Abstract
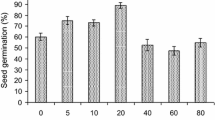
Quinoa is one of the crops well-adapted to high altitude regions that can grow relatively well under drought, humid, and high UV radiation conditions. This study was performed to investigate the effects of gamma-radiation on quinoa. Seeds were treated with various doses of 50 Gy, 100 Gy, 200 Gy, 300 Gy, 400 Gy, 600 Gy, 800 Gy, and 1000 Gy. We investigated germination, as well as plant height, chlorophyll content, and normalized difference vegetation index (NDVI) at 0, 30, 44, 58, and 88 days after transplanting (DAT) and panicle weight at 88 DAT. The plants grown from the seeds treated at radiation doses greater than 200 Gy showed reduced values in most growth and physiological characteristics. The germination rate and germination speed were higher in the 50 Gy-treated seeds than in 0 Gy-treated (control) seeds. Plant height and panicle weight were highest in the plants from 50 Gy-treated seeds. Chlorophyll content was higher in all treated samples than in the controls. NDVI value showed the highest value in 0 Gy controls and plants treated with 50 Gy. The antioxidant activity was also higher in the plants from the seeds treated with 50 Gy and 100 Gy, showing a steady increase as the radiation dose increased even at 200 Gy. The plants from seeds treated with 0 Gy showed higher expression of proteins related to photorespiration and tubulin chains. The plants from seeds treated with 50 Gy induced more stress-responsive proteins.






Similar content being viewed by others
References
Abdel HMS, Ali ZA (2006) Effect of gamma irradiation on wheat immature culture regenerated plant. Res J Appl Sci 2:310–316
Adolf VI, Jacobsen SE, Shabala S (2013) Salt tolerance mechanisms in quinoa (Chenopodium quinoa Willd.). Environ Exp Bot 92:43–54. https://doi.org/10.1016/j.envexpbot.2012.07.004
Agathokleous E, Kitao M, Calabrese EJ (2019) Hormesis: a compelling platform for sophisticated plant science. Trends Plant Sci 24:318–327. https://doi.org/10.1016/j.tplants.2019.01.004
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399. https://doi.org/10.1146/annurev.arplant.55.031903.141701
Asada K (1999) The water-water cycle in chloroplasts: scavenging of active oxygens and dissipation of excess photons. Annu Rev Plant Biol 50:601–639. https://doi.org/10.1146/annurev.arplant.50.1.601
Basyouni R, Dunn BL, Goad C (2015) Use of nondestructive sensors to assess nitrogen status in potted poinsettia (Euphorbia pulcherrima L. (Willd. ex Klotzsch)) production. Sci Hortic 192:47–53
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/j.scienta.2015.05.011
Buss W, Kammann C, Koyro HW (2012) Biochar reduces copper toxicity in Chenopodium quinoa Willd. in a sandy soil. J Environ Qual 41:1157–1165. https://doi.org/10.2134/jeq2011.0022
Caplin N, Willey N (2018) Ionizing radiation, higher plants, and radioprotection: from acute high doses to chronic low doses. Front Plant Sci 9:847. https://doi.org/10.3389/fpls.2018.00847
Dhakshanamoorthy D, Selvaraj R, Chidambaram ALA (2011) Induced mutagenesis in Jatropha curcas L. using gamma rays and detection of DNA polymorphism through RAPD marker. C R Biol 334:24–30. https://doi.org/10.1016/j.crvi.2010
Ellyfa K, Ahmed OH, Shaharudin S, Majid NMN (2007) Gamma radiosensitivity study on long bean (Vigna sesquipedalis). Am J Appl Sci 4:1090–1093. https://doi.org/10.3844/ajassp.2007.1090.1093
Foyer CH, Noctor G (2000) Tansley review No. 112: Oxygen processing in photosynthesis: regulation and signaling. New Phytol 146:359–388. https://doi.org/10.1046/j.1469-8137.2000.00667.x
Gamon JA, Field CB, Goulden ML, Griffin KL, Hartley AE, Joel G, Peñuelas J, Valentini R (1995) Relationships between NDVI, canopy structure, and photosynthesis in three Californian vegetation types. Ecol Appl 5:28–41. https://doi.org/10.2307/1942049
Gillespie KM, Chae JM, Ainsworth EA (2007) Rapid measurement of total antioxidant capacity in plants. Nat Protoc 2:867–870. https://doi.org/10.1038/nprot.2007.100
