Abstract
Background
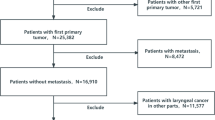
Prognostic risk factors of patients with initially diagnosed T2N0M0 glottic cancer remain unclear. This study was aimed to conduct a comprehensive analysis to identify valuable prognostic risk factors for initially diagnosed T2N0M0 glottic cancer.
Methods
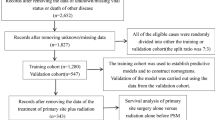
Data of patients with initially diagnosed T2N0M0 glottic cancer were extracted from the Surveillance, Epidemiology, and End Results database. Survival analyses and Cox regression analyses were conducted to evaluate overall survival (OS) and cancer-specific survival (CSS). In consideration of competing events, the competing risk (CR) analysis was applied. Furthermore, propensity-score matching (PSM) was applied to mimic randomized-controlled trials and reduce selection bias.
Results
A total of 923 eligible patients met the inclusion criteria. Survival analyses showed that age, marital status, primary site surgery, and radiation were independent predictors of OS. Besides, age, marital status, primary site surgery, radiation, and chemotherapy were independent predictors of CSS. Cox regression analyses and the CR analysis were basically consistent with this result. In addition, an internal validation and PSM were performed to explore the role of chemotherapy.
Conclusion
We conducted a comprehensive analysis to prove that age, marital status, primary site surgery, radiation, and chemotherapy may be valuable prognostic risk factors for initially diagnosed T2N0M0 glottic cancer. Primary site surgery and radiation should be recommended, whereas chemotherapy was likely not suitable so far. Furthermore, we constructed a CR nomogram to predict survival rates.



Similar content being viewed by others
Abbreviations
- OS:
-
Overall survival
- CSS:
-
Cancer-specific survival
- SEER:
-
Surveillance, epidemiology, and end results
- KM:
-
Kaplan–Meier
- CR:
-
Competing risk
- PSM:
-
Propensity-score matching
- AJCC:
-
American Joint Committee on Cancer
- HR:
-
Hazard ratio
- CI:
-
Confidence interval
- CIF:
-
Cumulative incidence function
- SHR:
-
Sub-distribution hazard ratio
References
Pfister DG, Spencer S, Adelstein D et al (2020) NCCN clinical practice guidelines in oncology: head and neck cancers (Version 1, 2020). Available at https://www.nccn.org/professionals/physician_gls/pdf/head-and-neck.pdf. Accessed 12 Feb 2020
Bhateja P, Ward MC, Hunter GH et al (2016) Impaired vocal cord mobility in T2N0 glottic carcinoma: suboptimal local control with radiation alone. Head Neck 38:1832–1836
Warner L, Lee K, Homer JJ (2017) Transoral laser microsurgery versus radiotherapy for T2 glottic squamous cell carcinoma: a systematic review of local control outcomes. Clin Otolaryngol 42:629–636
Hendriksma M, Heijnen BJ, Sjogren EV (2018) Oncologic and functional outcomes of patients treated with transoral CO2 laser microsurgery or radiotherapy for T2 glottic carcinoma: a systematic review of the literature. Curr Opin Otolaryngol Head Neck Surg 26:84–93
Yesensky J, Agrawal N, Bayan S et al (2017) AHNS series—Do you know your guidelines? Principles of treatment for glottic cancer: a review of the National Comprehensive Cancer Network guidelines. Head Neck 39:1729–1732
Mantsopoulos K, Psychogios G, Koch M, Zenk J, Waldfahrer F, Iro H (2012) Comparison of different surgical approaches in T2 glottic cancer. Head Neck 34:73–77
Al FK, Youssef BY, Mohamed A et al (2020) Outcomes after radiation therapy for T2N0/stage II glottic squamous cell carcinoma. Head Neck. https://doi.org/10.1002/hed.26308 (Epub ahead of print)
Murakami R, Nishimura R, Baba Y, Yumoto E, Oya N, Yamashita Y (2006) Concurrent chemoradiation therapy with low-dose CDDP and UFT for glottic carcinomas: evaluation using the sixth edition of the UICC TNM staging system. Acta Oncol 45:162–167
Katz SJ (2015) Cancer Care Delivery Research and the National Cancer Institute SEER Program: challenges and opportunities. JAMA Oncol 1:677–678
Lim H, Devesa SS, Sosa JA, Check D, Kitahara CM (2017) Trends in thyroid cancer incidence and mortality in the United States, 1974–2013. JAMA 317:1338–1348
Morgan CJ (2018) Reducing bias using propensity score matching. J Nucl Cardiol 25:404–406
