Abstract
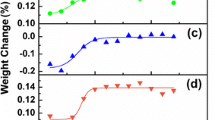
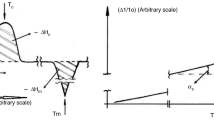
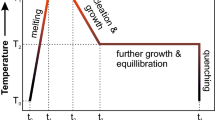
Graphite capsules are commonly used in high-temperature, high-pressure experiments, particularly for nominally anhydrous experiments and iron-bearing silicate samples. Due to the presence of graphite in the sample assembly, the oxygen fugacity for these experiments is thought to be relatively low, typically at or below the graphite-CO-CO2 buffer (CCO). The detailed mechanism and kinetics of redox control in graphite capsule experiments are, however, poorly understood. This is especially problematic for short duration experiments (e.g. kinetic experiments), because it is uncertain whether the experimental product will preserve its initial oxygen fugacity, or become reduced during the experiment. In this study, a set of basaltic glasses after high-temperature experiments in graphite capsules were analyzed by micro X-ray absorption near-edge structure (µ-XANES) to obtain their Fe3+/ΣFe profiles near the graphite–melt interface. The results show rapid reduction of ferric iron in the basaltic melt, reaching near-equilibrium in half an hour for samples of 2 mm diameter and 1.3–1.9 mm thickness. Even for a “time-zero” experiment, which was quenched immediately after reaching the target temperature, the reduction profile is over 100 µm in length. By comparing experiments at the same temperature and pressure but with different durations, the reduction reaction progress is found to be linear to the square root of duration, indicating that the reduction process is diffusion-controlled. Such a rapid reduction of the basaltic melt requires a mechanism that is significantly faster than divalent cation diffusion or oxygen diffusion, and is best explained by molecular hydrogen diffusion. It has been shown by previous studies that nominally anhydrous high-pressure experiments could contain significant amounts of water. Thousands of ppm of H2O could remain in the graphite capsule even after drying at 120 °C for an extended time period. At high temperatures, H2O reacts with graphite to produce molecular hydrogen, which then diffuses into the basaltic glass and causes reduction. This mechanism is also supported by a compensating H2O profile of equivalent length in the basaltic glass, showing evidence for H2O produced by molecular hydrogen reacting with ferric iron. A quantitative model is proposed and it successfully reproduces the Fe3+/ΣFe profiles in our experiments. The model helps explain the kinetics of the reduction process and demonstrates that for a basaltic glass with reasonable initial FeO* content, Fe3+/ΣFe ratios, and thicknesses, the equilibrium oxidation state can usually be reached in one hour at ~ 1300 °C and ~ 0.5 GPa. Although extrapolating our conclusion to the large range of graphite capsule experiments requires knowledge on how H2 solubility and diffusivity varies as a function of silicate composition, temperature, and pressure, the reduction process is expected to be rapid in general because H2 diffusivity is high in silicate melts. Our study elucidates the mechanism and rate of oxygen fugacity change in graphite capsule experiments. Based on thermodynamic calculations, the reaction between graphite capsule and H2O is expected to produce a C-O-H fluid with an intrinsic oxygen fugacity of CCO −0.9, which agrees well with the measured Fe3+/ΣFe ratios in the basaltic glasses and the estimated oxygen fugacity for graphite and Pt–graphite capsule experiments from a previous study. Future studies are needed to better constrain the kinetics of this process at different temperature, pressure, and in silicate melts of different compositions. The dynamic process of H2 diffusing and reducing Fe3+ to Fe2+ shown in our experiments also provides a potential way to determine the diffusivity and solubility of molecular hydrogen in silicate melts, which are crucial in understanding volatile behaviors on reducing planetary bodies, such as the Moon and Mercury.







