Abstract
Background
Head and neck squamous cell carcinoma (HNSC) poses a global health challenge. Effective biomarkers for early detection are necessary to improve the survival rate of HNSC patient. The purpose of this study was using integrated bioinformatic analysis to investigate the potential biological roles of GSDME in HNSC.
Methods
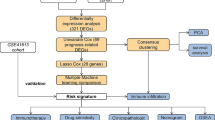
The Gene Expression Omnibus (GEO) and Cancer Gnome Atlas (TCGA) databases were used to analyze the expression of GSDME in different cancer types. The correlation between GSDME expression and immune cell infiltration or immune checkpoint genes was examined by Spearman correlation analysis. DNA methylation analysis of the GSDME gene was conducted using the MethSurv database. Kaplan–Meier (K–M) survival curves, diagnostic receiver operating characteristic (ROC) curves, nomogram model, and Cox regression analysis were chosen to evaluate the diagnostic and prognostic predictive value of GSDME. Connectivity Map (Cmap) online platform, Protein Data Bank (PDB) database and Chem3D, AutoDock Tool and PyMol software were used to predict and visualize potential molecular drugs aimed for GSDME.
Results
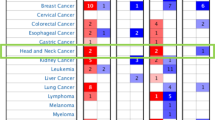
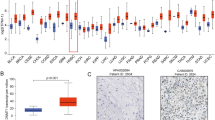
GSDME expression level in HNSC was significantly higher than in the controls (p < 0.001). Differentially expressed genes (DEGs) correlation with GSDME were enriched in the GO pathways, such as protein activation cascade, complement activation and classical pathway (p < 0.05). According to GSEA, GSDME-associated differentially expressed genes were significantly enriched in KRAS signaling pathway and cytokine signaling molecule (p < 0.05). There is a significant relation between GSDME expression and immune cell infiltration in HNSC tissues, as well as immune checkpoint genes expression (p < 0.001). DNA methylation status of cg17790129 CpG islands of GSDME gene is correlated with HNSC prognosis (p < 0.05). Based on Cox regression analysis of HNSC patients, GSDME as a potential risk gene has high correlation with overall survival (OS) and disease specific survival (DSS) (p < 0.05). In a ROC curve analysis, HNSC tissues were differentiated from adjacent peritumoral tissues based on GSDME expression levels (AUC = 0.928). Totally six potential drugs targeted for GSDME were screened and the molecular docking tests between GSDME protein and candidate drugs were conducted.
Conclusions
GSDME is a promising therapeutic target as well as a potential clinical biomarker in HNSC patients.










Similar content being viewed by others
Availability of data and materials
Our data can be found in the GEO and TCGA database.
References
Johnson DE, Burtness B, Leemans CR, Lui VWY, Bauman JE, Grandis JR (2020) Head and neck squamous cell carcinoma. Nat Rev Dis Primers 6:92
Li ZX, Zheng ZQ, Wei ZH, Zhang LL, Li F, Lin L, Liu RQ, Huang XD, Lv JW, Chen FP, He XJ, Guan JL, Kou J, Ma J, Zhou GQ, Sun Y (2019) Comprehensive characterization of the alternative splicing landscape in head and neck squamous cell carcinoma reveals novel events associated with tumorigenesis and the immune microenvironment. Theranostics 9:7648–7665
Siegel RL, Miller KD, Fuchs HE, Jemal A (2021) Cancer statistics. CA Cancer J Clin 71(2021):7–33
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F, Global Cancer Statistics 2020 (2021) GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249
