Abstract
Purpose
The maintaining of asthma control is difficult due to high variability in response to therapy among patients. Since matrix metalloproteinase 9 (MMP9) is implicated in inflammation and remodeling of asthmatic airways, it could be associated with adequate response to asthma therapy. The aim of this study was to investigate whether variants in 3′ end of the MMP9 gene are associated with clinical phenotype and responsiveness to treatment in children with asthma.
Methods
The study included 127 asthmatic children from Slovenia. Variants in the 3′ end of the MMP9 gene were analyzed by direct DNA sequencing and the obtained results were correlated with clinical parameters.
Results
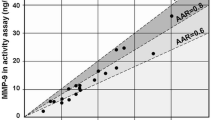
Two variants were detected, rs13925 and rs20544. For the variant rs20544, statistically significant difference in airway hyperresponsiveness (p = 0.011) and asthma control (p = 0.049) between genotypes was found. Patients with TT genotype had lower airway sensitivity, and after 12 months of treatment showed significant improvement in Asthma Control Test (ACT) scores compared to CC and CT genotype. For the variant rs13925, the association with lung function was observed. The carriers of A allele showed noticeable improvement of lung function after the first 6 months of treatment in comparison to the carriers of G allele (p = 0.046).
Conclusion
The main finding of our study is the association of MMP9 genotypes rs20544 TT and rs13925 AA and AG with better asthma control, and indirectly better response to treatment. Based on these results, MMP9 deserves further research as a potential predictive biomarker for asthma.
Similar content being viewed by others
References
Asher MI, Montefort S, Björkstén B, Lai CK, Strachan DP, Weiland SK, Williams H, ISAAC Phase Three Study Group (2006) Worldwide time trends in the prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and eczema in childhood: ISAAC Phases One and Three repeat multicountry cross-sectional surveys. Lancet 368(9537):733–743. https://doi.org/10.1016/S0140-6736(06)69283-0
(2018) Global Strategy for Asthma Management and Prevention. In: Global Initiative for Asthma
Beckett PA, Howarth PH (2003) Pharmacotherapy and airway remodelling in asthma? Thorax 58(2):163–174
Simpson JL, Scott RJ, Boyle MJ, Gibson PG (2005) Differential proteolytic enzyme activity in eosinophilic and neutrophilic asthma. Am J Respir Crit Care Med 172(5):559–565. https://doi.org/10.1164/rccm.200503-369OC
Mautino G, Oliver N, Chanez P, Bousquet J, Capony F (1997) Increased release of matrix metalloproteinase-9 in bronchoalveolar lavage fluid and by alveolar macrophages of asthmatics. Am J Respir Cell Mol Biol 17(5):583–591. https://doi.org/10.1165/ajrcmb.17.5.2562
Vignola AM, Riccobono L, Mirabella A, Profita M, Chanez P, Bellia V, Mautino G, D’accardi P, Bousquet J, Bonsignore G (1998) Sputum metalloproteinase-9/tissue inhibitor of metalloproteinase-1 ratio correlates with airflow obstruction in asthma and chronic bronchitis. Am J Respir Crit Care Med 158(6):1945–1950. https://doi.org/10.1164/ajrccm.158.6.9803014
St Jean PL, Zhang XC, Hart BK, Lamlum H, Webster MW, Steed DL, Henney AM, Ferrell RE (1995) Characterization of a dinucleotide repeat in the 92 kDa type IV collagenase gene (CLG4B), localization of CLG4B to chromosome 20 and the role of CLG4B in aortic aneurysmal disease. Ann Hum Genet 59(Pt 1):17–24
Yuan M, Zhan Q, Duan X, Song B, Zeng S, Chen X, Yang Q, Xia J (2013) A functional polymorphism at miR-491-5p binding site in the 3′-UTR of MMP-9 gene confers increased risk for atherosclerotic cerebral infarction in a Chinese population. Atherosclerosis 226(2):447–452. https://doi.org/10.1016/j.atherosclerosis.2012.11.026
Sun B, Qiu H, Zhao F, Qiao N, Fan W, Lu D, Chen H, Hu J, Fu C, Zhou L, Gu Y, Zhao Y, Mao Y (2012) The rs9509 polymorphism of MMP-9 is associated with risk of hemorrhage in brain arteriovenous malformations. J Clin Neurosci 19(9):1287–1290. https://doi.org/10.1016/j.jocn.2011.09.036
Bérubé D, Djandji M, Sampalis JS, Becker A (2014) Effectiveness of montelukast administered as monotherapy or in combination with inhaled corticosteroid in pediatric patients with uncontrolled asthma: a prospective cohort study. Allergy Asthma Clin Immunol 10(1):21. https://doi.org/10.1186/1710-1492-10-21
