng Z, Lemieux ME, Faber J, Vempati S, Sinha AU, Xia X, Jesneck J et al (2008) H3K79 methylation profiles define murine and human MLL-AF4 leukemias. Cancer Cell 14:355–368. doi:10.1016/j.ccr.2008.10.001
Lacoste N, Utley RT, Hunter JM, Poirier GG, Côte J (2002) Disruptor of telomeric silencing-1 is a chromatin-specific histone H3 methyltransferase. J Biol Chem 277:30421–30424. doi:10.1074/jbc.C200366200
Lammers JH, Offenberg HH, van Aalderen M, Vink AC, Dietrich AJ, Heyting C (1994) The gene encoding a major component of the lateral elements of synaptonemal complexes of the rat is related to X-linked lymphocyte-regulated genes. Mol Cell Biol 14:1137–1146. doi:10.1128/MCB.14.2.1137.Updated
Li XC, Li X, Schimenti JC (2007) Mouse Pachytene Checkpoint 2 (Trip13) is required for completing meiotic recombination but not synapsis. PLoS Genet 3:e130. doi:10.1371/journal.pgen.0030130
Liebe B, Alsheimer M, Höög C, Benavente R, Scherthan H (2004) Telomere attachment, meiotic chromosome condensation, pairing, and bouquet stage duration are modified in spermatocytes lacking axial elements. Mol Biol Cell 15:827–837. doi:10.1091/mbc.E03-07-0524
Macqueen AJ, Hochwagen A (2011) Checkpoint mechanisms: the puppet masters of meiotic prophase. Trends Cell Biol 21:393–400. doi:10.1016/j.tcb.2011.03.004
McGinty RK, Kim J, Chatterjee C, Roeder RG, Muir TW (2008) Chemically ubiquitylated histone H2B stimulates hDot1L-mediated intranucleosomal methylation. Nature 453:812–816. doi:10.1038/nature06906
Meneghini MD, Wu M, Madhani HD (2003) Conserved histone variant H2A.Z protects euchromatin from the ectopic spread of silent heterochromatin. Cell 112:725–736
Miao F, Natarajan R (2005) Mapping global histone methylation patterns in the coding regions of human genes. Mol Cell Biol 25:4650–4661. doi:10.1128/MCB.25.11.4650-4661.2005
Mohan M, Herz H, Takahashi Y, Lin C, Lai KC, Zhang Y, Washburn MP, Florens L et al (2010) Linking H3K79 trimethylation to Wnt signaling through a novel Dot1-containing complex (DotCom). Genes Dev 24:574–589. doi:10.1101/gad.1898410
Mueller JL, Mahadevaiah SK, Park PJ, Warburton PE, Page DC, Turner JMA (2008) The mouse X chromosome is enriched for multicopy testis genes showing postmeiotic expression. Nat Genet 40:794–799. doi:10.1038/ng.126
Namekawa SH, Park PJ, Zhang L-F, Shima JE, McCarrey JR, Griswold MD, Lee JT (2006) Postmeiotic sex chromatin in the male germline of mice. Curr Biol 16:660–667. doi:10.1016/j.cub.2006.01.066
Namekawa SH, VandeBerg JL, McCarrey JR, Lee JT (2007) Sex chromosome silencing in the marsupial male germ line. Proc Natl Acad Sci U S A 104:9730–9735. doi:10.1073/pnas.0700323104
Ng HH, Feng Q, Wang H, Erdjument-Bromage H, Tempst P, Zhang Y, Struhl K (2002) Lysine methylation within the globular domain of histone H3 by Dot1 is important for telomeric silencing and Sir protein association. Genes Dev 16:1518–1527. doi:10.1101/gad.1001502
Ng HH, Ciccone DN, Morshead KB, Oettinger MA, Struhl K (2003) Lysine-79 of histone H3 is hypomethylated at silenced loci in yeast and mammalian cells: a potential mechanism for position-effect variegation. Proc Natl Acad Sci U S A 100:1820–1825. doi:10.1073/pnas.0437846100
Nguyen AT, Zhang Y (2011) The diverse functions of Dot1 and H3K79 methylation. Genes Dev 25:1345–1358. doi:10.1101/gad.2057811
Nguyen AT, Xiao B, Neppl RL, Kallin EM, Li J, Chen T, Wang D-Z, Xiao X et al (2011) DOT1L regulates dystrophin expression and is critical for cardiac function. Genes Dev 25:263–274. doi:10.1101/gad.2018511
