Abstract
Purpose
Although adenomyosis is a common and benign gynecological disease, the specific pathogenesis of this condition is yet to be fully elucidated. It is difficult to culture primary cells of the ectopic endometrial epithelia and stroma from human adenomyosis lesions. Most of the previous of studies on adenomyosis were based on primary eutopic endometrium cells. However, as yet, no efficient protocols have been developed for the isolation, culture or purification of primary ectopic epithelial and stromal cells from human adenomyosis lesions. Therefore, the present study aimed to develop an efficient protocol for the isolation and culture of primary ectopic epithelial and stromal cells from human adenomyosis lesions.
Methods
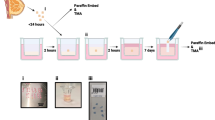
In the present study, we aimed to obtain ectopic endometrium tissue from human adenomyosis foci and use a simple and operable type I collagenase digestion method for primary culture. Cells were isolated by sterile cell strainer filtration and flow cytometry was performed to identify, purify, and evaluate the viability of isolated ectopic endometrial cells.
Results
Using our method, we successfully isolated and cultured highly purified and active ectopic endometrial epithelial and stromal cells from human adenomyosis foci. Ep-CAM was expressed in ectopic epithelial cells of human adenomyosis with a purity of 93.74% and a viability of 80.58%. In addition, CD10 were robustly expressed by ectopic stromal cells in human adenomyosis. Cellular purity and viability were determined to be 96.37 and 93.49%, respectively.
Conclusion
Our method provides a new experimental model for studying the molecular pathogenesis of human adenomyosis.






Similar content being viewed by others
Data availability
All the data we did were uploaded and saved in our hospital’s library. As it involves patient privacy and other relevant national policies, it is not provided in this manuscript. However, if anyone is interested in it, please contact the corresponding author directly.
References
Harada T, Khine YM, Kaponis A, Nikellis T, Decavalas G, Taniguchi F (2016) The impact of adenomyosis on women’s fertility. Obstet Gynecol Surv 71:557–568. https://doi.org/10.1097/OGX.0000000000000346
Chapron C, Vannuccini S, Santulli P, Abrão MS, Carmona F, Fraser IS, Gordts S, Guo S-W, Just P-A, Noël J-C, Pistofidis G, Van den Bosch T, Petraglia F (2020) Diagnosing adenomyosis: an integrated clinical and imaging approach. Hum Reprod Update 26:392–411. https://doi.org/10.1093/humupd/dmz049
Donnez J, Stratopoulou CA, Dolmans M-M (2021) Uterine adenomyosis: from disease pathogenesis to a new medical approach using GnRH antagonists. Int J Environ Res Public Health. https://doi.org/10.3390/ijerph18199941
Donnez O, Donnez J (2020) Gonadotropin-releasing hormone antagonist (linzagolix): a new therapy for uterine adenomyosis. Fertil Steril 114:640–645. https://doi.org/10.1016/j.fertnstert.2020.04.017
Camboni A, Marbaix E (2021) Ectopic endometrium: the pathologist’s perspective. Int J Mol Sci. https://doi.org/10.3390/ijms222010974
Pistofidis G, Makrakis E, Koukoura O, Bardis N, Balinakos P, Anaf V (2014) Distinct types of uterine adenomyosis based on laparoscopic and histopathologic criteria. Clin Exp Obstet Gynecol 41:113–118
Bulun SE, Yildiz S, Adli M, Wei J-J (2021) Adenomyosis pathogenesis: insights from next-generation sequencing. Hum Reprod Update 27:1086–1097. https://doi.org/10.1093/humupd/dmab017
关永格, 李坤寅, 何昱雯, 宋阳, 子宫腺肌病病灶细胞的培养与鉴定, 中国妇幼健康研究, (2013) 827–829+838,
Habiba M, Benagiano G (2021) Classifying adenomyosis: progress and challenges. Int J Environ Res Public Health. https://doi.org/10.3390/ijerph182312386
Zafari P, Rafiei A, Faramarzi F, Ghaffari S, Amiri AH, Taghadosi M (1992) Human fibroblast-like synoviocyte isolation matter: a comparison between cell isolation from synovial tissue and synovial fluid from patients with rheumatoid arthritis. Rev Assoc Med Bras 67(2021):1654–1658. https://doi.org/10.1590/1806-9282.20210706
