Abstract
Background
Pertuzumab is a humanized mAb that binds to the extracellular region of HER2/ErbB2 and is approved for treating breast cancer. Although ovarian cancer and breast cancer have comparable levels of HER2/ErbB2 expression, clinical studies of pertuzumab in epithelial ovarian cancer patients have not met the same level of success.
Objectives
To investigate the molecular mechanisms by which pertuzumab exerts its anti-tumor effects in ovarian cancer and the mechanisms by which cancer cells achieve pertuzumab resistance.
Methods
We examined expression of miR-150 in ovarian cancer cells treated with pertuzumab or not. miR-150 knockdown impacts on pertuzumab treatment were analyzed by cell proliferation assay, apoptosis analysis and cell cycle analysis. Cell signal pathway was examined by western blot assay.
Results
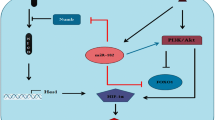
Pertuzumab induced miRNA-150 expression in SKOV3 and SNU119 cells. Furthermore, suppression of miRNA-150 in both cell lines resulted in decreased drug sensitivity to pertuzumab and cell apoptosis. The blockage of G1/S checkpoint by pertuzumab was rescued as well. miRNA-150 knockdown activated PI3K-Akt pathway and LY294002 reversed the effect of miR-150 knockdown.
Conclusions
miRNA-150 downregulation may contribute to the pertuzumab resistance in ovarian cancer via, at least in part, PI3K-akt pathway.






Similar content being viewed by others
References
Siegel RL, Miller KD (2015) Jemal A (2015) Cancer statistics. CA Cancer J Clin 65(1):5–29. doi:10.3322/caac.21254
Lengyel E (2010) Ovarian cancer development and metastasis. Am J Pathol 177(3):1053–1064. doi:10.2353/ajpath.2010.100105
Yap TA, Carden CP, Kaye SB (2009) Beyond chemotherapy: targeted therapies in ovarian cancer. Nat Rev Cancer 9(3):167–181. doi:10.1038/nrc2583
Tuefferd M, Couturier J, Penault-Llorca F, Vincent-Salomon A, Broet P, Guastalla JP, Allouache D, Combe M, Weber B, Pujade-Lauraine E, Camilleri-Broet S (2007) HER2 status in ovarian carcinomas: a multicenter GINECO study of 320 patients. PLoS One 2(11):e1138. doi:10.1371/journal.pone.0001138
Baselga J, Swain SM (2009) Novel anticancer targets: revisiting ERBB2 and discovering ERBB3. Nat Rev Cancer 9(7):463–475. doi:10.1038/nrc2656
Langdon SP, Faratian D, Nagumo Y, Mullen P, Harrison DJ (2010) Pertuzumab for the treatment of ovarian cancer. Exp Opin Biol Ther 10(7):1113–1120. doi:10.1517/14712598.2010.487062
Metzger-Filho O, Winer EP, Krop I (2013) Pertuzumab: optimizing HER2 blockade. Clin Cancer Res 19(20):5552–5556. doi:10.1158/1078-0432.ccr-13-0518
He L, Hannon GJ (2004) MicroRNAs: small RNAs with a big role in gene regulation. Nat Rev Genet 5(7):522–531. doi:10.1038/nrg1379
Calin GA, Croce CM (2006) MicroRNA signatures in human cancers. Nat Rev Cancer 6(11):857–866. doi:10.1038/nrc1997
He Y, Jiang X, Chen J (2014) The role of miR-150 in normal and malignant hematopoiesis. Oncogene 33(30):3887–3893. doi:10.1038/onc.2013.346
Warth SC, Hoefig KP, Hiekel A, Schallenberg S, Jovanovic K, Klein L, Kretschmer K, Ansel KM, Heissmeyer V (2015) Induced miR-99a expression represses Mtor cooperatively with miR-150 to promote regulatory T-cell differentiation. EMBO J. doi:10.15252/embj.201489589
Jin M, Yang Z, Ye W, Xu H, Hua X (2014) MicroRNA-150 predicts a favorable prognosis in patients with epithelial ovarian cancer, and inhibits cell invasion and metastasis by suppressing transcriptional repressor ZEB1. PLoS One 9(8):e103965. doi:10.1371/journal.pone.0103965
Mraz M, Chen L, Rassenti LZ, Ghia EM, Li H, Jepsen K, Smith EN, Messer K, Frazer KA, Kipps TJ (2014) miR-150 influences B-cell receptor signaling in chronic lymphocytic leukemia by regulating expression of GAB1 and FOXP1. Blood 124(1):84–95. doi:10.1182/blood-2013-09-527234
