Abstract
Introduction
Hereditary factor (F) XIII-deficiency is a known risk factor for postoperative complications, but data of acquired FXIII-deficiency in malignancies are limited. Therefore, we evaluated the role of acquired FXIII-deficiency in surgery for advanced epithelial ovarian cancer (EOC).
Materials and methods
We performed a retrospective analysis of patients with known serum FXIII status and treatment between 2011 and 2018 at our center. We defined cohorts according to FXIII with values > 75% as normal (group A), 55–75% as reduced (group B) and < 55% as low (group C). Complications were classified according to the Clavien–Dindo Classification, class III–V complications were defined as severe.
Results
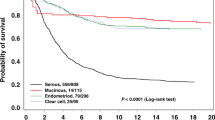
347 patients with EOC were identified. 180 patients (51.2%) were in group A, 82 patients (23.6%) in group B, and 85 patients (24.4%) in group C. Lower levels of FXIII were associated with higher amount of ascites, FIGO IV, high grade serous histology, low albumin, and higher CA-125 levels. Regarding intraoperative variables, low FXIII was associated with longer duration of surgery, higher blood loss, higher surgical complexity score/number of bowel anastomosis and a higher probability for macroscopic residual disease. The risk of severe complications in group A was 12.2%, 24.4% in group B, and 31.8% in group C. In a multivariate model, low FXIII (OR 2.8), > 1 bowel anastomosis (OR 2.7), age-adjusted Charlson comorbidity index ≥ 4 (OR 3.6) and a longer duration of surgery (> 285 min.) were significant predictive factors for severe complications.
Conclusion
FXIII is associated with tumor and treatment burden. A low level of FXIII is associated with postoperative complications. The knowledge about the presurgical serum FXIII-level might be helpful to plan the treatment strategy.


Similar content being viewed by others
References
S3-Leitlinie (2019) Maligne ovarialtumoren, Ovarialkarzinom, p 79
Nagy JA, Masse E, Herzberg K et al (1995) Pathogenesis of ascites tumor growth: fibrinogen influx and fibrin accumulation in tissues lining the peritoneal cavity. Cancer Res 55:369–375
Dvorak HF (1986) Tumors: wounds that do not heal Similarities between tumor stroma generation and wound healing. N Engl J Med 315:1650–1659
Wang X, Wang E, Kavanagh J et al (2005) Ovarian cancer, the coagulation pathway and inflammation. J Transl Med 3:25
Shi DY, Wang SJ (2017) Advances of coagulation factor XIII. Chin Med J 130:219–223
Dufner GS, Marbet GA (2002) Factor XIII in man: a review. Hamostaseologie 22(1):11–19
Andersson C, Kvist P, McEllhiney K et al (2015) Factor XIII transglutaminase supports the resolution of mucosal damage in experimental colitis. PLoS ONE 10:e0128113
Inbal A, Lubetsky A, Krapp T et al (2005) Impaired wound healing in factor XIII deficient mice. Thromb Haemost 94:432–437
Higaki S, Nakano K, Onaka S et al (2006) Clinical significance of measuring blood coagulation factor XIIIA regularly and continuously in patients with Crohn’s disease. J Gastroenterol Hepatol 21:1407–1411
Palumbo JS, Barney KA, Blevins EA et al (2008) Factor XIII transglutaminase supports hematogenous tumor cell metastasis through a mechanism dependent on natural killer cell function. J Thromb Haemost 6:812–819
Muszbek L, Katona É, Kerényi A (2017) Assessment of factor XIII. Methods Mol Biol 1646:277–293
Dorgalaleh A, Tabibian S, Shams M et al (2019) A unique factor XIII mutation in southeastern iran with an unexpectedly high prevalence: khash factor XIII. Semin Thromb Hemost 45(1):43–49
Ichinose A (2017) Japanese Collaborative Research Group on AH13. Autoimmune acquired factor XIII deficiency due to anti-factor XIII/13 antibodies: A summary of 93 patients. Blood Rev 31(1):37–45
Pötzsch B, Madlener K (2002) Gerinnungskonsil. Thieme Verlag, Rationelle Diagnostik und Therapie von Gerinnungsstörungen. Stuttgart
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213
Lee SH, Suh IB, Lee EJ et al (2013) Relationships of coagulation factor XIII activity with cell-type and stage of non-small cell lung cancer. Yonsei Med J 54(6):1394–1399
Kappelmayer J, Simon A, Katona E et al (2005) Coagulation factor XIII-A. A flow cytometric intracellular marker in the classification of acute myeloid leukemias. Thromb Haemost 94(2):454–9.
