Abstract
Background
Mannitol is a novel osmotic indirect bronchial challenge agent used to aid asthma diagnosis and management and is thought to reflect underlying inflammatory processes in asthma. Our objective was to evaluate relationships between mannitol airway hyperresponsiveness (AHR) and other measures of airway inflammation as well as direct-acting methacholine challenge in persistent asthmatics receiving inhaled corticosteroids.
Methods
We analysed screening data of mild to moderate persistent asthmatics, all receiving inhaled corticosteroids (ICS), who had mannitol and/or methacholine challenges, fractional exhaled nitric oxide (FeNO), and salivary eosinophilic cationic protein (ECP) performed as part of the same screen. Mannitol AHR was grouped by PD10 (cumulative provocative dose required to produce a 10 % fall in FEV1): mild (315–635 mg), moderate (75–315 mg), and severe (0–75 mg). FeNO groups were low (<25 ppb), medium (25–50 ppb), and high (> 50 ppb) and methacholine PC20 (provocative concentration of methacholine required to cause a 20 % fall in FEV1) groups were mild (2–8 mg/ml), moderate (0.5–2 mg/ml), and severe (0–0.5 mg/ml).
Results
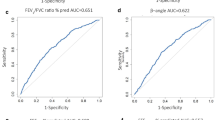
Mannitol PD10 groups were significantly different overall for FeNO (p = 0.023): 43 % higher in the severe vs. the mild group. There was a significant overall difference for methacholine PC20 (p = 0.006): a 2.1 doubling dilution difference between severe vs. mild mannitol groups. FeNO groups were significantly different overall for mannitol PD10 (p = 0.01) and methacholine PC20 (p = 0.029). Methacholine PC20 groups were significantly different overall for mannitol PD10 (p < 0.001) and FeNO (p = 0.005). No significant differences were found across any groups for salivary ECP, FEV1 % predicted, or ICS dose. Mannitol PD10, methacholine PC20, and FeNO as continuous variables all correlated with each other.
Conclusions
Mannitol challenge reflects underlying inflammation using FeNO and direct AHR using methacholine. Thus, mannitol may be a useful screening tool for the assessment of asthmatic patients receiving inhaled corticosteroids.



Similar content being viewed by others
References
Anderson SD, Brannan J, Spring J, Spalding N, Rodwell LT, Chan K, Gonda I, Walsh A, Clark AR (1997) A new method for bronchial-provocation testing in asthmatic subjects using a dry powder of mannitol. Am J Respir Crit Care Med 156:758–765
Sverrild A, Porsbjerg C, Thomsen SF, Backer V (2010) Airway hyperresponsiveness to mannitol and methacholine and exhaled nitric oxide: a random-sample population study. J Allergy Clin Immunol 126:952–958
Sverrild A, Porsbjerg C, Thomsen SF, Backer V (2009) Diagnostic properties of inhaled mannitol in the diagnosis of asthma: a population study. J Allergy Clin Immunol 124(928–32):e1
Anderson SD, Smith CM (1991) Osmotic challenges in the assessment of bronchial hyperresponsiveness. Am Rev Respir Dis 143:S43–S46
Crapo RO, Casaburi R, Coates AL, Enright PL, Hankinson JL, Irvin CG, MacIntyre NR, McKay RT, Wanger JS, Anderson SD, Cockcroft DW, Fish JE, Sterk PJ (2000) Guidelines for methacholine and exercise challenge testing-1999. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. Am J Respir Crit Care Med 161:309–329
Holzer K, Anderson SD, Chan HK, Douglass J (2003) Mannitol as a challenge test to identify exercise-induced bronchoconstriction in elite athletes. Am J Respir Crit Care Med 167:534–537
Cockcroft DW, Davis BE (2009) Diagnostic and therapeutic value of airway challenges in asthma. Curr Allergy Asthma Rep 9:247–253
Lipworth BJ, Short PM, Williamson PA, Clearie KL, Fardon TC, Jackson CM (2012) A randomized primary care trial of steroid titration against mannitol in persistent asthma: STAMINA trial. Chest 141:607–615
