Abstract
Introduction
Idiopathic pulmonary fibrosis (IPF) is a lethal disease with an unclear pathogenic mechanism. Components of the renin–angiotensin system (RAS) have a role in the pathogenesis of IPF, specifically, the aspartyl protease renin acts as a profibrotic factor in the lung. However, the concentration of the RAS components renin and soluble (pro)renin receptor (sPRR) have not been previously evaluated neither in serum nor in bronchoalveolar lavage fluid (BAL) of patients with IPF or chronic Hypersensitivity pneumonitis (cHP), a disease which may be confused with IPF.
Methods
The serum levels of renin [IPF patients (n = 70), cHP patients (n = 83), and controls (n = 26)] and sPRR [IPF (n = 28), cHP (37), and controls (n = 20)] were measured by ELISA. Renin was also quantified in BALs of IPF patients and controls by Western blot.
Results
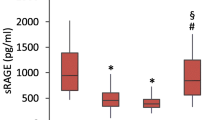
We found that the levels of renin were higher in serum samples from IPF patients when compared with cHP patients and controls. Furthermore, BALs from IPF patients had more renin than BALs from controls. Unlike renin, the serum levels of sPRR were lower in IPF and cHP patients than in control individuals.
Conclusions
The high levels of renin in sera and BALs of IPF patients suggest that renin might play a major role in the pathogenesis of IPF. Results from BAL confirm that renin is produced locally in the lung. Serum levels of renin could be used to differentiate IPF from cHP.



Similar content being viewed by others
Abbreviations
- IPF:
-
Idiopathic pulmonary fibrosis
- cHP:
-
Hypersensitivity pneumonitis
- BAL:
-
Bronchoalveolar lavage fluid
- EMT:
-
Epithelial to mesenchymal
- sPRR:
-
Soluble (pro)renin receptor
- PRR:
-
(Pro)renin receptor
- RAS:
-
Renin–angiotensin system
References
Ley B, Brown KK, Collard HR (2014) Molecular biomarkers in idiopathic pulmonary fibrosis. Am J Physiol Lung Cell Mol Physiol 307(9):L681–L691. https://doi.org/10.1152/ajplung.00014.2014
Ley B, Collard HR, King TE Jr (2011) Clinical course and prediction of survival in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 183(4):431–440. https://doi.org/10.1164/rccm.201006-0894CI
Martinez FJ, Collard HR, Pardo A, Raghu G, Richeldi L, Selman M, Swigris JJ, Taniguchi H, Wells AU (2017) Idiopathic pulmonary fibrosis. Nat Rev Dis Primers 3:17074. https://doi.org/10.1038/nrdp.2017.74
Eickelberg O, Kohler E, Reichenberger F, Bertschin S, Woodtli T, Erne P, Perruchoud AP, Roth M (1999) Extracellular matrix deposition by primary human lung fibroblasts in response to TGF-beta1 and TGF-beta3. Am J Physiol 276(5 Pt 1):L814–L824
Fine A, Goldstein RH (1987) The effect of transforming growth factor-beta on cell proliferation and collagen formation by lung fibroblasts. J Biol Chem 262(8):3897–3902
Willis BC, Borok Z (2007) TGF-beta-induced EMT: mechanisms and implications for fibrotic lung disease. Am J Physiol Lung Cell Mol Physiol 293(3):L525–L534. https://doi.org/10.1152/ajplung.00163.2007
Uhal BD, Kim JK, Li X, Molina-Molina M (2007) Angiotensin-TGF-beta 1 crosstalk in human idiopathic pulmonary fibrosis: autocrine mechanisms in myofibroblasts and macrophages. Curr Pharm Des 13(12):1247–1256