González JA, Rosa M, Parrado MF, Hilal M, Prado FE (2009) Morphological and physiological responses of two varieties of a highland species (Chenopodium quinoa Willd.) growing under near-ambient and strongly reduced solar UV-B in a lowland location. J Photochem Photobiol B 96:144–151. https://doi.org/10.1016/j.jphotobiol.2009.05.003
Gygi SP, Rochon Y, Franza BR, Aebersold R (1999) Correlation between protein and mRNA abundance in yeast. Nat Rev Mol Cell Biol 19:1720–1730. https://doi.org/10.1128/MCB.19.3.1720
Hampton JG, Tekrony DM (1995) Handbook of Vigor Test Methods, 3rd edn. ISTA, p 117
Hanafy RS, Akladious SA (2018) Physiological and molecular studies on the effect of gamma radiation in fenugreek (Trigonella foenum-graecum L.) plant. J Genet Eng Biotechnol 16:683–692. https://doi.org/10.1016/j.jgeb.2018.02.012
Hayashi G, Moro CF, Rohila JS, Shibato J, Kubo A, Imanaka T, Kimura S, Ozawa S, Fukutani S, Endo S, Ichikawa K (2015) 2D-DIGE-based proteome expression changes in leaves of rice seedlings exposed to low-level gamma radiation at Iitate village, Fukushima. Plant Signal Behav 10:e1103406. https://doi.org/10.1080/15592324.2015.1103406
Hegazi AZ, Hamideldin N (2010) The effect of gamma irradiation on enhancement of growth and seed yield of okra [Abelmoschus esculentus (L.) Moench] and associated molecular changes. J Hortic for 2:38–51
Hilal M, Parrado MF, Rosa M, Gallardo M, Orce L, Massa EM, Prado FE (2004) Epidermal lignin deposition in quinoa cotyledons in response to UV-B radiation. J Photochem Photobiol 79:205–210. https://doi.org/10.1562/0031-8655(2004)079%3c0205:eldiqc%3e2.0.co;2
Jaipo N, Kosiwikul M, Panpuang N, Prakrajang K (2019) Low dose gamma radiation effects on seed germination and seedling growth of cucumber and okra. J Phys Conf Ser 1380:012106. https://doi.org/10.1088/1742-6596/1380/1/012106
Jan S, Parween T, Siddiqi TO, Mahmooduzzafar (2012) Effect of gamma radiation on morphological, biochemical and physiological aspects of plants and plant products. Environ Rev 20:17–39. https://doi.org/10.1139/a11-021
Keresztes Á, Kovács E (1991) Ultrastructural effects of ionizing radiation on plant cells. Scann Microsc 5:287–296. https://doi.org/10.1007/BF03030408
Khan S, Goyal S (2009) Improvement of mungbean varieties through induced mutations. Afr J Plant Sci 3:174–180
Kim JS, Lee YG, Kim DH, Park HS, Back MH (2000) Influence of low dose gamma radiation on the growth of maize (Zea mays L.) varieties. Korean J Environ Agric 19:328–331
Kim JH, Back MH, Chung BY, Wi SG, Kim JS (2004) Alterations in the photosynthetic pigments and antioxidant machineries of red pepper (Capsicum annuum L.) seedlings from gamma irradiated seeds. J Plant Biol 47:314–321. https://doi.org/10.1007/BF03030546
Kim DS, Kim JB, Goh EJ, Kim WJ, Kim SH, Seo YW, Kang SY (2011) Antioxidant response of Arabidopsis plants to gamma irradiation: genome-wide expression profiling of the ROS scavenging and signal transduction pathways. J Plant Physiol 168:1960–1971. https://doi.org/10.1016/j.jplph.2011.05.008
Kon E, Ahmed OH, Saamin S, Majid NM (2007) Gamma radiosensitivity study on long bean (Vigna sesquipedalis). Am J Appl Sci 4:1090–1093. https://doi.org/10.3844/ajassp.2007.1090.1093
Kovács E, Keresztes A (2002) Effect of gamma and UV-B/C radiation on plant cells. Micron 33:199–210. https://doi.org/10.1016/S0968-4328(01)00012-9
Li L, Jiang Q, Niu F, Hu Z, Zhang H (2016) Research progress on salt tolerance mechanisms in quinoa. J Agr Sci Technol (beijing) 18:31–40
Mackerness SAH, Surplus SL, Blake P, John CF, Buchanan-Wollaston V, Jordan BR, Thomas B (1999) Ultraviolet-B-induced stress and changes in gene expression in Arabidopsis thaliana: role of signalling pathways controlled by jasmonic acid, ethylene and reactive oxygen species. Plant Cell Environ 22:1413–1423. https://doi.org/10.1046/j.1365-3040.1999.00499.x
Maity JP, Mishra D, Chakraborty A, Saha A, Santra SC, Chanda S (2005) Modulation of some quantitative and qualitative characteristics in rice (Oryza sativa L.) and mung (Phaseolus mungo L.) by ionizing radiation. Radiat Phys Chem 74:391–394. https://doi.org/10.1016/j.radphyschem.2004.08.005