Valabrega G, Montemurro F, Aglietta M (2007) Trastuzumab: mechanism of action, resistance and future perspectives in HER2-overexpressing breast cancer. Ann Oncol 18(6):977–984. doi:10.1093/annonc/mdl475
Faratian D, Zweemer AJM, Nagumo Y, Sims AH, Muir M, Dodds M, Mullen P, Um I, Kay C, Hasmann M, Harrison DJ, Langdon SP (2011) Trastuzumab and pertuzumab produce changes in morphology and estrogen receptor signaling in ovarian cancer xenografts revealing new treatment strategies. Clin Cancer Res 17(13):4451–4461. doi:10.1158/1078-0432.ccr-10-2461
Nakajima H, Mizuta N, Sakaguchi K, Fujiwara I, Yoshimori A, Magae J, Tanuma S (2010) Enhancement of paclitaxel-induced apoptosis in HER2-overexpressing human breast cancer cells by a pertuzumab mimetic peptide,HRAP. J Biosci Bioeng 110(2):250–253. doi:10.1016/j.jbiosc.2010.02.007
Yao E, Zhou W, Lee-Hoeflich ST, Truong T, Haverty PM, Eastham-Anderson J, Lewin-Koh N, Gunter B, Belvin M, Murray LJ, Friedman LS, Sliwkowski MX, Hoeflich KP (2009) Suppression of HER2/HER3-mediated growth of breast cancer cells with combinations of GDC-0941 PI3K inhibitor, trastuzumab, and pertuzumab. Clin Cancer Res 15(12):4147–4156. doi:10.1158/1078-0432.ccr-08-2814
Gajria D, Chandarlapaty S (2011) HER2-amplified breast cancer: mechanisms of trastuzumab resistance and novel targeted therapies. Expert Rev Anticancer Ther 11(2):263–275. doi:10.1586/era.10.226
Mullen P, Cameron DA, Hasmann M, Smyth JF, Langdon SP (2007) Sensitivity to pertuzumab (2C4) in ovarian cancer models: cross-talk with estrogen receptor signaling. Mol Cancer Ther 6(1):93–100. doi:10.1158/1535-7163.mct-06-0401
Vivanco I, Sawyers CL (2002) The phosphatidylinositol 3-kinase AKT pathway in human cancer. Nat Rev Cancer 2(7):489–501. doi:10.1038/nrc839
Greer EL, Brunet A (2005) FOXO transcription factors at the interface between longevity and tumor suppression. Oncogene 24(50):7410–7425. doi:10.1038/sj.onc.1209086
Wu Y, Shang X, Sarkissyan M, Slamon D, Vadgama JV (2010) FOXO1A is a target for HER2-overexpressing breast tumors. Cancer Res 70(13):5475–5485. doi:10.1158/0008-5472.can-10-0176
Nahta R, Esteva FJ (2006) Herceptin: mechanisms of action and resistance. Cancer Lett 232(2):123–138. doi:10.1016/j.canlet.2005.01.041
Moasser MM (2007) The oncogene HER2: its signaling and transforming functions and its role in human cancer pathogenesis. Oncogene 26(45):6469–6487. doi:10.1038/sj.onc.1210477
Davis R, Singh KP, Kurzrock R, Shankar S (2009) Sulforaphane inhibits angiogenesis through activation of FOXO transcription factors. Oncol Rep 22(6):1473–1478. doi:10.3892/or_00000589
Han J, Pan XY, Xu Y, Xiao Y, An Y, Tie L, Pan Y, Li XJ (2012) Curcumin induces autophagy to protect vascular endothelial cell survival from oxidative stress damage. Autophagy 8(5):812–825. doi:10.4161/auto.19471
Zhou J, Liao W, Yang J, Ma K, Li X, Wang Y, Wang D, Wang L, Zhang Y, Yin Y, Zhao Y, Zhu WG (2012) FOXO3 induces FOXO1-dependent autophagy by activating the AKT1 signaling pathway. Autophagy 8(12):1712–1723. doi:10.4161/auto.21830
Engelman JA (2009) Targeting PI3K signalling in cancer: opportunities, challenges and limitations. Nat Rev Cancer 9(8):550–562. doi:10.1038/nrc2664
Fruman DA, Rommel C (2014) PI3K and cancer: lessons, challenges and opportunities. Nat Rev Drug Discovery 13(2):140–156. doi:10.1038/nrd4204
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
D. Wuerkenbieke and J. Wang contributed equally.
Rights and permissions
About this article
Cite this article
Wuerkenbieke, D., Wang, J., Li, Y. et al. miRNA-150 downregulation promotes pertuzumab resistance in ovarian cancer cells via AKT activation. Arch Gynecol Obstet 292, 1109–1116 (2015). https://doi.org/10.1007/s00404-015-3742-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-015-3742-x