Kazani S, Wechsler ME, Israel E (2010) The role of pharmacogenomics in improving the management of asthma. J Allergy Clin Immunol 125(2):295–302. https://doi.org/10.1016/j.jaci.2009.12.014 (quiz 303–294)
Altraja S, Jaama J, Altraja A (2010) Proteome changes of human bronchial epithelial cells in response to pro-inflammatory mediator leukotriene E4 and pro-remodelling factor TGF-beta1. J Proteomics 73(6):1230–1240. https://doi.org/10.1016/j.jprot.2010.02.017
Ito K, Chung KF, Adcock IM (2006) Update on glucocorticoid action and resistance. J Allergy Clin Immunol 117(3):522–543. https://doi.org/10.1016/j.jaci.2006.01.032
Malmstrom K, Rodriguez-Gomez G, Guerra J, Villaran C, Piñeiro A, Wei LX, Seidenberg BC, Reiss TF (1999) Oral montelukast, inhaled beclomethasone, and placebo for chronic asthma. A randomized, controlled trial. Montelukast/Beclomethasone Study Group. Ann Intern Med 130(6):487–495
Israel E, Chervinsky PS, Friedman B, Van Bavel J, Skalky CS, Ghannam AF, Bird SR, Edelman JM (2002) Effects of montelukast and beclomethasone on airway function and asthma control. J Allergy Clin Immunol 110(6):847–854
Szefler SJ, Phillips BR, Martinez FD, Chinchilli VM, Lemanske RF, Strunk RC, Zeiger RS, Larsen G, Spahn JD, Bacharier LB, Bloomberg GR, Guilbert TW, Heldt G, Morgan WJ, Moss MH, Sorkness CA, Taussig LM (2005) Characterization of within-subject responses to fluticasone and montelukast in childhood asthma. J Allergy Clin Immunol 115(2):233–242. https://doi.org/10.1016/j.jaci.2004.11.014
Muraro A, Werfel T, Hoffmann-Sommergruber K, Roberts G, Beyer K, Bindslev-Jensen C, Cardona V, Dubois A, duToit G, Eigenmann P, Fernandez Rivas M, Halken S, Hickstein L, Høst A, Knol E, Lack G, Marchisotto MJ, Niggemann B, Nwaru BI, Papadopoulos NG, Poulsen LK, Santos AF, Skypala I, Schoepfer A, Van Ree R, Venter C, Worm M, Vlieg-Boerstra B, Panesar S, de Silva D, Soares-Weiser K, Sheikh A, Ballmer-Weber BK, Nilsson C, de Jong NW, Akdis CA, EAACI Food Allergy and Anaphylaxis Guidelines Group (2014) EAACI food allergy and anaphylaxis guidelines: diagnosis and management of food allergy. Allergy 69 (8): 1008–1025 https://doi.org/10.1111/all.12429
Duellman T, Warren C, Yang J (2014) Single nucleotide polymorphism-specific regulation of matrix metalloproteinase-9 by multiple miRNAs targeting the coding exon. Nucleic Acids Res 42(9):5518–5531. https://doi.org/10.1093/nar/gku197
Omair A, Mannion AF, Holden M, Leivseth G, Fairbank J, Hägg O, Fritzell P, Brox JI (2016) Age and pro-inflammatory gene polymorphisms influence adjacent segment disc degeneration more than fusion does in patients treated for chronic low back pain. Eur Spine J 25(1):2–13. https://doi.org/10.1007/s00586-015-4181-x
Grzela K, Zagorska W, Krejner A, Litwiniuk M, Zawadzka-Krajewska A, Banaszkiewicz A, Kulus M, Grzela T (2015) Prolonged treatment with inhaled corticosteroids does not normalize high activity of matrix metalloproteinase-9 in exhaled breath condensates of children with asthma. Arch Immunol Ther Exp (Warsz) 63(3):231–237. https://doi.org/10.1007/s00005-015-0328-z
Grzela K, Zagórska W, Krejner A, Banaszkiewicz A, Litwiniuk M, Kulus M, Grzela T (2016) Inhaled corticosteroids do not reduce initial high activity of matrix metalloproteinase (MMP)-9 in exhaled breath condensates of children with asthma exacerbation: a proof of concept study. Cent Eur J Immunol 41(2):221–227. https://doi.org/10.5114/ceji.2016.60998
Funding
The research was supported by the Grant P3-0360 from the Slovenian Research Agency and by the Grant 173008 of the Ministry of Education, Science and Technological Development of the Republic of Serbia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval and Informed Consent
The study was in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Written informed consent was obtained for all patients and the investigation was approved by the hospital ethical committee.
Rights and permissions
About this article
Cite this article
Dragicevic, S., Kosnik, M., Divac Rankov, A. et al. The Variants in the 3′ Untranslated Region of the Matrix Metalloproteinase 9 Gene as Modulators of Treatment Outcome in Children with Asthma. Lung 196, 297–303 (2018). https://doi.org/10.1007/s00408-018-0113-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-018-0113-y