source of H and O in the system (i.e. the bulk ratio of H:O equals 2:1 by mass balance)


Similar content being viewed by others
References
Botcharnikov RE, Koepke J, Holtz F et al (2005) The effect of water activity on the oxidation and structural state of Fe in a ferro-basaltic melt. Geochim Cosmochim Acta 69:5071–5085. https://doi.org/10.1016/j.gca.2005.04.023
Chen Y, Zhang Y (2008) Olivine dissolution in basaltic melt. Geochim Cosmochim Acta 72:4756–4777. https://doi.org/10.1016/j.gca.2008.07.014
Cochain B, Neuville DR, de Ligny D et al (2009) Kinetics of iron redox reaction in silicate melts: a high temperature Xanes study on an alkali basalt. J Phys Conf Ser 190:012182. https://doi.org/10.1088/1742-6596/190/1/012182
Cook GB, Cooper RF, Wu T (1990) Chemical diffusion and crystalline nucleation during oxidation of ferrous iron-bearing magnesium aluminosilicate glass. J Non-Cryst Solids 120:207–222. https://doi.org/10.1016/0022-3093(90)90205-Z
Cooper RF, Fanselow JB, Poker DB (1996) The mechanism of oxidation of a basaltic glass: chemical diffusion of network-modifying cations. Geochim Cosmochim Acta 60:3253–3265. https://doi.org/10.1016/0016-7037(96)00160-3
Cottrell E, Kelley KA (2011) The oxidation state of Fe in MORB glasses and the oxygen fugacity of the upper mantle. Earth Planet Sci Lett 305:270–282. https://doi.org/10.1016/j.epsl.2011.03.014
Cottrell E, Kelley KA (2013) Redox heterogeneity in mid-ocean ridge basalts as a function of mantle source. Science 340:1314–1317. https://doi.org/10.1126/science.1233299
Cottrell E, Kelley KA, Lanzirotti A, Fischer RA (2009) High-precision determination of iron oxidation state in silicate glasses using XANES. Chem Geol 268:167–179. https://doi.org/10.1016/j.chemgeo.2009.08.008
Cottrell E, Lanzirotti A, Mysen B et al (2018) A Mössbauer-based XANES calibration for hydrous basalt glasses reveals radiation-induced oxidation of Fe. Am Mineral 103:489–501. https://doi.org/10.2138/am-2018-6268
Fiege A, Ruprecht P, Simon AC et al (2017) Calibration of Fe XANES for high-precision determination of Fe oxidation state in glasses: comparison of new and existing results obtained at different synchrotron radiation sources. Am Mineral 102:369–380. https://doi.org/10.2138/am-2017-5822
Gaillard F, Scaillet B, Pichavant M (2002) Kinetics of iron oxidation-reduction in hydrous silicic melts. Am Mineral 87:829–837. https://doi.org/10.2138/am-2002-0704
Gaillard F, Pichavant M, Mackwell S et al (2003) Chemical transfer during redox exchanges between H2 and Fe-bearing silicate melts. Am Mineral 88:308–315. https://doi.org/10.2138/am-2003-2-308
Goldman DS, Gupta PK (1983) Diffusion-controlled redox kinetics in a glassmelt. J Am Ceram Soc 66:188–190. https://doi.org/10.1111/j.1151-2916.1983.tb10014.x
Guo C, Zhang Y (2018) Multicomponent diffusion in basaltic melts at 1350 °C. Geochim Cosmochim Acta 228:190–204. https://doi.org/10.1016/j.gca.2018.02.043
Hauri EH, Gaetani GA, Green TH (2006) Partitioning of water during melting of the Earth’s upper mantle at H2O-undersaturated conditions. Earth Planet Sci Lett 248:715–734. https://doi.org/10.1016/j.epsl.2006.06.014
Holloway JR, Pan V, Gudmundsson G (1992) High-pressure fluid-absent melting experiments in the presence of graphite : oxygen fugacity, ferric/ferrous ratio and dissolved CO2. Eur J Mineral 4:105–114
Hui H, Zhang Y, Xu Z, Behrens H (2008) Pressure dependence of the speciation of dissolved water in rhyolitic melts. Geochim Cosmochim Acta 72:3229–3240. https://doi.org/10.1016/j.gca.2008.03.025
Jakobsson S, Oskarsson N (1994) The system C-O in equilibrium with graphite at high pressure and temperature: an experimental study. Geochim Cosmochim Acta 58:9–17. https://doi.org/10.1016/0016-7037(94)90442-1
Kress VC, Carmichael ISE (1988) Stoichiometry of the iron oxidation reaction in silicate melts. Am Mineral 73:1267–1274
Laporte D, Toplis MJ, Seyler M, Devidal J-L (2004) A new experimental technique for extracting liquids from peridotite at very low degrees of melting: application to partial melting of depleted peridotite. Contrib Mineral Petrol 146:463–484. https://doi.org/10.1007/s00410-003-0509-3
Lesher CE, Hervig RL, Tinker D (1996) Self diffusion of network formers (silicon and oxygen) in naturally occurring basaltic liquid. Geochim Cosmochim Acta 60:405–413. https://doi.org/10.1016/0016-7037(95)00400-9
