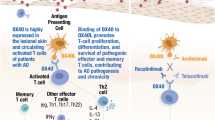
Abstract
Vitiligo is an acquired pigmentary skin disorder that currently lacks standardized treatment and validated biomarkers to objectively evaluate disease state or therapeutic response. Although prior studies have linked vitiligo autoimmunity with CXCL10/CXCL9-mediated recruitment of leukocytes to the skin, only limited clinical data are available regarding CXCL10 as vitiligo biomarker. To evaluate the utility of systemic CXCL10 as a predictor of disease progression and treatment response on a large cohort of vitiligo patients. CXCL10 levels in lesional, perilesional, and unaffected skin of vitiligo patient (n = 30) and in the serum (n = 51) were measured by quantitative ELISA. CXCL10 expression, recruitment of leukocytes, and inflammatory infiltrates were evaluated by histochemical (n = 32) and immunofluorescence (n = 10) staining. Rigorous cross-sectional and longitudinal biostatistical analysis were employed to correlate CXCL10 levels with disease variables, treatment response, and outcome. We demonstrated that elevated CXCL10 level (2 pg/mm2 and higher) in lesional skin correlates with increased leukocytic infiltrate, disease duration (< 2 year), and its higher level in the serum (50 pg/ml and higher). Changes in CXCL10 serum levels in patients treated with psoralen plus UVA (PUVA) phototherapy, narrowband UVB (NB-UVB) phototherapy, and systemic steroids (SS) correlated with changes in the intralesional CXCL10 levels in repigmented skin. NB-UVB and SS regimens provided most consistent CXCL10 mean change, suggesting that these regimens are most effective in harnessing CXCR3-mediated inflammatory response. Serum CXCL10 is a useful vitiligo biomarker, which predicts lesional skin leukocytic infiltration, and vitiligo treatment response and outcome.




Similar content being viewed by others
Notes
Average concentrations are provided in parentheses.
References
Aaronson DS, Horvath CM (2002) A road map for those who don’t know JAK-STAT. Science 296:1653–1655. https://doi.org/10.1126/science.1071545
Abdallah M, El-Mofty M, Anbar T, Rasheed H, Esmat S, Al-Tawdy A, Fawzy MM, Abdel-Halim D, Hegazy R, Gawdat H, Bassiouny D, Ibrahim MA, Sany I, El-Bassiouny M, Khalil M, Abdel-Aziz A, El Maadawi ZM, Mostafa WZ (2018) CXCL-10 and Interleukin-6 are reliable serum markers for vitiligo activity: a multicenter cross-sectional study. Pigment Cell Melanoma Res 31:330–336. https://doi.org/10.1111/pcmr.12667
Alexeev V (2017) CXCL12 as a predictor of vitiligo activity and disease progression. J Invest Dermatol 137:1588–1590. https://doi.org/10.1016/j.jid.2017.03.020
Alhowaish AK, Dietrich N, Onder M, Fritz K (2013) Effectiveness of a 308-nm excimer laser in treatment of vitiligo: a review. Lasers Med Sci 28:1035–1041. https://doi.org/10.1007/s10103-012-1185-1
Anbar TS, Westerhof W, Abdel-Rahman AT, El-Khayyat MA (2006) Evaluation of the effects of NB-UVB in both segmental and non-segmental vitiligo affecting different body sites. Photodermatol Photoimmunol Photomed 22:157–163. https://doi.org/10.1111/j.1600-0781.2006.00222.x
Boniface K, Jacquemin C, Darrigade AS, Dessarthe B, Martins C, Boukhedouni N, Vernisse C, Grasseau A, Thiolat D, Rambert J, Lucchese F, Bertolotti A, Ezzedine K, Taieb A, Seneschal J (2018) Vitiligo Skin Is Imprinted with Resident Memory CD8 T Cells Expressing CXCR3. J Invest Dermatol 138:355–364. https://doi.org/10.1016/j.jid.2017.08.038
de Menezes AF, Oliveira de Carvalho F, Barreto RS, de Santana SB, Shanmugam S, Gurgel RQ, de Souza Araujo AA (2017) Pharmacologic treatment of vitiligo in children and adolescents: a systematic review. Pediatr Dermatol 34:13–24. https://doi.org/10.1111/pde.13024
