Abstract
Objective
To evaluate the disease control rate (DCR) in heavily pretreated and relapsed ovarian cancer patients re-challenged with a weekly paclitaxel schedule and to establish whether a correlation between dose intensity, progression-free interval (PFI) and overall survival (OS) exists.
Methods
Retrospective data were collected from 30 heavily pretreated metastatic ovarian cancer patients who received 80 mg/m2/week paclitaxel regimen.
Results
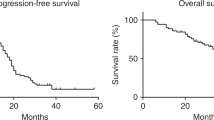
The treatment was well tolerated and showed a DCR in 70% of the patients, with only one case of grade 3 hematological toxicity. One patient (3%) showed a complete response, 15 patients (50%) a partial response and five patients (17%) a stabilization of their disease. The regimen was mostly used as a fourth-line chemotherapy (range 2–7). The median dose intensity in responding patients was 57.5 mg/m2/week and in those with progressive disease 49.7 mg/m2/week. (p = 0.20). PFI and OS were increased in the responder patient groups with a log-rank test of 25.64 (p < 0.001) and 15.10 (p = 0.0001), respectively.
Conclusions
Weekly administration of paclitaxel was active and well tolerated as a salvage therapy for heavily pretreated ovarian cancer patients.


Similar content being viewed by others
References
McGuire WP, Hoskins WJ, Brady MF et al (1996) Cyclophosphamide and cisplatin compared with paclitaxel and cisplatin in patients with stage III and stage IV ovarian cancer. N Engl J Med 334:1–6
Piccart MJ, Bertelsen K, James K et al (2000) Randomized intergroup trial of cisplatin-paclitaxel versus cisplatin cyclophosphamide in women with advanced epithelial ovarian cancer: three-year results. J Natl Cancer Inst 92:699–708
Norton L (2001) Theoretical concepts and the emerging role of taxanes in adjuvant therapy. Oncologist 3(Suppl):30–35
Katsumata N, Yasuda M, Takahashi F et al (2009) Japanese Gynecologic Oncology Group. Dose-dense paclitaxel once a week in combination with carboplatin every 3 weeks for advanced ovarian cancer: a phase 3, open-label, randomised controlled trial. Lancet 374:1331–1338
Thomas H, Rosenberg P (2002) Role of weekly paclitaxel in the treatment of advanced ovarian cancer. Crit Rev Oncol Hematol 44(Suppl):S43–S51
Markman M, Hall J, Spitz D et al (2002) Phase II trial of weekly single-agent Paclitaxel in platinum/Paclitaxel-refractory ovarian cancer. J Clin Oncol 20:2365–2369
Le T, Hopkins L, Baines KA et al (2006) Prospective evaluations of continuous weekly paclitaxel regimen in recurrent platinum-resistant epithelial ovarian cancer. Gynecol Oncol 102:49–53
Kaern J, Baekelandt M, Trope CG (2002) A phase II study of weekly paclitaxel in platinum and Paclitaxel-resistant ovarian cancer patients. Eur J Gynaecol Oncol 23:383–389
Linch M, Stavridi F, Hook J et al (2008) Experience in a UK cancer centre of weekly paclitaxel in the treatment of relapsed ovarian and primary peritoneal cancer. Gynecol Oncol 109:27–32
Ghamande S, Lele S, Marchetti D et al (2003) Weekly paclitaxel in patients with recurrent or persistent advanced ovarian cancer. Int J Gynecol Cancer 13:142–147
Breier S, Lebedinsky C, Pelayes L et al (1997) Phase I/II weekly paclitaxel (P) 80 mg/m2 in pretreated patients (pts) with breast (BC) and ovarian cancer (OC) [abstract]. Proc Am Soc Clin Oncol 16:a568
Thirapakawong C, Senapad S, Pedangsutt P et al (2001) Phase II study of weekly paclitaxel (Taxol) as a second line chemotherapy in refractory epithelial ovarian cancer (EOC): a multicenter study. Pro Am Soc Clin Oncol 20:2506 (abstract)
Fennelly D, Aghajanian C, Shapiro F et al (1997) Phase I and pharmacologic study of paclitaxel administered weekly in patients with relapsed ovarian cancer. J Clin Oncol 15:187–192
Löffler TM, Freund W, Lipke J et al (1996) Schedule- and dose-intensified paclitaxel as weekly 1-hour infusion in pretreated solid tumors: results of a phase I/II trial. Semin Oncol 23:32–34
Klaassen U, Wilke H, Strumberg D et al (1996) Phase I study with a weekly 1 h infusion of paclitaxel in heavily pretreated patients with metastatic breast and ovarian cancer. Eur J Cancer 32:547–549
Dunder I, Berker B, Atabekoglu C et al (2005) Preliminary experience with salvage weekly paclitaxel in women with advanced recurrent ovarian carcinoma. Eur J Gynaecol Oncol 26:79–82
Kita T, Kikuchi Y, Takano M et al (2004) The effect of single weekly paclitaxel in heavily pretreated patients with recurrent or persistent advanced ovarian cancer. Gynecol Oncol 92:813–818
Rosenberg P, Andersson H, Boman K et al (2002) Randomized trial of single agent Paclitaxel given weekly versus every three weeks and with peroral versus intravenous steroid premedication to patients with ovarian cancer previously treated with platinum. Acta Oncol 41:418–424
Molckowsky A, Vijay SM, Hopman WM et al (2008) Decreased dose density of standard chemotherapy does not compromise survival for ovarian cancer patients. Int J Gynecol Cancer 18:8–13
Grant DS, Williams TL, Iliakis G et al (2003) Comparison of antiangiogenic activities using paclitaxel (taxol) and docetaxel (taxotere). Int J Cancer 104:121–129
Pasquier E, Carrè M, Pourroy B et al (2004) Antiangiogenic activity of paclitaxel is associated with its cytostatic effect, mediated by the initiation but not completion of a mitochondrial apoptotic signaling pathway. Mol Cancer Ther 3:1301–1310
Pasquier E, Honore S, Pourroy B et al (2005) Antiangiogenic concentrations of paclitaxel induce an increase in microtubule dynamics in endothelial cells but not in cancer cells. Cancer Res 65:2433–2440
Kerbel RS, Kamen BA (2004) The antiangiogenic basis of metronomic chemotherapy. Nat Rev Cancer 4:423–436
Zhijung X, Kaern J, Davidson B et al (2004) Kallikrein 4 is associated with paclitaxel resistance in ovarian cancer. Ginecol Oncol 94:80–85
Dong Y, Kaushal A, Bui L et al (2001) Human kallikrein 4 (KLK4) is highly expressed in serous ovarian carcinomas. Clin Cancer Res 7:2363–2371
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miolo, G., Bidoli, E., Lombardi, D. et al. Weekly paclitaxel in heavily pretreated ovarian cancer patients: does this treatment still provide further advantages?. Arch Gynecol Obstet 285, 499–503 (2012). https://doi.org/10.1007/s00404-011-1976-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-011-1976-9