van Camp G, Coucke P, Balemans W, van Velzen D, van de Bilt C, van Laer L, Smith RJ, Fukushima K, Padberg GW, Frants RR et al (1995) Localization of a gene for non-syndromic hearing loss (DFNA5) to chromosome 7p15. Hum Mol Genet 4:2159–2163
Orning P, Lien E, Fitzgerald KA (2019) Gasdermins and their role in immunity and inflammation. J Exp Med 216:2453–2465
Zhang CC, Li CG, Wang YF, Xu LH, He XH, Zeng QZ, Zeng CY, Mai FY, Hu B, Ouyang DY (2019) Chemotherapeutic paclitaxel and cisplatin differentially induce pyroptosis in A549 lung cancer cells via caspase-3/GSDME activation. Apoptosis 24:312–325
Wang CJ, Tang L, Shen DW, Wang C, Yuan QY, Gao W, Wang YK, Xu RH, Zhang H (2013) The expression and regulation of DFNA5 in human hepatocellular carcinoma DFNA5 in hepatocellular carcinoma. Mol Biol Rep 40:6525–6531
Lage H, Helmbach H, Grottke C, Dietel M, Schadendorf D (2001) DFNA5 (ICERE-1) contributes to acquired etoposide resistance in melanoma cells. FEBS Lett 494:54–59
Xia X, Wang X, Cheng Z, Qin W, Lei L, Jiang J, Hu J (2019) The role of pyroptosis in cancer: pro-cancer or pro-"host"? Cell Death Dis 10:650
Zhang Z, Zhang Y, Xia S, Kong Q, Li S, Liu X, Junqueira C, Meza-Sosa KF, Mok TMY, Ansara J, Sengupta S, Yao Y, Wu H, Lieberman J (2020) Gasdermin E suppresses tumour growth by activating anti-tumour immunity. Nature 579:415–420
Wang Y, Gao W, Shi X, Ding J, Liu W, He H, Wang K, Shao F (2017) Chemotherapy drugs induce pyroptosis through caspase-3 cleavage of a Gasdermin. Nature 547:99–103
Wang Q, Wang Y, Ding J, Wang C, Zhou X, Gao W, Huang H, Shao F, Liu Z (2020) A bioorthogonal system reveals antitumour immune function of pyroptosis. Nature 579:421–426
Lamb J (2007) The connectivity map: a new tool for biomedical research. Nat Rev Cancer 7:54–60
Qiang W, Dai Y, Xing X, Sun X (2021) Identification and validation of a prognostic signature and combination drug therapy for immunotherapy of head and neck squamous cell carcinoma. Comput Struct Biotechnol J 19:1263–1276
Kim S, Chen J, Cheng T, Gindulyte A, He J, He S, Li Q, Shoemaker BA, Thiessen PA, Yu B, Zaslavsky L, Zhang J, Bolton EE (2019) PubChem 2019 update: improved access to chemical data. Nucleic Acids Res 47(2019):D1102–D1109
Yu J, Li S, Qi J, Chen Z, Wu Y, Guo J, Wang K, Sun X, Zheng J (2019) Cleavage of GSDME by caspase-3 determines lobaplatin-induced pyroptosis in colon cancer cells. Cell Death Dis 10:193
Tan JQ, Li Z, Chen G, Wu M, Feng JL, Kong SY, Shi XQ, Zhang HM, Lao YZ, Zhou H, Zhang L, Xu HX (2022) The natural compound from Garcinia bracteata mainly induces GSDME-mediated pyroptosis in esophageal cancer cells. Phytomedicine 102:154142
Lu H, Zhang S, Wu J, Chen M, Cai MC, Fu Y, Li W, Wang J, Zhao X, Yu Z, Ma P, Zhuang G (2018) Molecular targeted therapies elicit concurrent apoptotic and GSDME-dependent pyroptotic tumor cell death. Clin Cancer Res 24:6066–6077
Zhang S, Wang B, Ma F, Tong F, Yan B, Liu T, Xie H, Song L, Yu S, Wei L (2021) Characteristics of B lymphocyte infiltration in HPV(+) head and neck squamous cell carcinoma. Cancer Sci 112:1402–1416
Zhao Y, Niu C, Cui J (2018) Gamma-delta (gamma delta) T cells: friend or foe in cancer development? J Transl Med. https://doi.org/10.1186/s12967-017-1378-2
Saura-Esteller J, de Jong M, King LA, Ensing E, Winograd B, de Gruijl TD, Parren P, van der Vliet HJ (2022) Gamma delta T-Cell based cancer immunotherapy: past-present-future. Front Immunol 13:915837
Xiao Y, Zhang T, Ma X, Yang QC, Yang LL, Yang SC, Liang M, Xu Z, Sun ZJ (2021) Microenvironment-responsive prodrug-induced pyroptosis boosts cancer immunotherapy. Adv Sci (Weinh) 8:e2101840
