Abstract
Purpose
To provide a narrative review of anogenital screening for human papillomavirus in solid organ transplant recipients.
Methods
Keyword searches of PubMed and Ovid MEDLINE databases were performed. Keywords included human papillomavirus, malignancy, cervical cancer, Pap smear, solid organ transplant, and immunosuppression. Manual searches were also conducted of other relevant journals and reference lists of primary articles.
Results
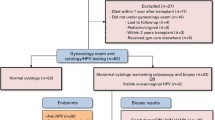
Forty-one studies, articles, or clinical practice guidelines across 25 years of literature were included. Eligible literature was written in English or offered an English translation.
Conclusion
Human papillomavirus-related anogenital malignancies disproportionately affect transplant recipients compared to the general population. Evidence-based guidelines for cervical cancer screening and prevention in transplant patients are lacking. Current practice guidelines generally agree on increased Pap screening for transplant recipients compared to the general population. However, recommended screening frequency differs between organizations and amongst medical specialties. Vaccination against HPV remains the most effective strategy to prevent HPV-driven pre-malignant and malignant lesions.
Similar content being viewed by others
Availability of data and materials
Not applicable to this work.
Code availability
Not applicable to this work.
References
Engels EA, Pfeiffer RM, Fraumeni JF, Kasiske BL, Israni AK, Snyder JJ, Wolfe RA, Goodrich NP, Bayakly AR, Clarke CA (2011) Spectrum of cancer risk among US solid organ transplant recipients. JAMA 306(17):1891–1901
Grulich AE, Van Leeuwen MT, Falster MO, Vajdic CM (2007) Incidence of cancers in people with HIV/AIDS compared with immunosuppressed transplant recipients: a meta-analysis. The Lancet 370(9581):59–67
Kasiske BL, Snyder JJ, Gilbertson DT, Wang C (2004) Cancer after kidney transplantation in the United States. Am J Transplant 4(6):905–913
Vajdic CM, McDonald SP, McCredie MR, Van Leeuwen MT, Stewart JH, Law M, Chapman JR, Webster AC, Kaldor JM, Grulich AE (2006) Cancer incidence before and after kidney transplantation. JAMA 296(23):2823–2831
Adami J, Gäbel H, Lindelöf B, Ekström K, Rydh B, Glimelius B, Ekbom A, Adami H-O, Granath F (2003) Cancer risk following organ transplantation: a nationwide cohort study in Sweden. Br J Cancer 89(7):1221–1227
Wiley D, Douglas J, Beutner K, Cox T, Fife K, Moscicki A-B, Fukumoto L (2002) External genital warts: diagnosis, treatment, and prevention. Clin Infect Dis 35(2):S210–S224
Euvrard S, Kanitakis J, Chardonnet Y, Noble CP, Touraine JL, Faure M, Thivolet J, Claudy A (1997) External anogenital lesions in organ transplant recipients: a clinicopathologic and virologic assessment. Arch Dermatol 133(2):175–178
Chin-Hong P, Kwak E (2013) Human papillomavirus in solid organ transplantation. Am J Transplant 13:189–200
(2016) Practice Bulletin No. 168: cervical cancer screening and prevention. Obstet Gynecol 128(4):e111–e130. https://doi.org/10.1097/aog.0000000000001708
Vaccarella S, Franceschi S, Engholm G, Lönnberg S, Khan S, Bray F (2014) 50 years of screening in the Nordic countries: quantifying the effects on cervical cancer incidence. Br J Cancer 111(5):965–969
Courtney AE, Leonard N, O’Neill CJ, McNamee PT, Maxwell AP (2009) The uptake of cervical cancer screening by renal transplant recipients. Nephrol Dial Transplant 24(2):647–652
Meeuwis KA, Melchers WJ, Bouten H, van de Kerkhof PC, Hinten F, Quint WG, Massuger LF, Hoitsma AJ, van Rossum MM, de Hullu JA (2012) Anogenital malignancies in women after renal transplantation over 40 years in a single center. Transplantation 93(9):914–922
