Abstract
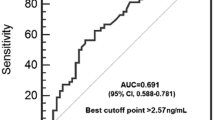
Goeckerman’s therapy (GT) of psoriasis is based on daily application of pharmacy grade coal tar on affected skin with subsequent exposure to UV light. Goeckerman’s therapy is still the first line therapy of psoriasis in the Czech Republic because of its low cost and long-term efficacy. Disturbances in angiogenic activity are characteristic for the immunopathogenesis of psoriasis. An abnormal spectrum of cytokines, growth factors and proangiogenic mediators is produced by keratinocytes and inflammatory cells in patients suffering from the disease. The aim of this study was to evaluate the influence of GT of psoriasis on angiogenic activities by comparing serum levels of vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF) in 44 patients with psoriasis in peripheral blood samples collected before and after therapy. Forty otherwise healthy blood donors serve as a control group. The efficacy of GT was delineated by psoriasis area and severity index (PASI). The disease activity was significantly diminished by GT (P < 0.001). The serum levels of both VEGF and bFGF were statistically significantly correlated to PASI value in patients before the treatment by GT. The serum levels of VEGF (329.4 ± 125.5 pg/ml) and bFGF (10.2 ± 5.04 pg/ml) in patients before GT were significantly higher than those measured in healthy blood donors (VEGF 236.4 ± 55.9 pg/ml, bFGF 7.3 ± 3.7 pg/ml). The serum levels of both VEGF and bFGF were significantly diminished by GT. The level of VEGF dropped from 329.4 ± 125.5 pg/ml before GT to 278.5 ± 109.9 pg/ml after GT (P = 0.0042) and the level of bFGF fell from 10.2 ± 5.04 to 7.78 ± 4.5 pg/ml (P = 0.019). Comparing to healthy controls, the serum level of bFGF in psoriasis patients was normalised (P = 0.5723) after GT. In contrast, the serum level of VEGF remained significantly increased in psoriasis patients after GT in comparison with healthy blood donors (P = 0.0319). In conclusion, we found that the angiogenic potential which is abnormally increased in patients with psoriasis is significantly alleviated by GT.






Similar content being viewed by others
References
Arbiser JL, Govindarajan B, Battle TE, Lynch R, Frank DA, Ushio-Fukai M, Perry BN, Stern DF, Bowden GT, Liu A, Klein E, Kolodziejski PJ, Eissa NT, Hossain CF, Nagle DG (2006) Carbazole is a naturally occurring inhibitor of angiogenesis and inflammation isolated from antipsoriatic coal tar. J Invest Dermatol 126:1396–1402
Beissert S, Cavazzana I, Mascia F, Meroni P, Pastore S, Tessari G, Girolomoni G (2006) Mechanisms of immune-mediated skin diseases: an overview. Clin Exp Rheumatol 24(1Suppl 40):S1–S6
Bhushan M, McLaughlin B, Weiss JB, Griffiths CEM (1999) Levels of endothelial cell stimulating angiogenesis factor and vascular endothelial growth factor are elevated in psoriasis. Br J Dermatol 141:1054–1060
Bhushan M, Young HS, Brenchley PEC, Griffiths CEM (2002) Recent advances in cutaneous angiogenesis. Br J Dermatol 147:418–425
Bielenberg DR, Bucana CD, Sanchez R, Donawho CHK, Kripke ML, Fidler IJ (1998) Molecular regulation of UVB-induced cutaneous angiogenesis. J Invest Dermatol 111:864–872
Bovenschen HJ, Seyger MM, van de Kerkhof PC (2005) Plaque psoriasis vs. atopic dermatitis and lichen planus: a comparison for lesional T-cell subsets, epidermal proliferation and differentiantion. Br J Dermatol 153:72–78
Chen CH, Poucher SM, Lu J, Henry PD (2004) Fibroblast growth factor 2: from laboratory evidence to clinical application. Curr Vasc Pharmacol 2:33–43
Creamer D, Sullivan D, Bicknell R, Barker J (2002) Angiogenesis in psoriasis. Angiogenesis 5:231–236
Cuevas P, Arrazola JM (2005) Dobesilate in the treatment of plaque psoriasis. Eur J Med Res 10:373–376
De Rie MA, Goedkoop AY, Bos JD (2004) Overview of psoriasis. Dermatol Ther 17:341–349