Okada Y, Feng Q, Lin Y, Jiang Q, Li Y, Coffield VM, Su L, Xu G et al (2005) hDOT1L links histone methylation to leukemogenesis. Cell 121:167–178. doi:10.1016/j.cell.2005.02.020
Ontoso D, Acosta I, van Leeuwen F, Freire R, San-Segundo PA (2013) Dot1-dependent histone H3K79 methylation promotes activation of the Mek1 meiotic checkpoint effector kinase by regulating the Hop1 adaptor. PLoS Genet 9:e1003262. doi:10.1371/journal.pgen.1003262
Ooga M, Inoue A, Kageyama S, Akiyama T, Nagata M, Aoki F (2008) Changes in H3K79 methylation during preimplantation development in mice. Biol Reprod 78:413–424. doi:10.1095/biolreprod.107.063453
Page J, de la Fuente R, Manterola M, Parra MT, Viera A, Berríos S, Fernández-Donoso R, Rufas JS (2012) Inactivation or non-reactivation: what accounts better for the silence of sex chromosomes during mammalian male meiosis? Chromosoma 121:307–326. doi:10.1007/s00412-012-0364-y
Parra MT, Viera A, Gómez R, Page J, Benavente R, Santos JL, Rufas JS, Suja JA (2004) Involvement of the cohesin Rad21 and SCP3 in monopolar attachment of sister kinetochores during mouse meiosis I. J Cell Sci 117:1221–1234. doi:10.1242/jcs.00947
Pawlowski WP, Cande WZ (2005) Coordinating the events of the meiotic prophase. Trends Cell Biol 15:674–681. doi:10.1016/j.tcb.2005.10.005
Pittman DL, Cobb J, Schimenti KJ, Wilson LA, Cooper DM, Brignull E, Handel MA, Schimenti JC (1998) Meiotic prophase arrest with failure of chromosome synapsis in mice deficient for Dmc1, a germline-specific RecA homolog. Mol Cell 1:697–705
Roig I, Dowdle JA, Toth A, de Rooij DG, Jasin M, Keeney S (2010) Mouse TRIP13/PCH2 is required for recombination and normal higher-order chromosome structure during meiosis. PLoS Genet 6:e1001062. doi:10.1371/journal.pgen.1001062
Romanienko PJ, Camerini-Otero RD (2000) The mouse Spo11 gene is required for meiotic chromosome synapsis. Mol Cell 6:975–987
San-Segundo PA, Roeder GS (1999) Pch2 links chromatin silencing to meiotic checkpoint control. Cell 97:313–324
San-Segundo PA, Roeder GS (2000) Role for the silencing protein Dot1 in meiotic checkpoint control. Mol Biol Cell 11:3601–3615
Smagulova F, Gregoretti IV, Brick K, Khil P, Camerini-Otero RD, Petukhova G V (2011) Genome-wide analysis reveals novel molecular features of mouse recombination hotspots. Nature 472:375–378. doi:10.1038/nature09869
Sommermeyer V, Béneut C, Chaplais E, Serrentino ME, Borde V (2013) Spp1, a member of the Set1 Complex, promotes meiotic DSB formation in promoters by tethering histone H3K4 methylation sites to chromosome axes. Mol Cell 49:43–54. doi:10.1016/j.molcel.2012.11.008
Steger DJ, Lefterova MI, Ying L, Stonestrom AJ, Schupp M, Zhuo D, Vakoc AL, Kim J-E et al (2008) DOT1L/KMT4 recruitment and H3K79 methylation are ubiquitously coupled with gene transcription in mammalian cells. Mol Cell Biol 28:2825–2839. doi:10.1128/MCB.02076-07
Sweet SMM, Li M, Thomas PM, Durbin KR, Kelleher NL (2010) Kinetics of re-establishing H3K79 methylation marks in global human chromatin. J Biol Chem 285:32778–32786. doi:10.1074/jbc.M110.145094
Tachiwana H, Kagawa W, Osakabe A, Kawaguchi K, Shiga T, Hayashi-Takanaka Y, Kimura H, Kurumizaka H (2010) Structural basis of instability of the nucleosome containing a testis-specific histone variant, human H3T. Proc Natl Acad Sci U S A 107:10454–10459. doi:10.1073/pnas.1003064107
