Abstract
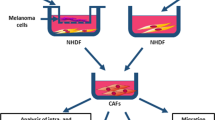
The dermis is the main site of melanoma invasion. Matrix metalloproteinases (MMPs), especially MMP-2, produced by melanoma or surrounding stromal cells, are essential for the destruction of dermal extracellular matrix. Here, we examined how dermal fibroblasts influenced proliferation, MMP-2 secretion and invasion of human melanoma cell lines in vitro. Human melanoma cell lines M3 Da and M1Dor were cocultured with dermal fibroblasts under non-contact and contact conditions in order to assess both soluble and insoluble factors, respectively. Zymographic analysis showed that the levels of MMP-2 and TIMP-2 in melanoma cells were not altered in non-contact cocultures when compared with those in individual cultures. However, in contact cocultures, the expression of MMP-2 in membrane extracts was enhanced. Under our coculture conditions, dermal fibroblasts failed to upregulate melanoma cell invasion through a three-dimensional type I collagen matrix. Since stromal and cancer cell contacts have been shown to occur after disruption of the extracellular matrix, we hypothesized that fibroblasts may influence melanoma cell invasion after the beginning of tumor progression through the dermis.





Similar content being viewed by others
References
Bellows CF, Belafsky P, Fortgang IS, Beech DJ (2001) Melanoma in African-Americans: trends in biological behavior and clinical characteristics over two decades. J Surg Oncol 78:10–16
Benbow U, Schoenermark MP, Mitchell TI, Rutter JL, Shimokawa K, Nagase H, Brinckerhoff CE (1999) A novel host/tumor cell interaction activates matrix metalloproteinase 1 and mediates invasion through type I collagen. J Biol Chem 274:25371–25378
Berthier-Vergnes O, Portoukalian J, Doré JF (1985) Organization and neuraminidase susceptibility of sialic acid residues in human melanoma cell lines with different heterotransplantabilities in nude mice. J Natl Cancer Inst 75:605–611
Berton A, Lorimier S, Emonard H, Laurent-Maquin D, Hornebeck W, Bellon G (2000) Contribution of the plasmin/matrix metalloproteinase cascade to the retraction of human fibroblasts populated collagen lattices. Mol Cell Biol Res Commun 3:173–180
Bogenrieder T, Herlyn M (2002) Cell-surface proteolysis, growth factor activation and intercellular communication in the progression of melanoma. Crit Rev Oncol Hematol 44:1–15
Borchers AH, Sanders LA, Powell MB, Bowden GT (1997) Melanocyte mediated paracrine induction of extracellular matrix degrading proteases in squamous carcinoma cells. Exp Cell Res 231:61–65
Boyd RS, Balkwill FR (1999) MMP-2 release and activation in ovarian carcinoma: the role of fibroblasts. Br J Cancer 80:315–321
Brew K, Dinakarpandian D, Nagase H (2000) Tissue inhibitors of metalloproteinases: evolution, structure and function. Biochim Biophys Acta 1477:267–283
Haas TL, Davis SJ, Madri JA (1998) Three-dimensional type I collagen lattices induce coordinate expression of matrix metalloproteinases MT1-MMP and MMP-2 in microvascular endothelial cells. J Biol Chem 273:3604–3610
Hofmann UB, Westphal JR, van Muijen GNP, Ruiter DJ (2000) Matrix metalloproteinases in human melanoma. J Invest Dermatol 115:337–344
Jacubovich R, Doré JF (1979) Tumor-associated antigens in culture medium of malignant melanoma cell strains. Cancer Immunol Immunother 7:59–64
Jacubovich R, Cabrillat H, Gerlier D, Bailly M, Doré JF (1985) Tumorigenic phenotypes of human melanoma cell lines in nude mice determined by an active antitumor mechanism. Br J Cancer 51:335–345
Kanekura T, Chen X, Kanzaki T (2002) Basigin (CD147) is expressed on melanoma cells and induces tumor cell invasion by stimulating production of metalloproteinases by fibroblasts. Int J Cancer 99:520–528
Kishnani NS, Staskus PW, Yang TT, Masiarz FR, Hawkes SP (1995) Identification and characterization of human tissue inhibitor of metalloproteinase-3 and detection of three additional metalloproteinase activities in extracellular matrix. Matrix Biol 14:479–488
Ko K, Yazumi S, Yoshikawa K, Konda Y, Nakajima M, Chiba T, Takahashi R (2000) Activation of fibroblast-derived matrix metalloproteinase-2 by colon-cancer cells in non-contact co-cultures. Int J Cancer 87:165–171
Kueng W, Silber E, Eppenberger U (1989) Quantification of cells cultured on 96-well plates. Ann Biochem 182:16–19
Kurschat P, Zigrino P, Nischt R, Breitkopf K, Steurer P, Klein CE, Krieg T, Mauch C (1999) Tissue inhibitor of matrix metalloproteinase-2 regulates matrix metalloproteinase-2 activation by modulation of membrane-type 1 matrix metalloproteinase activity in high and low invasive melanoma cell lines. J Biol Chem 274:21056–21062
Kurschat P, Wickenhauser C, Wolfgang G, Krieg T, Mauch C (2002) Identification of activated MMP-2 as the main gelatinolytic enzyme in malignant melanoma by in situ zymography. J Pathol 197:179–187
Lee DY, Lee JH, Lee ES, Cho KH, Yang JM (2003) Fibroblasts play a stimulatory role in keratinocyte proliferation but an inhibitory role in melanocyte growth and pigmentation in skin equivalent system from skin type IV. Arch Dermatol Res 294:444–446
Murphy G, Stanton H, Cowell S, Butler G, Knauper V, Atkinson S, Gavrilovic J (1999) Mechanisms for pro matrix metalloproteinase activation. APMIS 107:38–44
Ntayi C, Lorimier S, Berthier-Vergnes O, Hornebeck W, Bernard P (2001) Cumulative influence of matrix metalloproteinase-1 and -2 in the migration of melanoma cells within three-dimensional type I collagen lattices. Exp Cell Res 270 110–118
Saarialho-Kere UK (1998) Patterns of matrix metalloproteinase and TIMP expression in chronic ulcers. Arch Dermatol Res 290:S47–54
Seltzer JL, Lee AY, Akers KT, Sudbeck B, Southon EA, Wayner EA, Eisen AZ (1994) Activation of 72-kDa type IV collagenase/gelatinase by normal fibroblasts in collagen lattices is mediated by integrin receptors but is not related to lattices contraction. Exp Cell Res 213:365–374
Vaisanen A, Kallioinen M, Taskinen PJ, Turpeenniemi-Hujanen T (1998) Prognostic value of MMP-2 immunoreactive protein (72-kDa type IV collagenase) in primary skin melanoma. J Pathol 186:51–58
Wandel E, Raschke A, Hildebrandt G, Eberle J, Dummer R, Anderegg U, Saalbach A (2002) Fibroblasts enhance invasive capacity of melanoma cells in vitro. Arch Dermatol Res 293:601–608
Yin L, Morita A, Tsuji T (2000) Alterations of extracellular matrix induced by tobacco smoke extract. Arch Dermatol Res 292:188–194
Acknowledgement
C.N. is a grant holder of ARC (Association pour la Recherche sur le Cancer).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ntayi, C., Hornebeck, W. & Bernard, P. Influence of cultured dermal fibroblasts on human melanoma cell proliferation, matrix metalloproteinase-2 (MMP-2) expression and invasion in vitro. Arch Dermatol Res 295, 236–241 (2003). https://doi.org/10.1007/s00403-003-0429-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00403-003-0429-0