Abstract
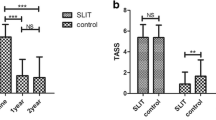
In the present study, we investigated the outcomes of sublingual immunotherapy (SLIT) in house dust mite-induced allergic rhinitis (HDM-AR) patients. In this prospective, multicentric study, 186 patients with AR who had positive skin prick test results for HDMs were included. The patients were administered SLIT using Staloral 300 for 1 year. Evaluation of the patients regarding symptom scores, clinical findings and Rhinitis Quality of Life Questionnaire (RQLQ) scores was performed at baseline, and then at 6 and 12 months of therapy. Our results showed that, for all of the evaluated items (symptom scores, clinical findings and RQLQ scores), 12-month values were significantly lower than those at 6 months and baseline. Similarly, 6-month values were significantly lower than those at baseline. There were no complications in any of our patients. SLIT for HDM-AR is a treatment modality that can be used safely. We obtained better results than expected, and the treatment showed a positive psychological effect; the patients believed that SLIT was the final step of treatment and, which made them feel better.



Similar content being viewed by others
References
Wilson DR, Lima MT, Durham SR (2005) Sublingual immunotherapy for allergic rhinitis. Systematic review and meta-analysis. Allergy 60:4–12
Bahceciler NN, Arikan C, Taylor A et al (2005) Impact of sublingual immunotherapy on specific antibody levels in asthmatic children allergic to house dust mites. Int Arch Allergy Immunol 136(3):287–294
Platts-Mills TAE, Erwin EA, Heymann PW, Woodfolk JA (2009) Pro: the evidence for a causal role of dust mites in asthma. Am J Respir Crit Care Med 180:109–121
Sporik R, Chapman MD, Platts-Mills TA (1992) House dust mite exposure as a cause of asthma. Clin Exp Allergy 22:897–906
Calderon MA, Casale TB, Nelson HS, Demoly P (2013) An evidence-based analysis of house dust mite allergen immunotherapy: a call for more rigorous clinical studies. J Allergy Clin Immunol 132(6):1322–1336
WMA, Declaration Of Helsinki (2008) Ethical principles for medical research ınvolving human subjects. In: 59th WMA general assembly, W.M. Association, Editor, Seoul
Juniper EF, Guyatt GH (1991) Development and testing of a new measure of health status for clinical trials in rhinoconjunctivitis. Clin Exper Allergy 21:77–83
Juniper EF, Guyatt GH, Griffith LE, Ferrie PJ (1996) Interpretation of rhinoconjunctivitis quality of life questionnaire data. J Allergy Clin Immunol 98:843–845
Lachapelle JM, Maibach HI (2009) Patch Testing and Prick Testing: A Practical Guide, vol 2. Springer, Berlin
Cingi C, Gunhan K, Gage-White L, Unlu H (2010) Efficacy of Leukotriene Antagonists as Concomitant Therapy in Allergic Rhinitis. Laryngoscope 120:1718–1723
Radulovic S, Calderon MA, Wilson D, Durham S (2010) Sublingual immunotherapy for allergic rhinitis. Cochrane Database Syst Rev 12:CD002893
Zureik M, Neukirch C, Leynaert B, Liard R, Bousquet J, Neukirch F (2002) Sensitisation to airborne moulds and severity of asthma: cross sectional study from European Community respiratory health survey. BMJ 325:411–414
Linneberg A, Henrik Nielsen N, Frolund L, Madsen F, Dirksen A, Jorgensen T (2002) The link between allergic rhinitis and allergic asthma: a prospective populationbased study. The Copenhagen Allergy Study. Allergy 57:1048–1052
Mosbech H (1985) House dust mite allergy. Allergy 40:81–91
Bergmann KC, Demoly P, Worm M et al (2014) Efficacy and safety of sublingual tablets of house dust mite allergen extractsin adults with allergic rhinitis. J Allergy Clin Immunol 133(6):1608.e6–1614.e6
Trebuchon F, Lhéritier-Barrand M, David M, Demoly P (2014) Characteristics and management of sublingual allergen immunotherapy in children with allergic rhinitis and asthma induced by house dust mite allergens. Clin Transl Allergy 29(4):15
Wise SK, Woody J, Koepp S, Schlosser RJ (2009) Quality of life outcomes with sublingual immunotherapy. Am J Otolaryngol 30(5):305–311. doi:10.1016/j.amjoto.2008.06.003
Wilson DR, Torres LI, Durham SR (2003) Sublingual immunotherapy for allergic rhinitis. Cochrane Database Syst Rev. 2:CD002893
Wang DH, Chen L, Cheng L et al (2013) Fast onset of action of sublingual immunotherapy in house dust mite-induced allergic rhinitis: a multicenter, randomized, double-blind, placebo-controlled trial. Laryngoscope 123(6):1334–1340. doi:10.1002/lary.23935
Bozek A, Ignasiak B, Filipowska B, Jarzab J (2013) House dust mite sublingual immunotherapy: a double-blind, placebo-controlled study in elderly patients with allergic rhinitis. Clin Exp Allergy 43(2):242–248
Queirós MG, Silva DA, Siman IL et al (2013) Modulation of mucosal/systemic antibody response after sublingual immunotherapy in mite-allergic children. Pediatr Allergy Immunol 24(8):752–761. doi:10.1111/pai.12163
Acknowledgments
“With the exception of data collection, preparation of this paper, including the design and planning, was supported by the Continuous Education and Scientific Research Association”. Scientific support from the association, but no grant or funding support, was received.
Conflict of interest
The authors declare that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cingi, C., Bayar Muluk, N., Ulusoy, S. et al. Efficacy of sublingual immunotherapy for house dust mite allergic rhinitis. Eur Arch Otorhinolaryngol 272, 3341–3346 (2015). https://doi.org/10.1007/s00405-014-3444-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-014-3444-1