Abstract
Purpose
Pirfenidone and nintedanib unequivocally inhibit FVC decline, but have been inconsistently linked to reduced mortality in phase III studies. On the contrary, real-world data show a survival benefit of antifibrotic drugs. However, it is unknown what this benefit is across different Gender, Age, and Physiology (GAP) stages.
Research Questions
Is there a difference in transplant-free (TPF) survival of IPF patients receiving antifibrotic drugs (IPFAF) compared with an untreated cohort (IPFnon−AF)? Is this different for patients with GAP stage I, II, or III.
Methods
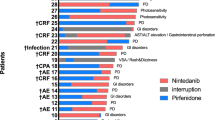
This is a single-center observational cohort study using prospectively included patients diagnosed with IPF between 2008–2018. Primary outcomes were TPF survival difference and 1-, 2-, and 3-year cumulative mortality for IPFAF and IPFnon−AF. This was repeated after stratification for GAP stage.
Results
In total, 457 patients were included. The median transplant-free survival was 3.4 years in IPFAF (n = 313) and 2.2 years in IPFnon−AF (n = 144, p = 0.005). For GAP stage II, a median survival of 3.1 and 1.7 years was noted for IPFAF (n = 143) and IPFnon−AF (n = 59, p < 0.001), respectively. A significantly lower 1-, 2-, and 3- year cumulative mortality was found for IPFAF with GAP stage II (1 yr: 7.0% vs 35.6%, 2 yr: 26.6% vs 55.9%, and 3 yr: 46.9% vs 69.5%). The 1-year cumulative mortality of IPFAF with GAP III was also significantly lower (19.0% vs 65.0%).
Conclusion
This large real-world study showed a survival benefit in IPFAF compared with IPFnon−AF. This especially holds true for patients with GAP stage II and III.


Similar content being viewed by others
Abbreviations
- AF:
-
Antifibrotic treatment
- DLCOc:
-
Diffusing capacity for carbon monoxide corrected for hemoglobin
- FPF:
-
Familial pulmonary fibrosis
- FVC:
-
Forced vital capacity
- GAP:
-
Gender, age, physiology model
- HRCT:
-
High-resolution computed tomography
- IPF:
-
Idiopathic pulmonary fibrosis
- IPFAF :
-
IPF patients treated with pirfenidone and/or nintedanib
- IPFnon − AF :
-
IPF patients not treated with antifibrotic drugs
- Ltx:
-
Lung transplantation
- MDD:
-
Multi-disciplinary discussion
- PANTHER-IPF:
-
Prednisone, azathioprine, and N-acetylcysteine: a study that evaluates response in IPF
- PF:
-
Pulmonary fibrosis
- PFT:
-
Pulmonary function test
- TPF:
-
Transplant-free
- UIP:
-
Usual interstitial pneumonia
References
Raghu G, Collard HR, Egan JJ et al (2011) An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med 183(6):788–824
Ley B, Collard HR, King TE (2011) Clinical course and prediction of survival in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 183(4):431–440
King TE, Bradford WZ, Castro-Bernardini S et al (2014) A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med 370(22):2083–2092
Noble PW, Albera C, Bradford WZ et al. Pirfenidone in patients with idiopathic pulmonary fibrosis (CAPACITY): Two randomised trials. Lancet. 2011;377(9779):1760–9. Available from: http://dx.doi.org/https://doi.org/10.1016/S0140-6736(11)60405-4
Richeldi L, Costabel U, Selman M et al (2011) Efficacy of a tyrosine kinase inhibitor in idiopathic pulmonary fibrosis. N Engl J Med. 365(12):1079–1087. https://doi.org/10.1056/NEJMoa1103690
Richeldi L, du Bois RM, Raghu G et al (2014) Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis. N Engl J Med 370(22):2071–2082
Taniguchi H, Ebina M, Kondoh Y et al (2010) Pirfenidone in idiopathic pulmonary fibrosis. Eur Respir J 35(4):821–829
Fang C, Huang H, Guo J et al (2020) Real-world experiences: Efficacy and tolerability of pirfenidone in clinical practice. PLoS ONE 15(1):1–15
Cameli P, Refini RM, Bergantini L et al (2020) Long-term follow-up of patients with idiopathic pulmonary fibrosis treated with pirfenidone or nintedanib: a real-life comparison study. Front Mol Biosci 7:1–8