Magnien V, Neuville DR, Cormier L et al (2004) Kinetics of iron oxidation in silicate melts: a preliminary XANES study. Chem Geol 213:253–263. https://doi.org/10.1016/j.chemgeo.2004.08.047
Magnien V, Neuville DR, Cormier L et al (2006) Kinetics of iron redox reactions in silicate liquids: a high-temperature X-ray absorption and Raman spectroscopy study. J Nucl Mater 352:190–195. https://doi.org/10.1016/j.jnucmat.2006.02.053
Magnien V, Neuville DR, Cormier L et al (2008) Kinetics and mechanisms of iron redox reactions in silicate melts: the effects of temperature and alkali cations. Geochim Cosmochim Acta 72:2157–2168. https://doi.org/10.1016/j.gca.2008.02.007
Medard E, McCammon CA, Barr JA, Grove TL (2008) Oxygen fugacity, temperature reproducibility, and H2O contents of nominally anhydrous piston-cylinder experiments using graphite capsules. Am Mineral 93:1838–1844. https://doi.org/10.2138/am.2008.2842
O’Neill HSTC, Berry AJ, Mallmann G (2018) The oxidation state of iron in Mid-Ocean Ridge Basaltic (MORB) glasses: implications for their petrogenesis and oxygen fugacities. Earth Planet Sci Lett 504:152–162. https://doi.org/10.1016/j.epsl.2018.10.002
Pommier A, Gaillard F, Pichavant M (2010) Time-dependent changes of the electrical conductivity of basaltic melts with redox state. Geochim Cosmochim Acta 74:1653–1671. https://doi.org/10.1016/j.gca.2009.12.005
Ravel B, Newville M (2005) ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J Synchrotron Radiat 12:537–541. https://doi.org/10.1107/S0909049505012719
Robie RA, Hemingway BS (1995) Thermodynamic properties of minerals and related substances at 298.15 K and 1 bar (105 Pascals) pressure and at higher temperatures. US Government Printing Office
Schreiber HD, Kozak SJ, Merkel RC et al (1986) Redox equilibria and kinetics of iron in a borosilicate glass-forming melt. J Non-Cryst Solids 84:186–195. https://doi.org/10.1016/0022-3093(86)90777-5
Spickenbom K, Sierralta M, Nowak M (2010) Carbon dioxide and argon diffusion in silicate melts: insights into the CO2 speciation in magmas. Geochim Cosmochim Acta 74:6541–6564. https://doi.org/10.1016/j.gca.2010.08.022
Wendlandt RF (1991) Oxygen diffusion in basalt and andesite melts: experimental results and discussion of chemical versus tracer diffusion. Contrib Mineral Petrol 108:463–471. https://doi.org/10.1007/BF00303450
Wojdyr M (2010) Fityk: a general-purpose peak fitting program. J Appl Crystallogr 43:1126–1128. https://doi.org/10.1107/S0021889810030499
Yang Y, Zhang Y, Simon A, Ni P (2016) Cassiterite dissolution and Sn diffusion in silicate melts of variable water content. Chem Geol 441:162–176. https://doi.org/10.1016/j.chemgeo.2016.07.021
Yu Y, Zhang Y, Chen Y, Xu Z (2016) Kinetics of anorthite dissolution in basaltic melt. Geochim Cosmochim Acta 179:257–274. https://doi.org/10.1016/j.gca.2016.02.002
Zhang Y (1999) H2O in rhyolitic glasses and melts: measurement, speciation, solubility, and diffusion. Rev Geophys 37:493–516. https://doi.org/10.1029/1999RG900012
Zhang Y (2008) Geochemical kinetics. Princeton University Press, Princeton
Zhang Y, Ni H (2010) Diffusion of H, C, and O components in silicate melts. Rev Mineral Geochem 72:171–225. https://doi.org/10.2138/rmg.2010.72.5
Zhang Y, Ni H, Chen Y (2010) Diffusion data in silicate melts. Rev Mineral Geochem 72:311–408. https://doi.org/10.2138/rmg.2010.72.8
Acknowledgements
P. Ni acknowledges the Rackham Summer Fellowship from University of Michigan and Carnegie Postdoctoral Fellowship from Carnegie Institution for Science for support while working on this project. This work is partially supported by Turner Awards to P. Ni from Department of Earth and Environmental Sciences, University of Michigan. The authors would like to acknowledge F. Gaillard and two anonymous reviewers for their constructive reviews that significantly improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Gordon Moore.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ni, P., Zhang, Y., Fiege, A. et al. Rapid reduction of basaltic glasses in piston-cylinder experiments: a XANES study. Contrib Mineral Petrol 176, 17 (2021). https://doi.org/10.1007/s00410-021-01773-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00410-021-01773-w