Tachiwana H, Osakabe A, Shiga T, Miya Y, Kimura H, Kagawa W, Kurumizaka H (2011) Structures of human nucleosomes containing major histone H3 variants. Acta Crystallogr D Biol Crystallogr 67:578–583. doi:10.1107/S0907444911014818
Turner JMA (2007) Meiotic sex chromosome inactivation. Development 134:1823–1831. doi:10.1242/dev.000018
van der Heijden GW, Derijck AAHA, Pósfai E, Giele M, Pelczar P, Ramos L, Wansink DG, van der Vlag J et al (2007) Chromosome-wide nucleosome replacement and H3.3 incorporation during mammalian meiotic sex chromosome inactivation. Nat Genet 39:251–258. doi:10.1038/ng1949
van Leeuwen F, Gafken PR, Gottschling DE (2002) Dot1p modulates silencing in yeast by methylation of the nucleosome core. Cell 109:745–756
Wojtasz L, Daniel K, Roig I, Bolcun-Filas E, Xu H, Boonsanay V, Eckmann CR, Cooke HJ et al (2009) Mouse HORMAD1 and HORMAD2, two conserved meiotic chromosomal proteins, are depleted from synapsed chromosome axes with the help of TRIP13 AAA-ATPase. PLoS Genet 5:e1000702. doi:10.1371/journal.pgen.1000702
Wu R, Terry AV, Singh PB, Gilbert DM (2005) Differential subnuclear localization and replication timing of histone H3 lysine 9 methylation states. Mol Biol Cell 16:2872–2881. doi:10.1091/mbc.E04
Wysocki R, Javaheri A, Allard S, Sha F, Côté J, Kron SJ (2005) Role of Dot1-dependent histone H3 methylation in G1 and S phase DNA damage checkpoint functions of Rad9. Mol Cell Biol 25:8430–8443. doi:10.1128/MCB.25.19.8430-8443.2005
Yao Y, Chen P, Diao J, Cheng G, Deng L, Anglin JL, Prasad BVV, Song Y (2011) Selective inhibitors of histone methyltransferase DOT1L: design, synthesis, and crystallographic studies. J Am Chem Soc 133:16746–16749. doi:10.1021/ja206312b
Yoshida K, Kondoh G, Matsuda Y, Habu T, Nishimune Y, Morita T (1998) The mouse RecA-like gene Dmc1 is required for homologous chromosome synapsis during meiosis. Mol Cell 1:707–718
Zee BM, Levin RS, Dimaggio PA, Garcia BA (2010) Global turnover of histone post-translational modifications and variants in human cells. Epigenetics & Chromatin 3:22. doi:10.1186/1756-8935-3-22
Zhang W, Hayashizaki Y, Kone BC (2004) Structure and regulation of the mDot1 gene, a mouse histone H3 methyltransferase. Biochem J 377:641–651. doi:10.1042/BJ20030839
Zhang Q, Xue P, Li H, Bao Y, Wu L, Chang S, Niu B, Yang F et al (2013) Histone modification mapping in human brain reveals aberrant expression of histone H3 lysine 79 dimethylation in neural tube defects. Neurobiol Dis 54:404–413. doi:10.1016/j.nbd.2013.01.014
Zhou VW, Goren A, Bernstein BE (2011) Charting histone modifications and the functional organization of mammalian genomes. Nat Rev Genet 12:7–18. doi:10.1038/nrg2905
Acknowledgments
We thank the members of the SK and PSS laboratories for useful comments and discussion. We also thank the reviewers for their constructive comments and suggestions improving the manuscript. DO was supported by a predoctoral fellowship (JAE-Predoc) from the CSIC (Spain) and an EMBO short-term fellowship. Research was supported by NIH grant R01 GM105421 (to Maria Jasin and SK) and grants from MINECO (BFU2012-35748), Junta de Castilla y León (CSI025A11-2), and Fundación Ramón Areces to PSS.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 1205 kb)
Rights and permissions
About this article
Cite this article
Ontoso, D., Kauppi, L., Keeney, S. et al. Dynamics of DOT1L localization and H3K79 methylation during meiotic prophase I in mouse spermatocytes. Chromosoma 123, 147–164 (2014). https://doi.org/10.1007/s00412-013-0438-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00412-013-0438-5