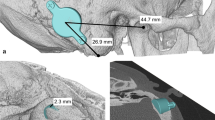
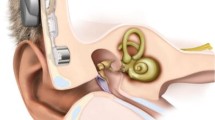
Abstract
The Bonebridge® (BB, Med-El) is a newly designed transcutaneous active bone conductive implant with functional outcome similar to percutaneous bone-anchored hearing systems (BAHS). It is currently approved only for patients ≥18 years. Since the BB allows the skin to remain intact and therefore should be able to overcome some of the issues related to percutaneous BAHS including skin reactions, wound infection and implant extrusion, it would be especially attractive for use in children. We present a preliminary series of the first three cases of BB implantation in children/adolescents (10–16 years). Two subjects were affected by conductive hearing loss (CHL) and one subject by single-sided deafness (SSD). The surgical procedure with transmastoid approach was completed in all cases without complications. Both subjects with CHL showed an increase in speech perception thresholds in quiet from preoperative unaided to 6 months postoperatively with BB of 37 dB, respectively, of 12 dB. The adolescent with SSD attained –3.1 dB unaided vs. –5.6 dB with the BB in the “speech and noise from the front” presentation and +0.5 unaided vs. –5.0 dB with the BB in the “speech from the unilateral deaf side/noise from the normal hearing side” presentation using the adaptive Oldenburg Sentence Test. The results show a straightforward surgical procedure and satisfactory functional gain after BB implantation also in children/adolescents. BB implantation in patients ≤18 years is currently an “off-label use” so that detailed information about alternative treatment options, operation risks and the lack of approval for use in children is essential.


Similar content being viewed by others
References
Håkansson B, Tjellström A, Rosenhall U, Carlsson P (1985) The bone-anchored hearing aid. Principal design and a psychoacoustical evaluation. Acta Otolaryngol 100(3-4):229–239
Vaneecloo FM, Ruzza I, Hanson JN, Gérard T, Dehaussy J, Cory M, Arrouet C, Vincent C (2001) The monaural pseudo-stereophonic hearing aid (BAHA) in unilateral total deafness: a study of 29 patients. Rev Laryngol Otol Rhinol 122(5):343–350
Wesarg T, Aschendorff A, Laszig R, Beck R, Schild C, Hassepass F, Kroeger S, Hocke T, Arndt S (2013) Comparison of speech discrimination in noise and directional hearing with 2 different sound processors of a bone-anchored hearing system in adults with unilateral severe or profound sensorineural hearing loss. Otol Neurotol 34(6):1064–1070
Linstrom CJ, Silverman CA, Yu GP (2009) Efficacy of the bone-anchored hearing aid for single-sided deafness. Laryngoscope 119(4):713–720
Christensen L, Dornhoffer JL (2008) Bone-anchored hearing aids for unilateral hearing loss in teenagers. Otol Neurotol 29(8):1120–1122
D’Eredità R, Caroncini M, Saetti R (2012) The new Baha implant: a prospective osseointegration study. Otolaryngol Head Neck Surg 146(6):979–983
House JW, Jr Kutz JW (2007) Bone-anchored hearing aids: incidence and management of postoperative complications. Otol Neurotol 28(2):213–217
Faber HT, Dun CA, Nelissen RC, Mylanus EA, Cremers CW, Hol MK (2013) Bone-anchored hearing implant loading at 3 weeks: stability and tolerability after 6 months. Otol Neurotol 34(1):104–110
de Wolf MJ, Hol MK, Mylanus EA, Cremers CW (2009) Bone-anchored hearing aid surgery in older adults: implant loss and skin reactions. Ann Otol Rhinol Laryngol 118(7):525–531
Kraai T, Brown C, Neeff M, Fisher K (2011) Complications of bone-anchored hearing aids in pediatric patients. Int J Pediatr Otorhinolaryngol 75(6):749–753
Gründer I, Seidl RO, Ernst A, Todt I (2008) Relative value of BAHA testing for the postoperative audiological outcome. HNO 56(10):1020–1024
Denoyelle F, Leboulanger N, Coudert C, Mazzaschi O, Loundon N, Vicaut E, Tessier N, Garabedian EN (2013) New closed skin bone-anchored implant: preliminary results in 6 children with ear atresia. Otol Neurotol 34(2):275–281
Arndt S, Aschendorff A, Laszig R, Beck R, Schild C, Kroeger S, Ihorst G, Wesarg T (2011) Comparison of pseudobinaural hearing to real binaural hearing rehabilitation after cochlear implantation in patients with unilateral deafness and tinnitus. Otol Neurotol 32(1):39–47
Mulla O, Agada F, Reilly PG (2012) Introducing the Sophono Alpha 1 abutment free bone conduction hearing system. Clin Otolaryngol 37(2):168–169
Barbara M, Biagini M, Monini S (2011) The totally implantable middle ear device “esteem” for rehabilitation of severe sensorineural hearing loss. Acta Otolaryngol 131(4):399–404
Sprinzl G, Lenarz T, Ernst A, Hagen R, Wolf-Magele A, Mojallal H, Todt I, Mlynski R, Wolframm MD (2013) First European multicenter results with a new transcutaneous bone conduction hearing implant system: short-term safety and efficacy. Otol Neurotol 34(6):1076–1083
Lassaletta L, Sanchez-Cuadrado I, Muñoz E, Gavilan J (2014) Retrosigmoid implantation of an active bone conduction stimulator in a patient with chronic otitis media. Auris Nasus Larynx 41(1):84–87
Garfin SR, Roux R, Botte MJ, Centeno R, Woo SL (1986) Skull osteology as it affects halo pin placement in children. J Pediatr Orthop 6(4):434–436
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hassepass, F., Bulla, S., Aschendorff, A. et al. The bonebridge as a transcutaneous bone conduction hearing system: preliminary surgical and audiological results in children and adolescents. Eur Arch Otorhinolaryngol 272, 2235–2241 (2015). https://doi.org/10.1007/s00405-014-3137-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-014-3137-9