Driessler F, Venstrom K, Sabat R, Asadullah K, Schottelius AJ (2004) Molecular mechanisms of interleukin-10-mediated inhibition of NF-kappaB activity: a role for p50. Clin Exp Immunol 135:64–73. https://doi.org/10.1111/j.1365-2249.2004.02342.x
Ferrari SM, Fallahi P, Santaguida G, Virili C, Ruffilli I, Ragusa F, Centanni M, Antonelli A (2017) Circulating CXCL10 is increased in non-segmental vitiligo, in presence or absence of autoimmune thyroiditis. Autoimmun Rev 16:946–950. https://doi.org/10.1016/j.autrev.2017.07.006
Halder RM, Walters CS, Johnson BA, Chakrabarti SG, Kenney JA Jr (1986) Aberrations in T lymphocytes and natural killer cells in vitiligo: a flow cytometric study. J Am Acad Dermatol 14:733–737. https://doi.org/10.1016/s0190-9622(86)70085-6
Jenkins MH, Brinckerhoff CE, Mullins DW (2015) CXCR3 signaling in BRAFWT melanoma increases IL-8 expression and tumorigenicity. PLoS ONE 10:e0121140. https://doi.org/10.1371/journal.pone.0121140
Jin WJ, Kim B, Kim D, Park Choo HY, Kim HH, Ha H, Lee ZH (2017) NF-kappaB signaling regulates cell-autonomous regulation of CXCL10 in breast cancer 4T1 cells. Exp Mol Med 49:e295. https://doi.org/10.1038/emm.2016.148
Kim SM, Lee HS, Hann SK (1999) The efficacy of low-dose oral corticosteroids in the treatment of vitiligo patients. Int J Dermatol 38:546–550. https://doi.org/10.1046/j.1365-4362.1999.00623.x
Kundu RV, Mhlaba JM, Rangel SM, Le Poole IC (2018) The convergence theory for vitiligo: a reappraisal. Exp Dermatol. https://doi.org/10.1111/exd.13677
Lacotte S, Brun S, Muller S, Dumortier H (2009) CXCR3, inflammation, and autoimmune diseases. Ann N Y Acad Sci 1173:310–317. https://doi.org/10.1111/j.1749-6632.2009.04813.x
Le Poole C, Boissy RE (1997) Vitiligo. Semin Cutan Med Surg 16:3–14. https://doi.org/10.1016/s1085-5629(97)80030-2
Lee EY, Lee ZH, Song YW (2009) CXCL10 and autoimmune diseases. Autoimmun Rev 8:379–383. https://doi.org/10.1016/j.autrev.2008.12.002
Loetscher M, Loetscher P, Brass N, Meese E, Moser B (1998) Lymphocyte-specific chemokine receptor CXCR3: regulation, chemokine binding and gene localization. Eur J Immunol 28:3696–3705. https://doi.org/10.1002/(sici)1521-4141(199811)28:11%3C3696::aid-immu3696%3E3.0.co;2-w
Lonsdorf AS, Hwang ST, Enk AH (2009) Chemokine receptors in T-cell-mediated diseases of the skin. J Invest Dermatol 129:2552–2566. https://doi.org/10.1038/jid.2009.122
Luster AD, Unkeless JC, Ravetch JV (1985) Gamma-interferon transcriptionally regulates an early-response gene containing homology to platelet proteins. Nature 315:672–676. https://doi.org/10.1038/315672a0
Mofty ME, Zaher H, Esmat S, Youssef R, Shahin Z, Bassioni D, Enani GE (2001) PUVA and PUVB in vitiligo–are they equally effective? Photodermatol Photoimmunol Photomed 17:159–163. https://doi.org/10.1034/j.1600-0781.2001.170403.x
Mohammed GF, Gomaa AH, Al-Dhubaibi MS (2015) Highlights in pathogenesis of vitiligo. World J Clin Cases 3:221–230. https://doi.org/10.12998/wjcc.v3.i3.221
Nordlund JJ (2017) The Medical Treatment of Vitiligo: An Historical Review. Dermatol Clin 35:107–116. https://doi.org/10.1016/j.det.2016.11.001
Patel NS, Paghdal KV, Cohen GF (2012) Advanced treatment modalities for vitiligo. Dermatol Surg 38:381–391. https://doi.org/10.1111/j.1524-4725.2011.02234.x
Pfeffer LM (2011) The role of nuclear factor kappaB in the interferon response. J Interferon Cytokine Res 31:553–559. https://doi.org/10.1089/jir.2011.0028
Radtke MA, Schafer I, Gajur A, Langenbruch A, Augustin M (2009) Willingness-to-pay and quality of life in patients with vitiligo. Br J Dermatol 161:134–139. https://doi.org/10.1111/j.1365-2133.2009.09091.x