Wang Y, Chen S, Yan Z, Pei M (2019) A prospect of cell immortalization combined with matrix microenvironmental optimization strategy for tissue engineering and regeneration. Cell Biosci 9:7. https://doi.org/10.1186/s13578-018-0264-9
Joplin R (1994) Isolation and culture of biliary epithelial cells. Gut 35:875–878
Abade Dos Santos FA, Carvalho CL, Almeida I, Fagulha T, Rammos F, Barros SC, Henriques M, Luís T, Duarte MD (2021) Simple method for establishing primary leporidae skin fibroblast cultures. Cells. https://doi.org/10.3390/cells10082100
Hu Z, Chen Y, Gao M, Chi X, He Y, Zhang C, Yang Y, Li Y, Lv Y, Huang Y, Deng X (2023) Novel strategy for primary epithelial cell isolation: combination of hyaluronidase and collagenase I. Cell Prolif 56:e13320. https://doi.org/10.1111/cpr.13320
Yu Q, Wang J, Li T, Xu X, Guo X, Ding S, Zhu L, Zou G, Chen Y, Zhang X (2021) RON mediates tumor-promoting effects in endometrial adenocarcinoma. Biomed Res Int 2021:2282916. https://doi.org/10.1155/2021/2282916
Fedele L, Bianchi S, Dorta M, Arcaini L, Zanotti F, Carinelli S (1992) Transvaginal ultrasonography in the diagnosis of diffuse adenomyosis. Fertil Steril 58:94–97
Mark AS, Hricak H, Heinrichs LW, Hendrickson MR, Winkler ML, Bachica JA, Stickler JE (1987) Adenomyosis and leiomyoma: differential diagnosis with MR imaging. Radiology 163:527–529
Zhang M, Bazot M, Tsatoumas M, Munro MG, Reinhold C (2023) MRI of adenomyosis: where are we today? Can Assoc Radiol J 74:58–68. https://doi.org/10.1177/08465371221114197
Celli V, Dolciami M, Ninkova R, Ercolani G, Rizzo S, Porpora MG, Catalano C, Manganaro L (2022) MRI and adenomyosis: what can radiologists evaluate? Int J Environ Res Public Health. https://doi.org/10.3390/ijerph19105840
Li T, Wang J, Guo X, Yu Q, Ding S, Xu X, Peng Y, Zhu L, Zou G, Zhang X (2020) Possible involvement of crosstalk between endometrial cells and mast cells in the development of endometriosis via CCL8/CCR1. Biomed Pharmacother 129:110476. https://doi.org/10.1016/j.biopha.2020.110476
Bonser LR, Koh KD, Johansson K, Choksi SP, Cheng D, Liu L, Sun DI, Zlock LT, Eckalbar WL, Finkbeiner WE, Erle DJ (2021) Flow-cytometric analysis and purification of airway epithelial-cell subsets. Am J Respir Cell Mol Biol 64:308–317. https://doi.org/10.1165/rcmb.2020-0149MA
Zhang Y, An J, Liu M, Li N, Wang W, Yao H, Li N, Yang X, Sun Y, Xu N, Wu L (2020) Efficient isolation, culture, purification, and stem cell expression profiles of primary tumor cells derived from uterine cervical squamous cell carcinoma. Am J Reprod Immunol 84:e13251. https://doi.org/10.1111/aji.13251
Guo S-W, Benagiano G, Bazot M (2022) In search of an imaging classification of adenomyosis: a role for elastography? J Clin Med. https://doi.org/10.3390/jcm12010287
Guo S-W (2022) Cracking the enigma of adenomyosis: an update on its pathogenesis and pathophysiology. Reproduction 164:R101–R121. https://doi.org/10.1530/rep-22-0224
Acknowledgements
This work was funded by National Key R&D Program of China (Grant number: 2022YFC2704003), National Natural Science Foundation of China (Grant numbers: 81974225, 82001518 and 82171636).
Author information
Authors and Affiliations
Contributions
ZF: conceptualization, writing—original draft, data curation, methodology, writing—review and editing, validation, resources. JW and TL: conceptualization, methodology, writing—review and editing, validation, supervision. MY and YP: data curation, methodology, resources, validation. XZ: supervision, conceptualization, writing—review and editing, project administration, validation. All the authors read the submitted version and approved it.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflict of interest. The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fang, Z., Wang, J., Li, T. et al. A method for isolating and culturing ectopic epithelial and stromal cells to study human adenomyosis. Arch Gynecol Obstet 309, 551–563 (2024). https://doi.org/10.1007/s00404-023-07254-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-023-07254-8