Weinstock H, Berman S, Cates W Jr (2004) Sexually transmitted diseases among American youth: incidence and prevalence estimates, 2000. Perspect Sex Reprod Health 36(1):6–10
Markowitz LE, Dunne EF, Saraiya M, Chesson HW, Curtis CR, Gee J, Bocchini JA Jr, Unger ER (2014) Human papillomavirus vaccination: recommendations of the Advisory Committee on Immunization Practices (ACIP). Morb Mortal Wkly Rep 63(5):1–30
Chelimo C, Wouldes TA, Cameron LD, Elwood JM (2013) Risk factors for and prevention of human papillomaviruses (HPV), genital warts and cervical cancer. J Infect 66(3):207–217
Centers for Disease Control and Prevention (2018) Cancers associated with human papillomavirus, United States—2011–2015 USCS data brief, no. 4. Atlanta, GA: Centers for Disease Control and Prevention, 2018. https://www.cdc.gov/cancer/uscs/about/data-briefs/no4-hpv-assoc-cancers-UnitedStates-2011-2015.htm. Accessed 24 Jan 2021
Kutlubay Z, Engin B, Zara T, Tüzün Y (2013) Anogenital malignancies and premalignancies: facts and controversies. Clin Dermatol 31(4):362–373
Connolly K, Manders P, Earls P, Epstein RJ (2014) Papillomavirus-associated squamous skin cancers following transplant immunosuppression: one Notch closer to control. Cancer Treat Rev 40(2):205–214
Tanaka Y, Ueda Y, Kakuda M, Kubota S, Matsuzaki S, Nakagawa S, Egawa-Takata T, Matsuzaki S, Kobayashi E, Yoshino K (2016) Clinical outcomes of abnormal cervical cytology and human papillomavirus-related lesions in patients with organ transplantation: 11-year experience at a single institution. Int J Clin Oncol 21(4):730–734
Casolati E, Agarossi A, Valieri M, Ferrazzi E (2003) Vulvar neoplasia in HIV positive women: a review. Med Wieku Rozwoj 7(4 Pt 1):487–493
Martelli-Marzagão F, Santos Junior G, Ogawa M, Enokihara M, Porro A, Tomimori J (2016) Human papillomavirus detected in viral warts of renal transplant recipients. Transpl Infect Dis 18(1):37–43
Reusser NM, Downing C, Guidry J, Tyring SK (2015) HPV carcinomas in immunocompromised patients. J Clin Med 4(2):260–281
Curry SJ, Krist AH, Owens DK, Barry MJ, Caughey AB, Davidson KW, Doubeni CA, Epling JW, Kemper AR, Kubik M (2018) Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA 320(7):674–686
Fontham ET, Wolf AM, Church TR, Etzioni R, Flowers CR, Herzig A, Guerra CE, Oeffinger KC, Shih YCT, Walter LC (2020) Cervical cancer screening for individuals at average risk: 2020 guideline update from the American Cancer Society. CA Cancer J Clin 70(5):321–346
Cuzick J, Sasieni P, Davies P, Adams J, Normand C, Frater A, Van Ballegooijen M, Van den Akker-Van Marle E (2000) A systematic review of the role of human papilloma virus (HPV) testing within a cervical screening programme: summary and conclusions. Br J Cancer 83(5):561–565
Ronco G, Dillner J, Elfström KM, Tunesi S, Snijders PJ, Arbyn M, Kitchener H, Segnan N, Gilham C, Giorgi-Rossi P (2014) Efficacy of HPV-based screening for prevention of invasive cervical cancer: follow-up of four European randomised controlled trials. The Lancet 383(9916):524–532
World Health Organization (2021) WHO guideline for screening and treatment of cervical pre-cancer lesions for cervical cancer prevention, 2nd ed. World Health Organization. https://apps.who.int/iris/handle/10665/342365. License: CC BY-NC-SA 3.0 IGO
European best practice guidelines for renal transplantation (2002) Section IV: long-term management of the transplant recipient. IV.6.3. Cancer risk after renal transplantation. Solid organ cancers: prevention and treatment. Nephrol Dial Transplant 17(4):32–36
