Abstract
Purpose
To study the prevalence of non-detectable chromosomal abnormalities by quantitative fluorescent polymerase chain reaction (QF-PCR) in a Chinese population referred for amniocentesis.
Methods
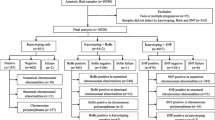
The karyotype results were reviewed in 8,466 amniotic fluid cultures performed for positive fetal Down syndrome screening or advanced maternal age between January 2002 and June 2012. The karyotype results were classified as detectable or not detectable by QF-PCR, using the assumption that all tests were conducted by this rapid molecular method.
Results
Of the 8,466 karyotypes obtained, 211 abnormal karyotypes were found (2.5 %). Out of these, 168 cases of common aneuploidies were identified by QF-PCR, and 43 cases of chromosomal abnormalities were missed. The 43 cases missed by QF-PCR included 31 cases predicted to confer no increased risk and 12 with a potential clinical significance. When QF-PCR shows a normal result, the overall residual risk is 0.1 % for any clinically significant chromosomal abnormality.
Conclusions
A normal QF-PCR result predicts a very low residual risk for patients who are referred solely for an increased risk of a common trisomy.
Similar content being viewed by others
References
Mann K, Ogilvie CM (2012) QF-PCR: application, overview and review of the literature. Prenat Diagn 32:309–314
Langlois S, Duncan A (2011) Use of a DNA method, QF-PCR, in the prenatal diagnosis of fetal aneuploidies. J Obstet Gynaecol Can 33:955–960
Faas BH, Cirigliano V, Bui TH (2011) Rapid methods for targeted prenatal diagnosis of common chromosome aneuploidies. Semin Fetal Neonatal Med 16:81–87
Shaffer LG, Bui TH (2007) Molecular cytogenetic and rapid aneuploidy detection methods in prenatal diagnosis. Am J Med Genet C Semin Med Genet 145C:87–98
Nicolini U, Lalatta F, Natacci F, Curcio C, Bui TH (2004) The introduction of QF-PCR in prenatal diagnosis of fetal aneuploidies: time for reconsideration. Hum Reprod Update 10:541–548
Leung WC, Lau ET, Lao TT, Tang MH (2003) Can amnio-polymerase chain reaction alone replace conventional cytogenetic study for women with positive biochemical screening for fetal Down syndrome? Obstet Gynecol 101:856–861
Speevak MD, McGowan-Jordan J, Chun K (2011) The detection of chromosome anomalies by QF-PCR and residual risks as compared to G-banded analysis. Prenat Diagn 31:454–458
Comas C, Echevarria M, Carrera M, Serra B (2010) Rapid aneuploidy testing versus traditional karyotyping in amniocentesis for certain referral indications. J Matern Fetal Neonatal Med 23:949–955
Caine A, Maltby AE, Parkin CA et al (2005) Prenatal detection of Down’s syndrome by rapid aneuploidy testing for chromosomes 13, 18, and 21 by FISH or PCR without a full karyotype: a cytogenetic risk assessment. Lancet 366:123–128
Gekas J, van den Berg DG, Durand A et al (2011) Rapid testing versus karyotyping in Down’s syndrome screening: cost-effectiveness and detection of clinically significant chromosome abnormalities. Eur J Hum Genet 19:3–9
Ogilvie CM, Lashwood A, Chitty L, Waters JJ, Scriven PN, Flinter F (2005) The future of prenatal diagnosis: rapid testing or full karyotype? An audit of chromosome abnormalities and pregnancy outcomes for women referred for Down’s syndrome testing. BJOG 112:1369–1375
Hills A, Donaghue C, Waters J et al (2010) QF-PCR as a stand-alone test for prenatal samples: the first 2 years’ experience in the London region. Prenat Diagn 30:509–517
Papoulidis I, Siomou E, Sotiriadis A et al (2012) Dual testing with QF-PCR and karyotype analysis for prenatal diagnosis of chromosomal abnormalities. Evaluation of 13,500 cases with consideration of using QF-PCR as a stand-alone test according to referral indications. Prenat Diagn 32:680–685
Ried T, Landes G, Dackowski W, Klinger K, Ward DC (1992) Multicolor fluorescence in situ hybridization for the simultaneous detection of probe sets for chromosomes 13, 18, 21, X and Y in uncultured amniotic fluid cells. Hum Mol Genet 1:307–313
Leung WC, Lam YH, Wong Y, Lau ET, Tang MHY (2002) The effect of fast reporting by amnio-PCR on anxiety levels in women with positive biochemical screening for Down syndrome: a randomized controlled trial. Prenat Diagn 22:256–259
Leung WC, Lau ET, Ngai C et al (2008) A prospective study on the effect of rapid aneuploidy testing (amnio-PCR) on anxiety levels and quality of life measures in women and their partners with positive Down screening result. Fetal Diagn Ther 24:165–169
Lam YH, Tang MH, Sin SY, Ghosh A (1998) Clinical significance of amniotic-fluid-cell culture failure. Prenat Diagn 18:343–347
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liao, C., Yi, CX., Li, FT. et al. The prevalence of non-detectable chromosomal abnormalities by QF-PCR in amniocentesis for certain referral indications: experience at a mainland Chinese hospital. Arch Gynecol Obstet 289, 75–78 (2014). https://doi.org/10.1007/s00404-013-2951-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-013-2951-4