Bando M, Yamauchi H, Ogura T et al (2016) Clinical experience of the long-term use of pirfenidone for idiopathic pulmonary fibrosis. Intern Med 55(5):443–448
Antoniou K, Markopoulou K, Tzouvelekis A et al (2020) Efficacy and safety of nintedanib in a Greek multicentre idiopathic pulmonary fibrosis registry: a retrospective, observational, cohort study. ERJ Open Res 6(1):00172–02019. https://doi.org/10.1183/23120541.00172-2019
Jo HE, Glaspole I, Grainge C et al (2017) Baseline characteristics of idiopathic pulmonary fibrosis: analysis from the australian idiopathic pulmonary fibrosis registry. Eur Respir J. https://doi.org/10.1183/13993003.01592-2016
Wright WA, Crowley LE, Parekh D et al (2021) Real-world retrospective observational study exploring the effectiveness and safety of antifibrotics in idiopathic pulmonary fibrosis. BMJ Open Respir Res 8(1):1–8
Behr J, Prasse A, Wirtz H et al (2020) Survival and course of lung function in the presence or absence of antifibrotic treatment in patients with idiopathic pulmonary fibrosis: Long-term results of the INSIGHTS-IPF registry. Eur Respir J. https://doi.org/10.1183/13993003.02279-2019
Ley B, Ryerson CJ, Vittinghoff E et al (2012) A multidimensional index and staging system for idiopathic pulmonary fibrosis. Ann Intern Med 156(10):684–695
Kim ES, Choi SM, Lee J et al (2015) Validation of the GAP SCORE in Korean patients with idiopathic pulmonary fibrosis. Chest 147(2):430–437
Hyldgaard C, Hilberg O, Muller A et al (2014) A cohort study of interstitial lung diseases in central Denmark. Respir Med 108(5):793–979. https://doi.org/10.1016/j.rmed.2013.09.002
Krauss E, Tello S, Wilhelm J et al (2020) Assessing the effectiveness of pirfenidone in idiopathic pulmonary fibrosis: long-term, real-world data from European IPF Registry (eurIPFreg). J Clin Med 9(11):3763
Gao J, Kalafatis D, Carlson L et al (2021) Baseline characteristics and survival of patients of idiopathic pulmonary fibrosis: a longitudinal analysis of the Swedish IPF Registry. Respir Res. https://doi.org/10.1186/s12931-021-01634-x
Guenther A, Krauss E, Tello S et al (2018) The European IPF registry (eurIPFreg): Baseline characteristics and survival of patients with idiopathic pulmonary fibrosis. Respir Res 19(1):1–10
Kaunisto J, Salomaa ER, Hodgson U et al (2019) Demographics and survival of patients with idiopathic pulmonary fibrosis in the FinnishIPF registry. ERJ Open Res. https://doi.org/10.1183/23120541.00170-2018
The Idiopathic Pulmonary Fibrosis Clinical Research Network*. Prednisone, Azathioprine, and N -Acetylcysteine for Pulmonary Fibrosis. N Engl J Med. 2012;366(21):1968–1977.
Raghu G, Rochwerg B, Zhang Y et al (2015) An official ATS/ERS/JRS/ALAT clinical practice guideline: Treatment of idiopathic pulmonary fibrosis: an update of the 2011 clinical practice guideline. Am J Respir Crit Care Med 192(2):e3-19
Salisbury ML, Tolle LB, Xia M et al (2017) Possible UIP pattern on high-resolution computed tomography is associated with better survival than definite UIP in IPF patients. Respir Med 131:229–235
Lederer DJ, Caplan-Shaw CE, O’Shea MK et al (2006) Racial and ethnic disparities in survival in lung transplant candidates with idiopathic pulmonary fibrosis. Am J Transplant 6(2):398–403
Ryerson CJ, Kolb M, Richeldi L et al (2019) Effects of nintedanib in patients with idiopathic pulmonary fibrosis by GAP stage. ERJ Open Res. https://doi.org/10.1183/23120541.00127-2018
Abe M, Tsushima K, Yoshioka K et al (2020) The gender–age–physiology system as a prognostic model in patients with idiopathic pulmonary fibrosis treated with nintedanib: A longitudinal cohort study. Adv Respir Med 88(5):369–376
Tomassetti S, Gurioli C, Ryu JH et al (2015) The impact of lung cancer on survival of idiopathic pulmonary fibrosis. Chest 147(1):157–164. https://doi.org/10.1378/chest.14-0359
Ozawa Y, Suda T, Naito T et al (2009) Cumulative incidence of and predictive factors for lung cancer in IPF. Respirology 14(5):723–728
