Abstract
Purpose
To investigate clinicopathological characteristics and oncological outcome of women with microinvasive BOTs.
Methods
A retrospective multicenter case–control study was conducted on 902 patients with BOT, who underwent surgery from January 2002 to December 2015 at six participating gynecologic oncology centers from Turkey. Among 902 patients, 69 had microinvasive BOT. For every patient with microinvasive BOT, two controls were randomly selected from another database based on decade of age and stage of disease at diagnosis. The clinical–pathological characteristics and oncological outcomes were compared between BOT patients with and without stromal microinvasion. Risk factors for poor oncological outcomes were investigated in a multivariate analysis model. Overall survival (OS) and disease-free survival (DFS) were estimated using the Kaplan–Meier method.
Results
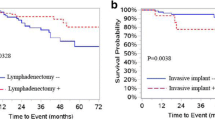
Patients with microinvasive BOT had a significantly higher rate of recurrence than patients without microinvasive BOT (17.4 vs 7.8%, OR 3.55, %95 CI 1.091–11.59, p = 0.03). Stage at diagnosis (stage I versus II/III) and type of surgery (cystectomy versus others) were found as other significant prognostic factors for recurrence in multivariate analysis (OR 8.63, %95 CI 2.48–29.9, p = 0.001 and OR 19.4, %95 CI 3.59–105.6, p = 0.001, respectively). Stromal microinvasion was found as a prognostic factor for significantly shorter DFS (26.7 vs 11.9 months, p = 0.031, log rank). However, there was no significant difference in OS between two groups (p = 0.99, log rank).
Conclusion
Stromal microinvasion is significantly associated with decreased DFS. In addition, our study confirms that the risk of recurrence is higher in patients with microinvasive BOT.
Similar content being viewed by others
References
Hart WR (2005) Borderline epithelial tumors of the ovary. Mod Pathol 18(Suppl 2):S33–S50
Skirnisdottir I et al (2008) Borderline ovarian tumors in Sweden 1960–2005: trends in incidence and age at diagnosis compared to ovarian cancer. Int J Cancer 123(8):1897–1901
Seidman JD, Kurman RJ (2003) Pathology of ovarian carcinoma. Hematol Oncol Clin North Am 17(4):909–925, vii
Tavassoli FA (1988) Serous tumor of low malignant potential with early stromal invasion (serous LMP with microinvasion). Mod Pathol 1(6):407–414
Bell DA, Scully RE (1990) Ovarian serous borderline tumors with stromal microinvasion: a report of 21 cases. Hum Pathol 21(4):397–403
Tinelli R et al (2006) Conservative surgery for borderline ovarian tumors: a review. Gynecol Oncol 100(1):185–191
Kaern J, Trope CG, Abeler VM (1993) A retrospective study of 370 borderline tumors of the ovary treated at the Norwegian Radium Hospital from 1970 to 1982. A review of clinicopathologic features and treatment modalities. Cancer 71(5):1810–1820
Lazarou A et al (2014) Long-term follow-up of borderline ovarian tumors clinical outcome and prognostic factors. Anticancer Res 34(11):6725–6730
McCluggage WG (2010) The pathology of and controversial aspects of ovarian borderline tumours. Curr Opin Oncol 22(5):462–472
Ewald-Riegler N et al (2012) Borderline tumors of the ovary: clinical course and prognostic factors. Onkologie 35(1–2):28–33
Kraus JA, Seidman JD (2010) The relationship between papillary infarction and microinvasion in ovarian atypical proliferative (“borderline”) serous and seromucinous tumors. Int J Gynecol Pathol 29(4):303–309
Ferrero A et al (2012) Clinical significance of microinvasion in borderline ovarian tumors and its impact on surgical management. Int J Gynecol Cancer 22(7):1158–1162
Longacre TA et al (2005) Ovarian serous tumors of low malignant potential (borderline tumors): outcome-based study of 276 patients with long-term (> or = 5-year) follow-up. Am J Surg Pathol 29(6):707–723
Prat J, De Nictolis M (2002) Serous borderline tumors of the ovary: a long-term follow-up study of 137 cases, including 18 with a micropapillary pattern and 20 with microinvasion. Am J Surg Pathol 26(9):1111–1128
Kennedy AW, Hart WR (1996) Ovarian papillary serous tumors of low malignant potential (serous borderline tumors). A long-term follow-up study, including patients with microinvasion, lymph node metastasis, and transformation to invasive serous carcinoma. Cancer 78(2):278–286
Katzenstein AL et al (1978) Proliferative serous tumors of the ovary. Histologic features and prognosis. Am J Surg Pathol 2(4):339–355