Huang Y, Wongamorntham S, Kasting J, McQuillan D, Owens RT, Yu L, Noble NA, Border W (2006) Renin increases mesangial cell transforming growth factor-beta1 and matrix proteins through receptor-mediated, angiotensin II-independent mechanisms. Kidney Int 69(1):105–113. https://doi.org/10.1038/sj.ki.5000011
Saris JJ, t Hoen PA, Garrelds IM, Dekkers DH, den Dunnen JT, Lamers JM, Jan Danser AH (2006) Prorenin induces intracellular signaling in cardiomyocytes independently of angiotensin II. Hypertension 48(4):564–571. https://doi.org/10.1161/01.HYP.0000240064.19301.1b
Montes E, Ruiz V, Checa M, Maldonado V, Melendez-Zajgla J, Montano M, Ordonez-Razo R, Cisneros J, Garcia-de-Alba C, Pardo A, Selman M (2012) Renin is an angiotensin-independent profibrotic mediator: role in pulmonary fibrosis. Eur Respir J 39(1):141–148. https://doi.org/10.1183/09031936.00130310
Burckle C, Bader M (2006) Prorenin and its ancient receptor. Hypertension 48(4):549–551. https://doi.org/10.1161/01.HYP.0000241132.48495.df
Cousin C, Bracquart D, Contrepas A, Corvol P, Muller L, Nguyen G (2009) Soluble form of the (pro)renin receptor generated by intracellular cleavage by furin is secreted in plasma. Hypertension 53(6):1077–1082. https://doi.org/10.1161/HYPERTENSIONAHA.108.127258
Sugulle M, Heidecke H, Maschke U, Herse F, Danser AHJ, Mueller DN, Staff AC, Dechend R (2017) Soluble (pro)renin receptor in preeclampsia and diabetic pregnancies. J Am Soc Hypertens 11(10):644–652. https://doi.org/10.1016/j.jash.2017.08.001
Bonakdaran S, Azami G, Tara F, Poorali L (2017) Soluble (pro)renin receptor is a predictor of gestational diabetes mellitus. Curr Diabetes Rev 13(6):555–559. https://doi.org/10.2174/1573399812666160919100253
Raghu G, Collard HR, Egan JJ, Martinez FJ, Behr J, Brown KK, Colby TV, Cordier JF, Flaherty KR, Lasky JA, Lynch DA, Ryu JH, Swigris JJ, Wells AU, Ancochea J, Bouros D, Carvalho C, Costabel U, Ebina M, Hansell DM, Johkoh T, Kim DS, King TE Jr, Kondoh Y, Myers J, Muller NL, Nicholson AG, Richeldi L, Selman M, Dudden RF, Griss BS, Protzko SL, Schunemann HJ, Fibrosis AEJACoIP (2011) An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med 183(6):788–824. https://doi.org/10.1164/rccm.2009-040GL
Mahmood T, Yang PC (2012) Western blot: technique, theory, and trouble shooting. N Am J Med Sci 4(9):429–434. https://doi.org/10.4103/1947-2714.100998
Buendia-Roldan I, Ruiz V, Sierra P, Montes E, Ramirez R, Vega A, Salgado A, Vargas MH, Mejia M, Pardo A, Selman M (2016) Increased expression of CC16 in patients with idiopathic pulmonary fibrosis. PLoS ONE 11(12):e0168552. https://doi.org/10.1371/journal.pone.0168552
Rosas IO, Richards TJ, Konishi K, Zhang Y, Gibson K, Lokshin AE, Lindell KO, Cisneros J, Macdonald SD, Pardo A, Sciurba F, Dauber J, Selman M, Gochuico BR, Kaminski N (2008) MMP1 and MMP7 as potential peripheral blood biomarkers in idiopathic pulmonary fibrosis. PLoS Med 5(4):e93. https://doi.org/10.1371/journal.pmed.0050093
Greene KE, King TE Jr, Kuroki Y, Bucher-Bartelson B, Hunninghake GW, Newman LS, Nagae H, Mason RJ (2002) Serum surfactant proteins-A and -D as biomarkers in idiopathic pulmonary fibrosis. Eur Respir J 19(3):439–446