Detmar M (2000) Tumor angiogenesis. J Investig Dermatol Symp Proc 5:20–23
Distler JH, Hirth A, Kurowska-Stolarska M, Gay RE, Gay S, Distler O (2003) Angiogenic and angiostatic factors in the molecular control of angiogenesis. Q J Nucl Med 47:149–161
Feldman SR (2005) Where has Goeckerman treatment gone? J Dermatolog Treat 16:73–74
Ferrara N (1999) Role of vascular endothelial growth factor in the regulation of angiogenesis. Kidney Int 56:794–814
Folkman J (2006) Angiogenesis. Annu Rev Med 57:1–18
Goeckerman WH (1925) Treatment of psoriasis. Northwest Med 24:229–231
Gupta AK, Langley R, Poulin Y, Lui H, Searles G, Carey W, Toole J, Inniss K (2004) Pathogenesis of psoriasis and current challenges. J Cutan Med Surg 8(Suppl):3–7
Kerbel RS (2000) Tumor angiogenesis: past, present and the near future. Carcinogenesis 21:505–515
Lebwohl M, Ali S (2001) Treatment of psoriasis. Part 1. Topical therapy and phototherapy. J Am Acad Dermatol 45:487–498
Lee E, Koo J (2005) Modern modified ‘ultra’ Goeckerman therapy: a PASI assessment of a very effective therapy for psoriasis resistant to both prebiologic and biologic therapies. J Dermatolog Treat 16:102–107
Leong TT, Fearon U, Veale DJ (2005) Angiogenesis in psoriasis and psoriatic arthritis: clues to disease pathogenesis. Curr Rheumatol Rep 7:325–329
Nielsen HJ, Christensen IJ, Svendsen MN, Hansen U, Werther K, Brünner N, Petersen LJ, Kristensen JK (2002) Elevated plasma levels of vascular endothelial growth factor and plasminogen activator inhibitor-1 decrease during improvement of psoriasis. Inflamm Res 51:563–567
Powers CJ, McLeskey SW, Wellstein A (2000) Fibroblast growth factors, their receptors and signaling. Endocr Relat Cancer 7:165–197
Smolej L, Andrys C, Maisnar V, Pour L, Maly J (2005) Plasma concentrations of vascular endothelial growth factor and basic fibroblast growth factor in lymphoproliferative disorders. Acta Medica (Hradec Kralove) 48:57–58
Staton CA, Stribbling SM, Tazzyman S, Hughes R, Brown NJ, Lewis CE (2004) Current methods for assaying angiogenesis in vitro and in vivo. Int J Exp Pathol 85:233–248
Stern RS (1997) Psoriasis. Lancet 350:349–353
Tille JC, Wood J, Mandriota SJ, Schnell C, Ferrari S, Mestan J, Zhu Z, Witte L, Pepper MS (2001) Vascular endothelial growth factor (VEGF) receptor-2 antagonists inhibit VEGF- and basic fibroblast growth factor-induced angiogenesis in vivo and in vitro. J Pharmacol Exp Ther 299:1073–1085
Yano K, Kadoya K, Kajiya K, Hong YK, Detmar M (2005) Ultraviolet B irradiation of human skin induces an angiogenic switch that is mediated by upregulation of vascular endothelial growth factor and by downregulation of thrombospondin-1. Br J Dermatol 152:115–121
Young HS, Summers AM, Bhushan M, Brenchley PE, Griffiths CE (2004) Single-nucleotide polymorphisms of vascular endothelial growth factor in psoriasis of early onset. J Invest Dermatol 122:209–215
Young HS, Summers AM, Read IR, Fairhust DA, Plant DJ, Campalani E, Smith CH, Barker JN, Detmar MJ, Brenchley PE, Griffiths CE (2006) Interaction between genetic control of vascular endothelial growth factor production and retinoid responsiveness in psoriasis. J Invest Dermatol 126:453–459
Zittermann SI, Issekutz AC (2006) Basic fibroblast growth factor (bFGF, FGF-2) potentiates leukocyte recruitment to inflammation by enhancing endothelial adhesion molecule expression. Am J Pathol 168:835–846
Acknowledgments
This study was supported by Ministry of Health Care, Czech Republic, project No. NR8154-3 and Ministry of Education, Czech Republic project No. MZO 00179906.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Andrys, C., Borska, L., Pohl, D. et al. Angiogenic activity in patients with psoriasis is significantly decreased by Goeckerman’s therapy. Arch Dermatol Res 298, 479–483 (2007). https://doi.org/10.1007/s00403-006-0723-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00403-006-0723-8