Renehan AG, Malcomson L, Emsley R et al (2016) Watch-and-wait approach versus surgical resection after chemoradiotherapy for patients with rectal cancer (the OnCoRe project): a propensity-score matched cohort analysis. Lancet Oncol 17:174–183
Jones EA, Shuman AG, Egleston BL, Liu JC (2019) common pitfalls of head and neck research using cancer registries. Otolaryngol Head Neck Surg 161:245–250
Thomas L, Li F, Pencina M (2020) Using propensity score methods to create target populations in observational clinical research. JAMA. https://doi.org/10.1001/jama.2019.21558 (Epub ahead of print)
Thomas LE, Li F, Pencina MJ (2020) Overlap weighting: a propensity score method that mimics attributes of a randomized clinical trial. JAMA. https://doi.org/10.1001/jama.2020.7819 (Epub ahead of print)
Canis M, Martin A, Ihler F et al (2014) Transoral laser microsurgery in treatment of pT2 and pT3 glottic laryngeal squamous cell carcinoma—results of 391 patients. Head Neck 36:859–866
Canis M, Ihler F, Martin A, Matthias C, Steiner W (2015) Transoral laser microsurgery for T1a glottic cancer: review of 404 cases. Head Neck 37:889–895
Zhang Y, Wang B, Sun G, Zhang G, Lu L, Liang G (2018) Carbon dioxide laser microsurgery versus low-temperature plasma radiofrequency ablation for T1a glottic cancer: a single-blind randomized clinical trial. Biomed Res Int 2018:4295960
Chung SY, Lee CG (2020) Feasibility of single vocal cord irradiation as a treatment strategy for T1a glottic cancer. Head Neck 42:854–859
Nomiya T, Nemoto K, Wada H, Takai Y, Yamada S (2008) Long-term results of radiotherapy for T1a and T1bN0M0 glottic carcinoma. Laryngoscope 118:1417–1421
De Seta D, Campo F, D’Aguanno V et al (2020) Transoral laser microsurgery for Tis, T1, and T2 glottic carcinoma: 5-year follow-up. Lasers Med Sci. https://doi.org/10.1007/s10103-020-03049-4 (Epub ahead of print)
Gorphe P, Blanchard P, Breuskin I, Temam S, Tao Y, Janot F (2015) Vocal fold mobility as the main prognostic factor of treatment outcomes and survival in stage II squamous cell carcinomas of the glottic larynx. J Laryngol Otol 129:903–909
Hendriksma M, Ruler M, Verbist BM et al (2019) Survival and prognostic factors for outcome after radiotherapy for T2 glottic carcinoma. Cancers (Basel) 11:E1319
Dixon LM, Douglas CM, Shaukat SI et al (2017) Conventional fractionation should not be the standard of care for T2 glottic cancer. Radiat Oncol 12:178
Forastiere AA, Goepfert H, Maor M et al (2003) Concurrent chemotherapy and radiotherapy for organ preservation in advanced laryngeal cancer. N Engl J Med 349:2091–2098
Wolf GT, Fisher SG, Hong WK et al (1991) Induction chemotherapy plus radiation compared with surgery plus radiation in patients with advanced laryngeal cancer. N Engl J Med 324:1685–1690
Jones DA, Mendenhall CM, Kirwan J et al (2010) Radiation therapy for management of t1–t2 glottic cancer at a private practice. Am J Clin Oncol 33:587–590
Nakayama M, Hayakawa K, Okamoto M, Niibe Y, Ishiyama H, Kotani S (2010) Phase I/II trial of concurrent use of S-1 and radiation therapy for T2 glottic cancer. Jpn J Clin Oncol 40:921–926
Itoh Y, Fuwa N (2006) Retrospective analysis: concurrent chemoradiotherapy using protracted continuous infusion of low-dose cisplatin and 5-fluorouracil for T2N0 glottic cancer. Radiat Med 24:277–281
Hirasawa N, Itoh Y, Naganawa S et al (2012) Multi-institutional analysis of early glottic cancer from 2000 to 2005. Radiat Oncol 7:122
Kitani Y, Kubota A, Furukawa M, Sato K (2016) Prognostic factors for local control in patients receiving radiation therapy for early glottic cancer: anterior commissure involvement and effect of chemoradiotherapy. Eur Arch Otorhinolaryngol 273:1011–1017
Cheraghlou S, Kuo P, Judson BL (2017) Treatment delay and facility case volume are associated with survival in early-stage glottic cancer. Laryngoscope 127:616–622
Vokes EE, Stenson KM (2003) Therapeutic options for laryngeal cancer. N Engl J Med 349:2087–2089
Laccourreye O, Marret G, Rubin F et al (2019) Ten-year outcome of curative “exclusive” chemotherapy in N0M0 squamous cell carcinoma of the larynx and pharynx with complete clinical response. Head Neck 41:2190–2196
Acknowledgements
We are grateful to Dr. Hui-Zi Li for his technical support.
