Abstract
Purpose
Chemoradiotherapy with docetaxel (DOC), cisplatin (CDDP), and 5-FU (TPF-CRT) for locally advanced external auditory canal cancer (EACC) has favorable oncological and functional outcomes. To establish TPF-CRT as a standard of care for advanced EACC, we conducted this study to determine the maximum tolerated (MTD) and recommended dose (RD) of DOC in TPF-CRT for locally advanced EACC.
Methods
To determine the recommended (RD) and maximum tolerated dose (MTD) of DOC in TPF-CRT for EACC, a phase I trial was conducted using the standard “3 + 3” design for maximum dose finding. DOC was administered twice every 4 weeks, CDDP at 70 mg/m2 and 5-FU at 700 mg/m2; patients were also receiving radiotherapy (66 Gy). Eight patients with T3 or T4 EACC were prospectively enrolled.
Results
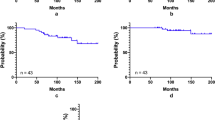
Two patients treated with DOC, 50 mg/m2, and one out of six patients treated with DOC, 40 mg/m2, had dose-limiting toxicities. Prolonged febrile neutropenia was observed in three patients. Grade 3 non-hematological toxicities were observed in only three patients. At study completion, six patients survived, five of whom were disease free.
Conclusion
The RD and MTD of DOC in TPF-CRT for locally advanced EACC are 40 mg/m2 when doses of CDDP and 5-FU are 70 mg/m2 and 700 mg/m2, respectively.

Similar content being viewed by others
Data availability
The data that support the findings of this study are openly available.
References
Morton RP, Stell PM, Derrick PP (1984) Epidemiology of cancer of the middle ear cleft. Cancer 53(7):1612–1617. https://doi.org/10.1002/1097-0142(19840401)53:7%3c1612::aid-cncr2820530733%3e3.0.co;2-p
Moody SA, Hirsch BE, Myers EN (2000) Squamous cell carcinoma of the external auditory canal: an evaluation of a staging system. Am J Otol 21(4):582–588
Bibas AG, Ward V, Gleeson MJ (2008) Squamous cell carcinoma of the temporal bone. Laryngol Otol 122(11):1156–1161. https://doi.org/10.1017/S0022215107001338
Moore MG, Deschler DG, McKenna MJ, Varvares MA, Lin DT (2007) Management outcomes following lateral temporal bone resection for ear and temporal bone malignancies. Otolaryngol Head Neck Surg 137(6):893–898. https://doi.org/10.1016/j.otohns.2007.09.010
Chi FL, Gu FM, Dai CF, Chen B, Li HW (2011) Survival outcomes in surgical treatment of 72 cases of squamous cell carcinoma of the temporal bone. Otology Neurotol 32(4):665–669. https://doi.org/10.1097/MAO.0b013e318210b90f
Morris LG, Mehra S, Shah JP, Bilsky MH, Selesnick SH, Kraus DH (2012) Predictors of survival and recurrence after temporal bone resection for cancer. Head Neck 34(9):1231–1239. https://doi.org/10.1002/hed.21883
Essig GF, Kitipornchai L, Adams F, Zarate D, Gandhi M, Porceddu S, Panizza B (2013) Lateral temporal bone resection in advanced cutaneous squamous cell carcinoma: report of 35 patients. Skull Base 74(1):54–59. https://doi.org/10.1055/s-0032-1331021
Shiga K, Nibu KI, Fujimoto Y, Asakage T, Homma A et al (2021) Multi-institutional survey of squamous cell carcinoma of the external auditory canal in Japan. Laryngoscope 131(3):E870–E874. https://doi.org/10.1002/lary.28936
Tsukuda M, Ishitoya J, Matsuda H, Horiuchi C, Taguchi T et al (2010) Randomized controlled phase II comparison study of concurrent chemoradiotherapy with docetaxel, cisplatin, and 5-fluorouracil versus CCRT with cisplatin, 5-fluorouracil, methotrexate and leucovorin in patients with locally advanced squamous cell carcinoma of the head and neck. Cancer Chemother Pharmacol 66(4):729–736. https://doi.org/10.1007/s00280-009-1217-0
Shiga K, Ogawa T, Maki A, Amano M, Kobayashi T (2011) Concomitant chemoradiotherapy as a standard treatment for squamous cell carcinoma of the temporal bone. Skull Base 21(3):153–158. https://doi.org/10.1055/s-0031-1275244
Shinomiya H, Hasegawa S, Yamashita D, Ejima Y, Kenji Y et al (2016) Concomitant chemoradiotherapy for advanced squamous cell carcinoma of the temporal bone. Head Neck 38:E949–E953. https://doi.org/10.1002/hed.24133 (Suppl 1)
Shiga K, Katagiri K, Saitoh D, Ogawa T, Higashi K, Ariga H (2018) Long-term outcomes of patients with squamous cell carcinoma of the temporal bone after concomitant chemoradiotherapy. Skull Base 79:S316–S321. https://doi.org/10.1055/s-0038-1651522 (Suppl 4)
Katori H, Tsukuda M (2005) Comparison of induction chemotherapy with docetaxel, cisplatin, and 5-fluorouracil (TPF) followed by radiation vs concurrent chemoradiotherapy with TPF in patients with locally advanced squamous cell carcinoma of the head and neck. Clin Oncol 17(3):148–152. https://doi.org/10.1016/j.clon.2004.09.013
Fuwa N, Shikama N, Hayashi N, Matsuzuka T, Toita T et al (2007) Treatment results of alternating chemoradiotherapy for nasopharyngeal cancer using cisplatin and 5-fluorouracil–a phase II study. Oral Oncol 43(9):948–955. https://doi.org/10.1016/j.oraloncology.2006.11.003
Funding
This study was conducted without grant supports.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have no conflicts of interest to report.
Ethical approval
The trial was approved by the Institutional Review Board of the University of Kobe (#1385).
Consent to participate and for publication
All participants gave a written informed consent to participate and for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shinomiya, H., Uehara, N., Fujita, T. et al. Phase I trial of concurrent chemoradiotherapy with docetaxel, cisplatin and 5-fluorouracil (TPF-CRT) for locally advanced squamous cell carcinoma of the external auditory canal. Eur Arch Otorhinolaryngol 279, 2805–2810 (2022). https://doi.org/10.1007/s00405-021-06974-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-021-06974-4



