Abstract
Background
Cardiopulmonary exercise testing (CPET) is a safe and clinically useful method to assess functional capacity and to follow disease progression and the response to treatment in several clinical conditions.
Aim
We set out to determine the relationship between outcome measures of CPET and high-resolution computed tomography (HRCT) findings in thoracic sarcoidosis.
Methods
A cross-sectional study was carried out in which 42 nonsmoking outpatients (22 females; median age = 46.5 years) were evaluated. All the patients underwent pulmonary function tests (PFTs) and CPET. By using CPET, the most probable causes of exercise limitation were separated into respiratory mechanics (n = 25) and cardiovascular (n = 17). By using HRCT, the following patterns were recorded: predominant nodules (n = 18), predominant ground-glass opacity (n = 10), and predominant traction bronchiectasis and honeycombing (n = 14).
Results
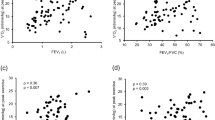
Although significant differences have been shown for both PFT parameters and CPET results, only the latter were able to distinguish between patients with ground-glass opacity and patients with traction bronchiectasis and honeycombing on HRCT. A statistically significant difference was found for peak VO2, breathing reserve, and P(A-a)O2 when patients with predominant traction bronchiectasis and honeycombing were compared to patients with other HRCT patterns (p < 0.0001). There was no statistical difference among the patterns with abnormal CPET and the patterns of abnormalities on HRCT (p > 0.05).
Conclusion
The functional capacity assessed by CPET was strongly influenced by HRCT patterns in sarcoidosis. Patients with traction bronchiectasis and honeycombing have lower exercise capacity measured by CPET.


Similar content being viewed by others
References
[No authors listed] (1999) Statement on sarcoidosis. Joint Statement of the American Thoracic Society (ATS), the European Respiratory Society (ERS) and the World Association of Sarcoidosis and Other Granulomatous Disorders (WASOG) adopted by the ATS Board of Directors and by the ERS Executive Committee, February 1999. Am J Respir Crit Care Med 160:736–755
Miller BH, Rosado-de-Christenson ML, McAdams HP, Fishback NF (1995) Thoracic sarcoidosis: radiologic-pathologic correlation. RadioGraphics 15:421–437
Criado E, Sánchez M, Ramírez J, Arguis P, Caralt TM, Perea RJ, Xaubet A (2010) Pulmonary sarcoidosis: typical and atypical manifestations at high-resolution CT with pathologic correlation. RadioGraphics 30:1567–1586
Remy-Jardin M, Giraud F, Remy J, Wattinne L, Wallaert B, Duhamel A (1994) Pulmonary sarcoidosis: role of CT in the evaluation of disease activity and functional impairment and in prognosis assessment. Radiology 191:675–680
Muers MF, Middleton WG, Gibson GJ, Prescott RJ, Mitchell DN, Connolly CK, Harrison BD (1997) A simple radiographic scoring method for monitoring pulmonary sarcoidosis: relations between radiographic scores, dyspnoea grade and respiratory function in the British Thoracic Society Study of Long-Term Corticosteroid Treatment. Sarcoidosis Vasc Diffuse Lung Dis 14:46–56
American Thoracic Society/American College of Chest Physicians (2003) ATS/ACCP statement on cardiopulmonary exercise testing. Am J Respir Crit Care Med 167:211–277
Weisman IM, Zeballos RJ (2001) Clinical exercise testing. Clin Chest Med 22:679–701
Barros WGP, Neder JA, Pereira CAC, Nery LE (2004) Clinical, radiographic and functional predictors of pulmonary gas exchange impairment at moderate exercise in patients with sarcoidosis. Respiration 71:367–373
Kollert F, Geck B, Suchy R, Jörres RA, Arzt M, Heidinger D, Hamer OW, Prasse A, Müller-Quernheim J, Pfeifer M, Budweiser S (2011) The impact of gas exchange measurement during exercise in pulmonary sarcoidosis. Respir Med 105:122–129