Marcu D, Damian G, Cosma C, Cristea V (2013) Gamma radiation effects on seed germination, growth and pigment content, and ESR study of induced free radicals in maize (Zea mays). J Biol Phys 39:625–634. https://doi.org/10.1007/s10867-013-9322-z
Martínez EA, Veas E, Jorquera C, San Martín R, Jara P (2009) Re-introduction of Chenopodium quinoa Willd. into arid Chile: cultivation of two lowland races under extremely low irrigation. J Agron Crop Sci 195:1–10. https://doi.org/10.1111/j.1439-037X.2008.00332.x
McCord JM, Fridovich I (1969) Superoxide dismutase: an enzymic function for erythrocuprein (hemocuprein). J Biol Chem 244:6049–6055. https://doi.org/10.1016/S0021-9258(18)63504-5
Melki M, Marouani A (2010) Effects of gamma rays irradiation on seed germination and growth of hard wheat. Environ Chem Lett 8:307–310. https://doi.org/10.1007/s10311-009-0222-1
Miller MW, Miller WM (1987) Radiation hormesis in plants. Health Phys 52:607–616. https://doi.org/10.1097/00004032-198705000-00012
Mittler R, Vanderauwera S, Gollery M, Van Breusegem F (2004) The reactive oxygen gene network in plants. Trends Plant Sci 9:490–498. https://doi.org/10.1016/j.tplants.2004.08.009
Nonogaki H, Bassel G, Bewley JD (2010) Germination—still a mystery. Plant Sci 179:574–581. https://doi.org/10.1016/j.plantsci.2010.02.010
Pilon M, Ravet K, Tapken W (2011) The biogenesis and physiological function of chloroplast superoxide dismutases. Biochim Biophys Acta 1807:989–998. https://doi.org/10.1016/j.bbabio.2010.11.002
Richardson AD, Duigan SP, Berlyn GP (2002) An evaluation of noninvasive methods to estimate foliar chlorophyll content. New Phytol 153:185–194. https://doi.org/10.1046/j.0028-646X.2001.00289.x
Sah NK, Pramanik S, Raychaudhuri SS (1996) Peroxidase changes in barley induced by ionizing and thermal radiation. Int J Radiat Biol 69:107–111. https://doi.org/10.1080/095530096146237
Shevchenko A, Wilm M, Vorm O, Mann M (1996) Mass spectrometric sequencing of proteins from silver-stained polyacrylamide gels. Anal Chem 68:850–858. https://doi.org/10.1021/ac950914h
Sosedov NI, Vakar AB (1963) Effect of gamma-rays on the biochemical properties of wheat. Proc Intern Congr Biochem 13:27–40
Volkova PY, Clement G, Makarenko ES, Kazakova EA, Bitarishvili SV, Lychenkova MA (2020) Metabolic profiling of γ-irradiated barley plants identifies reallocation of nitrogen metabolism and metabolic stress response. Dose Response 18:1559325820914186. https://doi.org/10.1177/1559325820914186
Wang X, Mao T (2019) Understanding the functions and mechanisms of plant cytoskeleton in response to environmental signals. Curr Opin Plant Biol 52:86–96. https://doi.org/10.1016/j.pbi.2019.08.002
Wang W, Tai F, Chen S (2008) Optimizing protein extraction from plant tissues for enhanced proteomics analysis. J Sep Sci 31:2032–2039. https://doi.org/10.1002/jssc.200800087
Yang A, Akhtar SS, Amjad M, Iqbal S, Jacobsen SE (2016) Growth and physiological responses of quinoa to drought and temperature stress. J Agron Crop Sci 202:445–453. https://doi.org/10.1111/jac.12167
Zaka R, Chenal C, Misset MT (2004) Effects of low doses of short-term gamma irradiation on growth and development through two generations of Pisum sativum. Sci Total Environ 320:121–129. https://doi.org/10.1016/j.scitotenv.2003.08.010
Zhang DW, Yuan S, Xu F, Zhu F, Yuan M, Ye HX (2016) Light intensity affects chlorophyll synthesis during greening process by metabolite signal from mitochondrial alternative oxidase in Arabidopsis. Plant Cell Environ 39:12–25. https://doi.org/10.1111/pce.12438
Zhang Y, Fu Q, Huang T, Liu CG, Lin A (2022) Ionizing radiation-induced DNA damage response affect cell compressibility. Biochem Biophys Res Commun 603:116–122. https://doi.org/10.1016/j.bbrc.2022.03.032
Acknowledgements
This study is a part of the results of the research funded by the Korean Ministry of Environment (grant no. 2018002270002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Song, K.E., Park, C.Y., Hong, S.H. et al. Beneficial effects of gamma-irradiation of quinoa seeds on germination and growth. Radiat Environ Biophys 61, 465–477 (2022). https://doi.org/10.1007/s00411-022-00986-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00411-022-00986-2