Abstract
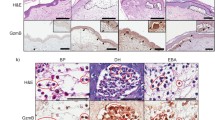
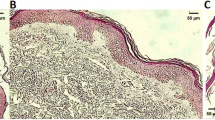
Proteinase activity is increased in psoriatic epidermis. To elucidate the involvement of enzymes in psoriatic epidermis, the expression of cathepsins L, B and D was investigated by Western blotting and immunohistological studies. Normal epidermis contained abundant inactive precursors (39 kDa) of cathepsins L and B and an inactive intermediate form (45 kDa) of cathepsin D. Cathepsin L in psoriasis was processed to a variable extent from the precursor to a single-chain form (30 kDa) and a mixture of single- and heavy-chain (25 kDa) forms of the active mature enzyme, corresponding to the immunohistological staining patterns ‘diffuse dense’, ‘small granular’, and unevenly distributed ‘condensed granular’. Cathepsin B showed a mixture of precursor form (39 kDa) and single-chain (30 kDa) forms and was expressed as a ‘diffuse dense’ staining pattern in the mid-spinous layer and as a ‘condensed’ pattern in the upper spinous and granular layers. Cathepsin D was processed to the heavy-chain (31 kDa) form of activated mature enzyme with small granular staining and a mixture of heavy-chain and degraded protein (28 kDa) with larger and more condensed granular staining. The distribution patterns of ‘small granular’ cathepsin L, and of cathepsins B and D expression in suprabasal keratinocytes were very similar to that of involucrin. After complete clinical resolution of psoriasis by 8-methoxypsoralen plus UVA treatment, the expression of the three cathepsins was normalized. These results suggest that cathepsins L, B and D in forms activated to a variable extent may be involved in the pathology of psoriasis.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 12 March 1996
Rights and permissions
About this article
Cite this article
Kawada, A., Hara, K., Kominami, E. et al. Processing of cathepsins L, B and D in psoriatic epidermis. Arch Dermatol Res 289, 87–93 (1997). https://doi.org/10.1007/s004030050160
Issue Date:
DOI: https://doi.org/10.1007/s004030050160