Harter P, Sehouli J, Lorusso D et al (2019) A randomized trial of lymphadenectomy in patients with advanced ovarian neoplasms. N Engl J Med 380:822–832
Kehoe S, Hook J, Nankivell M, Jayson GC, Kitchener H, Lopes T, Luesley D, Perren T, Bannoo S, Mascarenhas M, Dobbs S, Essapen S, Twigg J, Herod J, McCluggage G, Parmar M, Swart AM (2015) Primary chemotherapy versus primary surgery for newly diagnosed advanced ovarian cancer (CHORUS): an open-label, randomised, controlled, non-inferiority trial. Lancet 386(9990):249–257
Kahl A, du Bois A, Harter P et al (2017) Prognostic Value of the Age-Adjusted Charlson Comorbidity Index (ACCI) on Short- and Long-Term Outcome in Patients with Advanced Primary Epithelial Ovarian Cancer. Ann Surg Oncol 24(12):3692–3699
Obermair A, Hagenauer S, Tamandl D et al (2001) Safety and efficacy of low anterior en bloc resection as part of cytoreductive surgery for patients with ovarian cancer. Gynecol Oncol 83:115–120
Ataseven B, du Bois A, Reinthaller A et al (2015) Pre-operative serum albumin is associated with post-operative complication rate and overall survival in patients with epithelial ovarian cancer undergoing cytoreductive surgery. Gynecol Oncol 138(3):560–565
Moore KN, Reid NS, Fong DN et al (2008) Ovarian cancer in octogenarian: does the paradigm of cytoreductive surgery and chemotherapy still apply? Gynecol Oncol 110(2):133–139
Aletti GD, Eisenhauer EL, Santillan A et al (2011) Identification of patient groups at highest risk from traditional approach to ovarian cancer treatment. Gynecol Oncol 120(1):23–28
Nieuwenhuyzen-de Boer GM, Gerestein CG, Eijkemans MJ, Burger CW, Kooi GS (2016) Nomogram for 30-day morbidity after primary cytoreductive surgery for advanced stage ovarian cancer. Eur J Gynaecol Oncol 37(1):63–68
Grimm C, Harter P, Alesina PF et al (2017) The impact of type and number of bowel resections on anastomotic leakage risk in advanced ovarian cancer surgery. Gynecol Oncol 146(3):498–503
Bristow RE, Tomacruz RS, Armstrong DK, Trimble EL, Montz FJ (2002) Survival effect of maximal cytoreductive surgery for advanced ovarian carcinoma during the platinum era: a meta-analysis. J Clin Oncol 20(5):1248–1259
Du Bois A, Reuss A, Pujade-Lauraine E et al (2009) The role of surgical outcome as prognostic factor in advanced epithelial ovarian cancer. Cancer 15:1234–1244
Author information
Authors and Affiliations
Contributions
MY: Conceptualization, Investigation, Supervision, original draft; Writing—review and editing. HG: Conceptualization, Investigation, Writing—review and editing. BA: Investigation, Writing—review and editing. SS: Investigation, Writing—review and editing. TB: Investigation, Writing—review and editing. MB: Investigation, Writing—review and editing. AT: Statistical analysis, Writing—review and editing. AE-W: Investigation, Writing—review and editing. UB: Investigation, Writing—review and editing. RS: Investigation, Writing—review and editing. AdB: Investigation, Writing—review and editing. UW: Investigation, Writing—review and editing. PH: Conceptualization, Investigation, Supervision, original draft; Writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest regarding the submitted work. Dr. Ataseven reports personal fees and non-financial support from Roche, personal fees from Amgen, personal fees from Astra Zeneca, personal fees and non-financial support from Tesaro/GSK, personal fees from Clovis, personal fees from Celgene, non-financial support from PharmaMar, outside the submitted work. Prof. du Bois reports personal fees and non-financial support from Roche, Astra Zeneca, Tesaro/GSK, Clovis, BIOCAD, non-financial support from Pfizer, Genmab/Seattle Genetics and MSD, outside the submitted work. Dr. Harter reports grants and personal fees from Astra Zeneca, grants and personal fees from Roche, personal fees from Sotio, grants and personal fees from Tesaro, personal fees from Stryker, personal fees from Zai Lab, personal fees from MSD, grants and personal fees from Public funding (ASCO, DKH, DFG), personal fees from Clovis, personal fees from Immunogen, grants and personal fees from GSK, grants from Boehringer Ingelheim, grants from Medac, grants from Genmab, outside the submitted work.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yazdian, M., Groeben, H., Ataseven, B. et al. The role of factor XIII in surgery for advanced stage of epithelial ovarian cancer. Arch Gynecol Obstet 305, 1311–1318 (2022). https://doi.org/10.1007/s00404-021-06308-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-021-06308-z