Scadding JG (1961) Prognosis of intrathoracic sarcoidosis in England. BMJ 2:1165–1172
Alhamad EH, Shaik SA, Idrees MM, Alanezi MO, Isnani AC (2010) Outcome measures of the 6 minute walk test: relationships with physiologic and computed tomography findings in patients with sarcoidosis. BMC Pulm Med 10:42
Terasaki H, Fujimoto K, Müller NL, Sadohara J, Uchida M, Koga T, Aizawa H, Hayabuchi N (2005) Pulmonary sarcoidosis: comparison of findings of inspiratory and expiratory high-resolution CT and pulmonary function tests between smokers and nonsmokers. AJR Am J Roentgenol 185:333–338
Miller MR, Hankinson J, Brusasco V, ATS/ERS Task Force (2005) Standardization of spirometry. Eur Respir J 26:319–338
Medinger AE, Khouri S, Rohatgi PK (2001) Sarcoidosis: the value of exercise testing. Chest 120:93–101
Pereira CAC, Sato T, Rodrigues SC (2007) New reference values for forced spirometry in white adults in Brazil. J Bras Pneumol 33:397–406
Neder JA, Andreoni S, Peres C, Nery LE (1999) Reference values for lung function tests. III. Carbon monoxide diffusing capacity (transfer factor). Braz J Med Biol Res 32:729–737
Sietsema KE, Kraft M, Ginzton L, Sharma OP (1992) Abnormal oxygen uptake responses to exercise in patients with mild pulmonary sarcoidosis. Chest 102:838–845
Gibbons WJ, Levy RD, Nava S, Malcolm I, Marin JM, Tardif C, Magder S, Lisbona S, Cosio MG (1991) Subclinical cardiac dysfunction in sarcoidosis. Chest 100:44–50
Wasserman K, Hansen JE, Sue DY, Stringer WW, Whipp BJ (2005) Principles of exercise testing and interpretation: including pathophysiology and clinical applications, 4th ed. Lippincott Willians and Wilkins, Philadelphia
Neder JA, Nery LE, Castelo A, Andreoni S, Lerario MC, Sachs A, Silva AC, Whipp BJ (1999) Prediction of metabolic and cardio-pulmonary responses to maximum cycle ergometry: a randomized study. Eur Respir J 4:1304–1313
Leung AN, Miller RR, Müller NL (1993) Parenchymal opacification in chronic infiltrative lung disease: CT-pathologic correlation. Radiology 188:209–214
Hansell DM, Milne DG, Wilsher ML, Wells AU (1998) Pulmonary sarcoidosis: morphologic associations of airflow obstruction at thin-section CT. Radiology 209:697–704
Lamberto C, Nunes H, Le Toumelin P, Duperron F, Valeyre D, Clerici C (2004) Membrane and capillary blood components of diffusion capacity of the lung for carbon monoxide in pulmonary sarcoidosis: relation to exercise gas exchange. Chest 125:2061–2068
Akira M, Kozuka T, Inoue Y, Sakatani M (2005) Long-term follow-up scan evaluation in patients with pulmonary sarcoidosis. Chest 127:185–191
Harrison BDW, Shaylor JM, Stokes TC, Wilkes AR (1991) Airflow limitation in sarcoidosis—a study of pulmonary function in 107 patients with newly diagnosed disease. Respir Med 85:59–64
Alhamad EH, Lynch JP III, Martinez FJ (2001) Pulmonary function tests in interstitial lung disease: what role do they have? Clin Chest Med 22:715–750
Arcasoy SM, Christie JD, Pochettino A, Rosengard BR, Blumenthal NP, Bavaria JE, Kotloff RM (2001) Characteristics and outcomes of patients with sarcoidosis listed for lung transplantation. Chest 120:873–880
Miller A, Brown LK, Sloane MF, Bhuptani A, Teirstein AS (1995) Cardiorespiratory responses to incremental exercise in sarcoidosis patients with normal spirometry. Chest 107:323–329
Silverman KJ, Hutchins GM, Bulkley BH (1978) Cardiac sarcoid: a clinicopathologic study of 84 unselected patients with systemic sarcoidosis. Circulation 58:1204–1211
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lopes, A.J., de Menezes, S.L.S., Dias, C.M. et al. Comparison Between Cardiopulmonary Exercise Testing Parameters and Computed Tomography Findings in Patients with Thoracic Sarcoidosis. Lung 189, 425–431 (2011). https://doi.org/10.1007/s00408-011-9316-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-011-9316-1