Rashighi M, Agarwal P, Richmond JM, Harris TH, Dresser K, Su MW, Zhou Y, Deng A, Hunter CA, Luster AD, Harris JE (2014) CXCL10 is critical for the progression and maintenance of depigmentation in a mouse model of vitiligo. Sci Transl Med 6:223ra223. https://doi.org/10.1126/scitranslmed.3007811
Rezk AF, Kemp DM, El-Domyati M, El-Din WH, Lee JB, Uitto J, Igoucheva O, Alexeev V (2017) Misbalanced CXCL12 and CCL5 chemotactic signals in vitiligo onset and progression. J Invest Dermatol 137:1126–1134. https://doi.org/10.1016/j.jid.2016.12.028
Speeckaert R, Ongenae K, van Geel N (2017) Alterations of CXCL12 in serum of patients with vitiligo. J Invest Dermatol 137:1586–1588. https://doi.org/10.1016/j.jid.2017.02.012
Strassner JP, Rashighi M, Ahmed Refat M, Richmond JM, Harris JE (2017) Suction blistering the lesional skin of vitiligo patients reveals useful biomarkers of disease activity. J Am Acad Dermatol 76(847–855):e845. https://doi.org/10.1016/j.jaad.2016.12.021
Tembhre MK, Sharma VK, Sharma A, Chattopadhyay P, Gupta S (2013) T helper and regulatory T cell cytokine profile in active, stable and narrow band ultraviolet B treated generalized vitiligo. Int J Clin Chem 424:27–32. https://doi.org/10.1016/j.cca.2013.05.005
Tuck MK, Chan DW, Chia D, Godwin AK, Grizzle WE, Krueger KE, Rom W, Sanda M, Sorbara L, Stass S, Wang W, Brenner DE (2009) Standard operating procedures for serum and plasma collection: early detection research network consensus statement standard operating procedure integration working group. J Proteome Res 8:113–117. https://doi.org/10.1021/pr800545q
Van Raemdonck K, Van den Steen PE, Liekens S, Van Damme J, Struyf S (2015) CXCR3 ligands in disease and therapy. Cytokine Growth Factor Rev 26:311–327. https://doi.org/10.1016/j.cytogfr.2014.11.009
Wang XX, Wang QQ, Wu JQ, Jiang M, Chen L, Zhang CF, Xiang LH (2016) Increased expression of CXCR3 and its ligands in patients with vitiligo and CXCL10 as a potential clinical marker for vitiligo. Br J Dermatol 174:1318–1326. https://doi.org/10.1111/bjd.14416
Wenzel J, Wiechert A, Merkel C, Bieber T, Tuting T (2007) IP10/CXCL10 - CXCR3 interaction: a potential self-recruiting mechanism for cytotoxic lymphocytes in lichen sclerosus et atrophicus. Acta Derm Venereol 87:112–117. https://doi.org/10.2340/00015555-0194
Xiao BH, Wu Y, Sun Y, Chen HD, Gao XH (2015) Treatment of vitiligo with NB-UVB: a systematic review. J Dermatolog Treat 26:340–346. https://doi.org/10.3109/09546634.2014.952610
Yamamoto Y, Gaynor RB (2001) Therapeutic potential of inhibition of the NF-kappaB pathway in the treatment of inflammation and cancer. J Clin Investig 107:135–142. https://doi.org/10.1172/jci11914
Acknowledgements
These studies were supported in part by the Cultural and Educational Bureau of the Republic of Egypt and Egyptian Scholar Program for AFR and by the Jefferson Institute of Molecular Medicine, Department of Dermatology and Cutaneous Biology, Thomas Jefferson University for VA.
Funding
This work was supported in part by the Cultural and Educational Bureau of the Republic of Egypt for AFR and by the Jefferson Institute of Molecular Medicine, Thomas Jefferson University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
El-Domyati, M., El-Din, W.H., Rezk, A.F. et al. Systemic CXCL10 is a predictive biomarker of vitiligo lesional skin infiltration, PUVA, NB-UVB and corticosteroid treatment response and outcome. Arch Dermatol Res 314, 275–284 (2022). https://doi.org/10.1007/s00403-021-02228-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00403-021-02228-9