Abstract
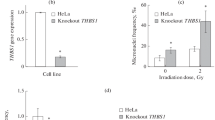
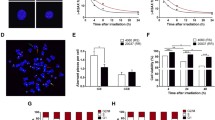
The immediate-early response gene 5 (IER5) was previously shown, using microarray analysis, to be upregulated by ionizing radiation. Here we further characterized the dose- and time-dependency of radiation-induced expression of IER5 at doses from 0.5 to 15 Gy by quantitative real-time PCR analyses in HeLa cells and human lymphoblastoid AHH-1 cells. A radiation-induced increase in the IER5 mRNA level was evident 2 h after irradiation with 2 Gy in both cell lines. In AHH-1 cells the expression reached a peak at 4 h and then quickly returned to the control level, while in HeLa cells the expression only remained increased for a short period of time at around 2 h after irradiation before returning to the control. After high-dose irradiation (10 Gy), the induction of the IER5 expression was lower and delayed in AHH-1 cells as compared with 2-Gy irradiated cells. In HeLa cells, at this dose, two peaks of increased expression were observed 2 h and 12–24 h post-irradiation, respectively. RNA interference technology was employed to silence the IER5 gene in HeLa cells. siRNA-mediated suppression of IER5 resulted in an increased proliferation of HeLa cells. Cell growth and survival analyses demonstrated that suppression of IER5 significantly increased the radioresistance of HeLa cells to radiation doses of up to 6 Gy, but barely affected the sensitivity of cells at 8 Gy. Moreover, suppression of IER5 potentiated radiation-induced arrest at the G2-M transition and led to an increase in the fraction of S phase cells. Taken together, we propose that the early radiation-induced expression of IER5 affects the radiosensitivity via disturbing radiation-induced cell cycle checkpoints.




Similar content being viewed by others
References
Ahmed MM, Venkatasubbarao K, Fruitwala SM, Muthukkumar S, Wood DP Jr, Sells SF, Mohiuddin M, Rangnekar VM (1996) EGR-1 induction is required for maximal radiosensitivity in A375–C6 melanoma cells. J Biol Chem 271:29231–29292
Amundson SA (2008) Functional genomics in radiation biology: a gateway to cellular systems-level studies. Radiat Environ Biophy 47:25–31
Amundson SA, Grace MB, McLeland CB, Epperly MW, Yeager A, Qimin Zhan, Greenberger JS, Fornace AJ Jr (2004) Human in vivo radiation-induced biomarkers: gene expression changes in radiotherapy patients. Cancer Res 64:6368–6371
Boerma M, van der Wees CGC, Vrieling H, Svensson JP, Wondergem J, van der Laarse A, Mullenders LHF, van Zeeland AA (2005) Microarray analysis of gene expression profiles of cardiac myocytes and fibroblasts after mechanical stress, ionising or ultraviolet radiation. BMC Genomics 6:6–15
Chaudhry MA, Chodosh LA, McKenna WG, Muschel RJ (2003) Gene expression profile of human cells irradiated in G1 and G2 phases of cell cycle. Cancer Lett 195:221–233
Cireli C, Tononi G (2000) Gene expression in the brain across the sleep-waking cycle. Brain Res 885:303–321
Ding LH, Shingyoji M, Chen F, Hwang JJ, Burma S, Lee C, Cheng JF, Chen DJ (2005) Gene expression profiles of normal human fibroblasts after exposure to ionizing radiation: a comparative study of low and high doses. Radiat Res 164:17–26
Franken NA, Ten Cate R, van Bree C, Haveman J (2004) Induction of the early response protein EGR-1 in human tumour cells after ionizing radiation is correlated with a reduction of repair of lethal lesions and an increase of repair of sublethal lesions. Int J Oncol 24:1027–1031
Guo WF, Lin RX, Huang J, Zhou Z, Yang J, Guo GZ, Wang SQ (2005) Identification of differentially expressed genes contributing to radioresistance in lung cancer cells using microarray analysis. Radiat Res 164:27–35