Green RH, Brightling CE, McKenna S, Hargadon B, Parker D, Bradding P, Wardlaw AJ, Pavord ID (2002) Asthma exacerbations and sputum eosinophil counts: a randomised controlled trial. Lancet 360:1715–1721
Currie GP, Haggart K, Lee DK, Fowler SJ, Wilson AM, Brannan JD, Anderson SD, Lipworth BJ (2003) Effects of mediator antagonism on mannitol and adenosine monophosphate challenges. Clin Exp Allergy 33:783–788
Currie GP, Haggart K, Brannan JD, Lee DK, Anderson SD, Lipworth BJ (2003) Relationship between airway hyperresponsiveness to mannitol and adenosine monophosphate. Allergy 58:762–766
Brannan JD, Koskela H, Anderson SD, Chew N (1998) Responsiveness to mannitol in asthmatic subjects with exercise- and hyperventilation-induced asthma. Am J Respir Crit Care Med 158:1120–1126
Leuppi JD, Salome CM, Jenkins CR, Koskela H, Brannan JD, Anderson SD, Andersson M, Chan HK, Woolcock AJ (2001) Markers of airway inflammation and airway hyperresponsiveness in patients with well-controlled asthma. Eur Respir J 18:444–450
Miedinger D, Mosimann N, Meier R, Karli C, Florek P, Frey F, Scherer K, Surber C, Villiger B, Michel F, Leuppi JD (2010) Asthma tests in the assessment of military conscripts. Clin Exp Allergy 40:224–231
Dupont LJ, Rochette F, Demedts MG, Verleden GM (1998) Exhaled nitric oxide correlates with airway hyperresponsiveness in steroid-naive patients with mild asthma. Am J Respir Crit Care Med 157:894–898
Porsbjerg C, Brannan JD, Anderson SD, Backer V (2008) Relationship between airway responsiveness to mannitol and to methacholine and markers of airway inflammation, peak flow variability and quality of life in asthma patients. Clin Exp Allergy 38:43–50
Dweik RA, Boggs PB, Erzurum SC, Irvin CG, Leigh MW, Lundberg JO, Olin AC, Plummer AL, Taylor DR (2011) An official ATS clinical practice guideline: interpretation of exhaled nitric oxide levels (FENO) for clinical applications. Am J Respir Crit Care Med 184:602–615
Short PM, Lipworth SI, Lipworth BJ (2011) Relationships between airway hyperresponsiveness, inflammation, and calibre in asthma. Lung 189:493–497
Miller MR, Hankinson J, Brusasco V, Burgos F, Casaburi R, Coates A, Crapo R, Enright P, van der Grinten CP, Gustafsson P, Jensen R, Johnson DC, MacIntyre N, McKay R, Navajas D, Pedersen OF, Pellegrino R, Viegi G, Wanger J (2005) Standardisation of spirometry. Eur Respir J 26:319–338
American Thoracic Society: European Respiratory Society (2005) ATS/ERS recommendations for standardized procedures for the online and offline measurement of exhaled lower respiratory nitric oxide and nasal nitric oxide, 2005. Am J Respir Crit Care Med 171:912–930
Koh GC, Shek LP, Kee J, Wee A, Ng V, Koh D (2010) Saliva and serum eosinophil cationic protein in asthmatic children and adolescents with and without allergic sensitization. J Asthma 47:61–65
Schmekel B, Ahlner J, Malmstrom M, Venge P (2001) Eosinophil cationic protein (ECP) in saliva: a new marker of disease activity in bronchial asthma. Respir Med 95:670–675
Bowton DL, Seeds MC, Bass DA (2003) Eosinophil cationic protein concentration in saliva does not correlate with eosinophil cationic protein concentration in sputum. Chest 123:372S
Fardon TC, Burns P, Barnes ML, Lipworth BJ (2006) A comparison of 2 extrafine hydrofluoroalkane-134a-beclomethasone formulations on methacholine hyperresponsiveness. Ann Allergy Asthma Immunol 96:422–430
Acknowledgments
We thank Pharmaxis which donated the mannitol challenge test kits as an educational gift to our department.
Conflict of interest
W. J. Anderson and B. J. Lipworth have no conflicts of interest or financial ties to disclose.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Anderson, W.J., Lipworth, B.J. Relationship of Mannitol Challenge to Methacholine Challenge and Inflammatory Markers in Persistent Asthmatics Receiving Inhaled Corticosteroids. Lung 190, 513–521 (2012). https://doi.org/10.1007/s00408-012-9396-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-012-9396-6