Reck M, Kaiser R, Mellemgaard A et al (2014) Docetaxel plus nintedanib versus docetaxel plus placebo in patients with previously treated non-small-cell lung cancer (LUME-Lung 1): A phase 3, double-blind, randomised controlled trial. Lancet Oncol 15(2):143–155
Gottfried M, Bennouna J, Bondarenko I, Douillard JY, Heigener DF, Krzakowski M et al (2017) Efficacy and safety of nintedanib plus docetaxel in patients with advanced lung adenocarcinoma: complementary and exploratory analyses of the phase III LUME-Lung 1 study. Target Oncol 12(4):475–485
Miura Y, Saito T, Tanaka T et al (2018) Reduced incidence of lung cancer in patients with idiopathic pulmonary fibrosis treated with pirfenidone. Respir Investig 56(1):72–79. https://doi.org/10.1016/j.resinv.2017.09.007
Krämer M, Markart P, Drakopanagiotakis F et al (2020) Pirfenidone inhibits motility of NSCLC cells by interfering with the urokinase system. Cell Signal 65:109432. https://doi.org/10.1016/j.cellsig.2019.109432
Dhooria S, Agarwal R, Sehgal IS et al (2020) A real-world study of the dosing and tolerability of pirfenidone and its effect on survival in idiopathic pulmonary fibrosis. Sarcoidosis Vasc Diffus Lung Dis 37(2):148–157
Dobashi M, Tanaka H, Taima K et al (2021) The efficacy of nintedanib in 158 patients with idiopathic pulmonary fibrosis in real-world settings: A multicenter retrospective study. SAGE Open Med 9:205031212110233
Fletcher SV, Jones MG, Renzoni EA et al (2018) Safety and tolerability of nintedanib for the treatment of idiopathic pulmonary fibrosis in routine UK clinical practice. ERJ Open Res 4(4):00049–02018
Okuda R, Hagiwara E, Baba T et al (2013) Safety and efficacy of pirfenidone in idiopathic pulmonary fibrosis in clinical practice. Respir Med 107(9):1431–1437. https://doi.org/10.1016/j.rmed.2013.06.011
Vancheri C, Sebastiani A, Tomassetti S et al (2019) Pirfenidone in real life: A retrospective observational multicentre study in Italian patients with idiopathic pulmonary fibrosis. Respir Med 156:78–84. https://doi.org/10.1016/j.rmed.2019.08.006
Wijsenbeek MS, Grutters JC, Wuyts WA (2015) Early experience of pirfenidone in daily clinical practice in Belgium and the Netherlands: a retrospective cohort analysis. Adv Ther 32(7):691–704
Cottin V, Koschel D, Günther A et al (2018) Long-term safety of pirfenidone: results of the prospective, observational PASSPORT study. ERJ Open Res. https://doi.org/10.1183/23120541.00084-2018
Antoniou K, Markopoulou K, Tzouvelekis A et al (2020) Efficacy and safety of nintedanib in a Greek multicentre idiopathic pulmonary fibrosis registry: a retrospective, observational, cohort study. ERJ Open Res. https://doi.org/10.1183/23120541.00172-2019
Acknowledgements
The authors would like to thanks the included patients and research assistants K. Verhoeven, A. Verkleij-van Laarhoven, and A. Wind.
Funding
This study was funded by a ZonMw TopZorg grant (Grant Numbers: 842002001 and 842002003) and a TZO grant (Grant Number: 10070012010004).
Author information
Authors and Affiliations
Contributions
MGJPP: Collected the data, performed the statistical analysis, drafted the manuscript and rewrote the manuscript after peer review (guarantor of the paper). CHMVM: Played a major role in the conception of this study and peer reviewed the article. IW: Peer reviewed the article. ADMV: Peer reviewed the article. MV: Peer reviewed the article. JCG: Played a major role in the conception of this study and peer reviewed the article.
Corresponding author
Ethics declarations
Conflict of Interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Platenburg, M.G.J.P., van Moorsel, C.H.M., Wiertz, I.A. et al. Improved Survival of IPF patients Treated With Antifibrotic Drugs Compared With Untreated Patients. Lung 201, 335–343 (2023). https://doi.org/10.1007/s00408-023-00628-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-023-00628-4