Hou J, Zhao R, Xia W, Chang CW, You Y, Hsu JM, Nie L, Chen Y, Wang YC, Liu C, Wang WJ, Wu Y, Ke B, Hsu JL, Huang K, Ye Z, Yang Y, Xia X, Li Y, Li CW, Shao B, Tainer JA, Hung MC (2020) PD-L1-mediated Gasdermin C expression switches apoptosis to pyroptosis in cancer cells and facilitates tumour necrosis. Nat Cell Biol 22:1264–1275
Kalbasi A, Ribas A (2020) Tumour-intrinsic resistance to immune checkpoint blockade. Nat Rev Immunol 20:25–39
De Schutter E, Croes L, Ibrahim J, Pauwels P, Op de Beeck K, Vandenabeele P, Van Camp G (2021) GSDME and its role in cancer: from behind the scenes to the front of the stage. Int J Cancer 148:2872–2883
Zhang Z, Zhang Y, Lieberman J (2021) Lighting a fire: can we harness pyroptosis to ignite antitumor immunity? Cancer Immunol Res 9:2–7
Tsai JY, Rédei D, Hohmann J, Wu CC (2020) 12-Deoxyphorbol esters induce growth arrest and apoptosis in human lung cancer A549 cells via activation of PKC-δ/PKD/ERK signaling pathway. Int J Mol Sci 21(20):7579
Plyasova AA, Berrino E, Khan TT, Veselovsky AV, Pokrovsky VS, Angeli A, Ferraroni M, Supuran CT, Pokrovskaya MV, Alexandrova SS, Gladilina YA, Sokolov NN, Hilal A, Carta F, Zhdanov DD (2021) Mechanisms of the antiproliferative and antitumor activity of novel telomerase-carbonic anhydrase dual-hybrid inhibitors. J Med Chem 64:11432–11444
Rogalska A, Gajek A, Marczak A (2019) Suppression of autophagy enhances preferential toxicity of epothilone A and epothilone B in ovarian cancer cells. Phytomedicine 61:152847
Bandyopadhyay G, Biswas T, Roy KC, Mandal S, Mandal C, Pal BC, Bhattacharya S, Rakshit S, Bhattacharya DK, Chaudhuri U, Konar A, Bandyopadhyay S (2004) Chlorogenic acid inhibits Bcr-Abl tyrosine kinase and triggers p38 mitogen-activated protein kinase-dependent apoptosis in chronic myelogenous leukemic cells. Blood 104:2514–2522
Liu J, Zhang Y, Sun S, Zhang G, Jiang K, Sun P, Zhang Y, Yao B, Sui R, Chen Y, Guo X, Tang T, Shi J, Liang H, Piao H (2019) Bufalin induces apoptosis and improves the sensitivity of human glioma stem-like cells to temozolamide. Oncol Res 27:475–486
Yu Z, Li Y, Li Y, Zhang J, Li M, Ji L, Tang Y, Zheng Y, Sheng J, Han Q, Li F, Guo J, Wang L, Sun X, Gao Y, Feng H (2022) Bufalin stimulates antitumor immune response by driving tumor-infiltrating macrophage toward M1 phenotype in hepatocellular carcinoma. J Immunother Cancer 10:e004297
Athuluri-Divakar SK, Vasquez-Del Carpio R, Dutta K, Baker SJ, Cosenza SC, Basu I, Gupta YK, Reddy MV, Ueno L, Hart JR, Vogt PK, Mulholland D, Guha C, Aggarwal AK, Reddy EP (2016) A small molecule RAS-mimetic disrupts RAS association with effector proteins to block signaling. Cell 165:643–655
Acknowledgements
We would like to acknowledge the TCGA, GEO network for providing data.
Funding
This article is supported by the Fundamental Research Funds for the Central Universities of Central South University. The funding number is 2022ZZTS0315.
Author information
Authors and Affiliations
Contributions
RH is responsible for the design of the study and collected the data. ML and LS are prepared and revised for the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, R., Lu, M. & She, L. Integrated analysis of diagnostic, prognostic value and potential drug treatment of GSDME in head and neck squamous cell carcinoma. Eur Arch Otorhinolaryngol 280, 4239–4253 (2023). https://doi.org/10.1007/s00405-023-08022-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-023-08022-9