Nayar R et al (1996) Microinvasion in low malignant potential tumors of the ovary. Hum Pathol 27(6):521–527
Buttin BM et al (2002) Epithelial ovarian tumors of low malignant potential: the role of microinvasion. Obstet Gynecol 99(1):11–17
Morice P et al (2012) Borderline ovarian tumour: pathological diagnostic dilemma and risk factors for invasive or lethal recurrence. Lancet Oncol 13(3):e103–e115
du Bois A et al (2013) Borderline tumours of the ovary: a cohort study of the Arbeitsgmeinschaft Gynakologische Onkologie (AGO) Study Group. Eur J Cancer 49(8):1905–1914
Nomura K, Aizawa S (2000) Noninvasive, microinvasive, and invasive mucinous carcinomas of the ovary: a clinicopathologic analysis of 40 cases. Cancer 89(7):1541–1546
Silva EG et al (1998) Tumor recurrence in stage I ovarian serous neoplasms of low malignant potential. Int J Gynecol Pathol 17(1):1–6
Harter P et al (2014) Gynecologic Cancer InterGroup (GCIG) consensus review for ovarian tumors of low malignant potential (borderline ovarian tumors). Int J Gynecol Cancer 24(9 Suppl 3):S5–S8
Seidman JD, Kurman RJ (2000) Ovarian serous borderline tumors: a critical review of the literature with emphasis on prognostic indicators. Hum Pathol 31(5):539–557
Leake JF et al (1991) Retroperitoneal lymphatic involvement with epithelial ovarian tumors of low malignant potential. Gynecol Oncol 42(2):124–130
Kanat-Pektas M et al (2011) Complete lymph node dissection: is it essential for the treatment of borderline epithelial ovarian tumors? Arch Gynecol Obstet 283(4):879–884
Hannibal CG et al (2014) A nationwide study of serous “borderline” ovarian tumors in Denmark 1978–2002: centralized pathology review and overall survival compared with the general population. Gynecol Oncol 134(2):267–273
Hendrickson MR, Longacre TA (1993) Classification of surface epithelial neoplasms of the ovary. Pathology (Phila) 1(2):189–254
Hogg R et al (2007) Microinvasion links ovarian serous borderline tumor and grade 1 invasive carcinoma. Gynecol Oncol 106(1):44–51
Laurent I et al (2009) Results after conservative treatment of serous borderline tumours of the ovary with stromal microinvasion but without micropapillary pattern. BJOG 116(6):860–862
Park JY et al (2011) Micropapillary pattern in serous borderline ovarian tumors: does it matter? Gynecol Oncol 123(3):511–516
Hoerl HD, Hart WR (1998) Primary ovarian mucinous cystadenocarcinomas: a clinicopathologic study of 49 cases with long-term follow-up. Am J Surg Pathol 22(12):1449–1462
Riopel MA, Ronnett BM, Kurman RJ (1999) Evaluation of diagnostic criteria and behavior of ovarian intestinal-type mucinous tumors: atypical proliferative (borderline) tumors and intraepithelial, microinvasive, invasive, and metastatic carcinomas. Am J Surg Pathol 23(6):617–635
Siriaunkgul S et al (1995) Ovarian mucinous tumors of low malignant potential: a clinicopathologic study of 54 tumors of intestinal and mullerian type. Int J Gynecol Pathol 14(3):198–208
Author information
Authors and Affiliations
Contributions
GB: project development, data collection, and manuscript writing; MCS: project development, data management, and manuscript writing; MG: manuscript writing, manuscript editing; NTO: data collection, and manuscript writing; HU: data collection; KG: manuscript writing and manuscript editing; VG: data collection; HA: data collection; TT: manuscript writing and data management; OK: manuscript writing and data management; HS: manuscript writing and data management; AE: data collection; NO: project development, data management, and manuscript writing; MMM: project development, data management, and manuscript writing; MS: manuscript writing and manuscript editing; TT: manuscript writing and manuscript editing; TG: manuscript writing and manuscript editing; TS: project development, data collection, and manuscript writing; ST: manuscript writing and manuscript editing; KY: project development, data collection, and manuscript writing
Corresponding author
Ethics declarations
Conflict of interest
There are no potential conflicts of interest to declare and no relevant sources of funding for this study.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Boyraz, G., Salman, M.C., Gultekin, M. et al. What is the impact of stromal microinvasion on oncologic outcomes in borderline ovarian tumors? A multicenter case–control study. Arch Gynecol Obstet 296, 979–987 (2017). https://doi.org/10.1007/s00404-017-4496-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-017-4496-4