Hsu JY, Sun ZW, Li X, Reuben M, Tatchell K, Bishop DK, Grushcow JM, Brame CJ, Caldwell JA, Hunt DF, Lin F, Smith MM, Allis CD (2000) Mitotic phosphorylation of histone H3 is governed by Ipl1/aurora kinase and Glc7/PP1 phosphatase in budding yeast and nematodes. Cell 102:279–291
Ishikawa K, Ishii H, Saito T (2006) DNA damage-dependent cell cycle checkpoints and genomic stability. DNA and Cell Biol 25:406–411
Jeggo PA, Löbrich M (2006) Contribution of DNA repair and cell cycle checkpoint arrest to the maintenance of genomic stability. DNA repair (Amst) 5:1192–1198
Jen KY, Cheung VG (2003) Transcriptional response of lymphoblastoid cells to ionizing radiation. Genome Res 13:2092–2100
Kis E, Szatmári T, Keszei M, Farkas R, Esik O, Lumniczky K, Falus A, Sáfrány G (2006) Microarray analysis of radiation response genes in primary human fibroblasts. Int J Radiat Oncol Biol Phys 66:1506–1514
Koike M, Shiomi T, Koike A (2005) Identification of skin injury-related genes induced by ionizing radiation in human keratinocytes using cDNA microarray. J Radiat Res 46:173–184
Lanza V, Pretazzoli V, Olivieri G, Pascarella G, Panconesi A, Negri A (2005) Transcriptional responses of human umbilical vein endothelial cells to low dose radiation of ionizing radiation. J Radiat Res 46:265–276
Long XH, Zhao ZQ, He XP, Wang HP, Xu QZ, An J, Bai B, Sui JL, Zhou PK (2007) Dose-dependent expression changes of early response genes to ionizing radiation in human lymphoblastoid cells. Int J Mol Med 19:607–615
Mori M, Benotmanea MA, Tironea I, Hooghe-Peters EL, Desaintesa C (2005) Transcriptional response to ionizing radiation in lymphocyte subsets. CMLS Cell Mol Life Sci 62:1489–1501
Ojima E, Inoue Y, Miki C, Mori M, Kusunoki M (2007) Effectiveness of gene expression profiling for response prediction of rectal cancer to preoperative radiotherapy. J Gastroenterol 42:730–736
Okada A, Kushima K, Aoki Y, Bialer M, Fujiwara M (2005) Identification of early-responsive genes correlated to valproic acid-induced neural tube defects in mice. Birth Defects Res (Part A): Clinic Mol Teratol 73:229–238
Svensson JP, Stalpers LJA, Esveldt–van Lange REE, Franken NAP, Haveman J, Klein B, Turesson I, Vrieling H, Giphart-Gassler M (2006) Analysis of gene expression using gene sets discriminates cancer patients with and without late radiation toxicity. PloS Med 3:1904–1914
Wang HP, Long XH, Sun ZZ, Rigaud O, Xu QZ, Huang YC, Sui JL, Bai B, Zhou PK (2006) Identification of differentially transcribed genes in human lymphoblastoid cells irradiated with 0.5 Gy of γ-ray and the involvement of low dose radiation inducible CHD6 gene in cell proliferation and radiosensitivity. Int J Radiat Biol 82:181–190
Williams M, Lyu MS, Yang YL, Lin EP, Dunbrack R, Birren B, Cunningham J, Hunter K (1999) IER5, a novel member of the slow-kinetics immediate-early genes. Genomics 55:327–334
Winkles JA (1998) Serum- and polypeptide growth factor-inducible gene expression in mouse fibroblast. Prog Nucleic Acid Res Mol Biol 58:41–78
Acknowledgments
This work was supported by grants from the National Basic Research Program of MOST, China (973 Program, Grant No: 2007CB914603), the National Natural Science Foundation of China (Grant No: 30371232, No.:30770533), and the Beijing Municipal Education Commission (Science and Technology for Development Program: Km200710025007).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ding, KK., Shang, ZF., Hao, C. et al. Induced expression of the IER5 gene by γ-ray irradiation and its involvement in cell cycle checkpoint control and survival. Radiat Environ Biophys 48, 205–213 (2009). https://doi.org/10.1007/s00411-009-0213-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00411-009-0213-4