Kasiske BL, Vazquez MA, Harmon WE, Brown RS, Danovitch GM, Gaston RS, Roth D, Scandling JD, Singer GG (2000) Recommendations for the outpatient surveillance of renal transplant recipients. J Am Soc Nephrol 11(suppl 1):S1–S86
Acuna S, Huang J, Scott A, Micic S, Daly C, Brezden-Masley C, Kim S, Baxter N (2017) Cancer screening recommendations for solid organ transplant recipients: a systematic review of clinical practice guidelines. Am J Transplant 17(1):103–114
Wong G, Chapman JR, Craig JC (2008) Cancer screening in renal transplant recipients: what is the evidence? Clin J Am Soc Nephrol 3(Supplement 2):S87–S100
Moscicki A-B, Flowers L, Huchko MJ, Long ME, MacLaughlin KL, Murphy J, Spiryda LB, Gold MA (2019) Guidelines for cervical cancer screening in immunosuppressed women without HIV infection. J Low Genit Tract Dis 23(2):87–101
Chin-Hong PV (2016) Human papillomavirus in kidney transplant recipients. Semin Nephrol 36(397):404
Benson CA, Brooks JT, Holmes KK, Kaplan JE, Masur H, Pau A (2009) Guidelines for prevention and treatment opportunistic infections in HIV-infected adults and adolescents; recommendations from CDC, the National Institutes of Health, and the HIV Medicine Association/Infectious Diseases Society of America
Moscicki A-B, Hills N, Shiboski S, Powell K, Jay N, Hanson E, Miller S, Clayton L, Farhat S, Broering J (2001) Risks for incident human papillomavirus infection and low-grade squamous intraepithelial lesion development in young females. JAMA 285(23):2995–3002
Medical Care Criteria Committee (2022) Screening for anal dysplasia and cancer in adults With HIV. Clinical Guidelines Program, New York State Department of Health AIDS Institute. https://www.hivguidelines.org/hivcare/anal-cancer/#tab_3
Food & Administration D (2018) Prescribing information [package insert]. Gardasil 9 (human papillomavirus 9-valent vaccine, recombinant). Silver Spring, MD: US Department of Health and Human Services. Food and Drug Administration
Palefsky JM, Giuliano AR, Goldstone S, Moreira ED Jr, Aranda C, Jessen H, Hillman R, Ferris D, Coutlee F, Stoler MH (2011) HPV vaccine against anal HPV infection and anal intraepithelial neoplasia. N Engl J Med 365(17):1576–1585
Petrosky E, Bocchini JA Jr, Hariri S, Chesson H, Curtis CR, Saraiya M, Unger ER, Markowitz LE (2015) Use of 9-valent human papillomavirus (HPV) vaccine: updated HPV vaccination recommendations of the advisory committee on immunization practices. MMWR Morb Mortal Wkly Rep 64(11):300
Eckert LON (2020) Human papillomavirus vaccination ACOG committee opinion, number 809. Obstetr Gynecol 136(2):E15–E21
Meites E, Szilagyi PG, Chesson HW, Unger ER, Romero JR, Markowitz LE (2019) Human papillomavirus vaccination for adults: updated recommendations of the Advisory Committee on Immunization Practices. In: Wiley Online Library
Funding
No funding was used for this project.
Author information
Authors and Affiliations
Contributions
All the authors have seen and approved this manuscript as it is written and have contributed significantly to its work. CP was involved with conceptualization, project development, literature search, data collection, data management, and manuscript writing and editing. ML was active in manuscript writing and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethics approval
Not applicable to this work.
Consent to participate
Not applicable to this work.
Consent for publication
Not applicable to this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Papastamelos, C., Linder, M. Human papillomavirus anogenital screening in solid organ transplant recipients: a narrative review. Arch Gynecol Obstet 307, 1277–1283 (2023). https://doi.org/10.1007/s00404-022-06577-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-022-06577-2