Hamai K, Iwamoto H, Ishikawa N, Horimasu Y, Masuda T, Miyamoto S, Nakashima T, Ohshimo S, Fujitaka K, Hamada H, Hattori N, Kohno N (2016) Comparative study of circulating MMP-7, CCL18, KL-6, SP-A, and SP-D as disease markers of idiopathic pulmonary fibrosis. Dis Markers 2016:4759040. https://doi.org/10.1155/2016/4759040
Zhang Y, Bi L, Qiu Y, Wang Y, Ding J, Zhuang Y, Tian Y, Cai H (2016) Elevated sL1-CAM levels in BALF and serum of IPF patients. Respirology 21(1):143–148. https://doi.org/10.1111/resp.12659
Huang Y, Noble NA, Zhang J, Xu C, Border WA (2007) Renin-stimulated TGF-beta1 expression is regulated by a mitogen-activated protein kinase in mesangial cells. Kidney Int 72(1):45–52. https://doi.org/10.1038/sj.ki.5002243
Narumi K, Hirose T, Sato E, Mori T, Kisu K, Ishikawa M, Totsune K, Ishii T, Ichihara A, Nguyen G, Sato H, Ito S (2015) A functional (pro)renin receptor is expressed in human lymphocytes and monocytes. Am J Physiol Ren Physiol 308(5):F487–F499. https://doi.org/10.1152/ajprenal.00206.2014
Byrne AJ, Maher TM, Lloyd CM (2016) Pulmonary macrophages: a new therapeutic pathway in fibrosing lung disease? Trends Mol Med 22(4):303–316. https://doi.org/10.1016/j.molmed.2016.02.004
Papiris SA, Kollintza A, Kitsanta P, Kapotsis G, Karatza M, Milic-Emili J, Roussos C, Daniil Z (2005) Relationship of BAL and lung tissue CD4+ and CD8+ T lymphocytes, and their ratio in idiopathic pulmonary fibrosis. Chest 128(4):2971–2977. https://doi.org/10.1378/chest.128.4.2971
Esposito I, Perna F, Ponticiello A, Perrella M, Gilli M, Sanduzzi A (2005) Natural killer cells in Bal and peripheral blood of patients with idiopathic pulmonary fibrosis (IPF). Int J Immunopathol Pharmacol 18(3):541–545. https://doi.org/10.1177/039463200501800314
Melnyk RA, Tam J, Boie Y, Kennedy BP, Percival MD (2009) Renin and prorenin activate pathways implicated in organ damage in human mesangial cells independent of angiotensin II production. Am J Nephrol 30(3):232–243. https://doi.org/10.1159/000220260
Nguyen G, Blanchard A, Curis E, Bergerot D, Chambon Y, Hirose T, Caumont-Prim A, Tabard SB, Baron S, Frank M, Totsune K, Azizi M (2014) Plasma soluble (pro)renin receptor is independent of plasma renin, prorenin, and aldosterone concentrations but is affected by ethnicity. Hypertension 63(2):297–302. https://doi.org/10.1161/HYPERTENSIONAHA.113.02217
Lu X, Wang F, Xu C, Soodvilai S, Peng K, Su J, Zhao L, Yang KT, Feng Y, Zhou SF, Gustafsson JA, Yang T (2016) Soluble (pro)renin receptor via beta-catenin enhances urine concentration capability as a target of liver X receptor. Proc Natl Acad Sci USA 113(13):E1898–E1906. https://doi.org/10.1073/pnas.1602397113
Gonzalez AA, Lara LS, Luffman C, Seth DM, Prieto MC (2011) Soluble form of the (pro)renin receptor is augmented in the collecting duct and urine of chronic angiotensin II-dependent hypertensive rats. Hypertension 57(4):859–864. https://doi.org/10.1161/HYPERTENSIONAHA.110.167957
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None declared.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Eduardo, M., Ivette, BR., Gabriela, DP. et al. Evaluation of Renin and Soluble (Pro)renin Receptor in Patients with IPF. A Comparison with Hypersensitivity Pneumonitis. Lung 197, 715–720 (2019). https://doi.org/10.1007/s00408-019-00278-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-019-00278-5