Abstract
Vascular endothelial growth factor (VEGF) is closely related to angiogenesis. Anticancer therapy by inhibiting VEGF signaling is well established. However, the role of VEGF in cell–cell communication during the response to ionizing radiation is not well understood. Here, we examined the role of VEGF on radiosensitivity of cells. The addition of recombinant VEGF (rVEGF) on cultured rat C6 glioma cells showed a radioprotective effects on X-ray irradiation and reduced oxidative stress. These effects were also observed by endogenous VEGF in supernatant of C6 glioma cells. Reduction of oxidative stress by VEGF is suggested to underlie the radioprotective effects. The mechanism of VEGF-induced reduction of oxidative stress was indicated by a decreased oxygen consumption rate (OCR) in mitochondria. However, the number of DNA double-strand breaks (DSB) immediately after irradiation was not reduced by the treatment with VEGF. These results suggest that VEGF plays a role in cell survival after irradiation by controlling the oxidative condition through mitochondrial function that is independent of the efficiency of DSB induction.





Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Barcellos-Hoff MH (2005) Integrative radiation carcinogenesis: interactions between cell and tissue responses to DNA damage. Semin Cancer Biol 15:138–148
Barker HE, Paget JTE, Khan AA et al (2015) The tumour microenvironment after radiotherapy: Mechanisms of resistance and recurrence. Nat Rev Cancer 15:409–425
Bhargava P, Robinson MO (2011) Development of second-generation VEGFR tyrosine kinase inhibitors: current status. Drug Des Devel Ther 15:2653–2665
Chekhonin VP, Shein SA, Korchagina AA et al (2013) VEGF in tumor progression and targeted therapy. Curr Cancer Drug Targets 13:423–443
Chen L, Lin G, Chen K et al (2020) VEGF knockdown enhances radiosensitivity of nasopharyngeal carcinoma by inhibiting autophagy through the activation of mTOR pathway. Sci Rep 10(1):16328. https://doi.org/10.1038/s41598-020-73310-x
Coates PJ, Lorimore SA, Wright EG (2005) Cell and tissue responses to genotoxic stress. J Pathol 205:221–235
Goel S, Duda DG, Xu L et al (2011) Normalization of the vasculature for treatment of cancer and other diseases. Physiol Rev 91:1071–1121
Hasegawa T, Takahashi J, Nagasawa S et al (2020) DNA strand break properties of protoporphyrin IX by X-ray irradiation against melanoma. Int J Mol Sci 21:2302
Hillman GG, Lonardo F, Hoogstra DJ et al (2014) Axitinib improves radiotherapy in murine xenograft lung tumors. Translational Oncology 7:400–409
Kashino G, Kondoh T, Nariyama N et al (2009) Induction of DNA double-strand breaks and cellular migration through bystander effects in cells irradiated with the slit-type microplanar beam of the spring-8 synchrotron. Int J Radiat Oncol Biol Phys 74:229–236
Kashino G, Hayashi K, Douhara K et al (2014) Comparison of the biological effects of (18)F at different intracellular levels. Biochem Biophys Res Commun 454:7–11
Keating GM (2015) Axitinib: a review in advanced renal cell carcinoma. Drugs 75:1903–1913
Kobashigawa S, Kashino G, Suzuki K et al (2015) Ionizing radiation-induced cell death is partly caused by increase of mitochondrial reactive oxygen species in normal human fibroblast cells. Radiat Res 183:455–464
Kobayashi M, Morinibu A, Koyasu S et al (2017) A circadian clock gene, PER2, activates HIF-1 as an effector molecule for recruitment of HIF-1α to promoter regions of its downstream genes. FEBS J 284:3804–3816
Leung DW, Cachianes G, Kuang WJ et al (1989) Vascular endothelial growth factor is a secreted angiogenic mitogen. Science 246:1306–1309
Liao G, Khan M, Zhao Z et al (2021) Bevacizumab treatment of radiation-induced brain necrosis: a systematic review. Front Oncol 11:593449. https://doi.org/10.3389/fonc.2021.593449
Liu Y, Cox SR, Morita T et al (1995) Hypoxia regulates vascular endothelial growth factor gene expression in endothelial cells. Identification of a 5′ enhancer. Circ Res 77:638–643
Lorimore SA, Coates PJ, Wright EG (2003) Radiation-induced genomic instability and bystander effects: inter-related nontargeted effects of exposure to ionizing radiation. Oncogene 22:7058–7069
Mireștean CC, Iancu RI, Iancu DPT (2022) New horizons in modulating the radio-sensitivity of head and neck cancer – 100 years after Warburg’ effect discovery. Front Oncol 12:908695
Powell BE, Chin KJ, Finger PT (2022) Early anti-VEGF treatment for radiation maculopathy and optic neuropathy: lessons learned. Eye. https://doi.org/10.1038/s41433-022-02200-5
Rao SS, Thompson C, Cheng J (2014) Axitinib sensitization of high single dose radiotherapy. Radiother Oncol 111:88–93
Sappington DR, Siegel ER, Hiatt G et al (2016) Glutamine drives glutathione synthesis and contributes to radiation sensitivity of A549 and H460 lung cancer cell lines. Biochim Biophys Acta 1860:836–843
Smolina N, Bruton J, Kostareva A et al (2017) Assaying mitochondrial respiration as an indicator of cellular metabolism and fitness. Methods Mol Biol 1601:79–87
Tamari Y, Kashino G, Mori H (2017) Acquisition of radioresistance by IL-6 treatment is caused by suppression of oxidative stress derived from mitochondria after γ-irradiation. J Radiat Res 58:412–420
Viallard C, Larrive´e B, (2017) Tumor angiogenesis and vascular normalization: alternative therapeutic targets. Angiogenesis 20:409–426
Zhuang H, Shi S, Yuan Z et al (2019) Bevacizumab treatment for radiation brain necrosis: mechanism, efficacy and issues. Mol Cancer. https://doi.org/10.1186/s12943-019-0950-1
Acknowledgements
We thank Professor Hiroki Kuniyasu, Dr. Rina Fujiwara-Tani and Dr. Shiori Mori (Department of Molecular Pathology, Nara Medical University School of Medicine) for their support in using the extracellular flux analyzer. This work was supported by JSPS Grants-in-Aid for Scientific Research (KAKENHI) (Grant numbers JP18K07764, JP26461863 and JP21K07736).
Author information
Authors and Affiliations
Contributions
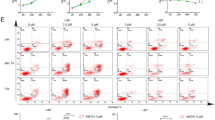
Kashino wrote the main manuscript text. Kashino and Uchikoshi prepared figure 1, Kashino and Kobashigawa prepared figure 2 and 4, Kashino and Tamari prepared figure 3, and Kashino prepared figure 5. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kashino, G., Kobashigawa, S., Uchikoshi, A. et al. VEGF affects mitochondrial ROS generation in glioma cells and acts as a radioresistance factor. Radiat Environ Biophys 62, 213–220 (2023). https://doi.org/10.1007/s00411-023-01021-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00411-023-01021-8