Funding
The work was supported by Grants from the National Natural Science Foundation of China (no. 81172561), the Ministry of Science and Technology of the People’s Republic of China (no. 2014CB541700), and Department of Science and Technology of Zhejiang Province (no. 2015C03035).
Author information
Authors and Affiliations
Contributions
G-JH and B-BY performed the conception and design of this manuscript. G-JH and B-BY performed data analysis and prepared the figure. G-JH and B-BY drafted and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest in the work.
Ethical approval
Ethics approval and patient written informed consent were not required, because all analyses in our study were mainly performed based on data from the SEER database.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
405_2020_6557_MOESM2_ESM.tif
Supplementary file1Multivariable analyses based on patients’ characteristics. A, Forest plot for the OS. Patients who were married or underwent primary site surgery or underwent radiation would have a better OS, whereas patients of age≥65 would suffer a worse OS. Chemotherapy (p=0.13) was not considered as an independent predictor of OS. B, Forest plot for the CSS. Patients who were married or underwent primary site surgery or underwent radiation would have a better CSS, whereas patients who were age≥65 or underwent chemotherapy would suffer a worse CSS. Abbreviations: OS, overall survival; CSS, cancer-specific survival (TIF 4609 KB)
405_2020_6557_MOESM3_ESM.tif
Supplementary file2Cumulative incidence of cancer-specific death and death of other causes on various factors (1, cancer-specific death; 2, death of other causes). A, Age. B, Sex. C, Race. D, Marital status. E, Grade. F, Year of diagnosis. G, Primary site surgery. H, Radiation. I, Chemotherapy (TIF 4639 KB)
405_2020_6557_MOESM4_ESM.tif
Supplementary file3Survival analyses of chemotherapy based on the training and the validation cohorts. A, Patients’ OS after chemotherapy in the training cohort. B, Patients’ CSS after chemotherapy in the training cohort. C, Patients’ OS after chemotherapy in the validation cohort. D, Patients’ CSS after chemotherapy in the validation cohort. Abbreviations: OS, overall survival; CSS, cancer-specific survival (TIF 3701 KB)
405_2020_6557_MOESM5_ESM.docx
Supplementary file4Survival analyses between chemotherapy (Yes) and chemotherapy (No/unknown) groups after PSM. A, Patients’ OS after chemotherapy. B, Patients’ CSS after chemotherapy. C, Patients’ OS after chemotherapy plus primary site surgery. D, Patients’ CSS after chemotherapy plus primary site surgery. E, Patients’ OS after chemotherapy plus radiation. F, Patients’ CSS after chemotherapy plus radiation. G, Patients’ OS after chemotherapy plus primary site surgery and radiation. H, Patients’ CSS after chemotherapy plus primary site surgery and radiation. Abbreviations: OS, overall survival; CSS, cancer-specific survival; PSM, propensity-score matching (TIF 9348 KB)
Rights and permissions
About this article
Cite this article
Huang, GJ., Yang, BB. Prognostic risk factors for initially diagnosed T2N0M0 glottic cancer: competing risk analysis and propensity-score matched cohort analysis. Eur Arch Otorhinolaryngol 278, 3921–3931 (2021). https://doi.org/10.1007/s00405-020-06557-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-020-06557-9