Abstract
Introduction
Secondary knee osteoarthritis (OA) is currently associated with meniscal injuries, but the pathogenesis is unclear. We analyzed the distribution of cells and cartilage oligomeric matrix protein (COMP) and its changes in the early stages of degeneration in meniscus.
Method
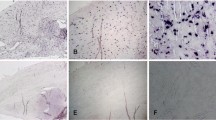
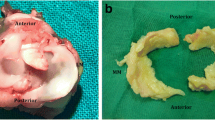
Ten New Zealand rabbits underwent anterior cruciate ligament (ACL)-transection of the right knee-joint. Left knee-joints were used as controls. The animals were killed at 4 and 12 weeks. Gross injuries in meniscus and articular cartilage were scored. Meniscal tissues were immunostained with a specific antibody against COMP, with Ki-67, using TUNEL-assay and alcian blue stain. The number of cells was counted.
Results
At 4 weeks post-ACL-transection, 2/5 of the operated knees showed articular damages and medial menisci tears. Menisci showed a weak increase of cells, higher in cells under division and an increase of apoptosis, COMP and proteoglycans.
At 12 weeks, 5/5 of the medial menisci and 2/5 of lateral menisci presented tears, and osteoarthritic changes were seen in the cartilage of all the operated knees. Meniscal cells reverted to normal number, while active cell division decreased below normal, apoptotic events were still high, COMP remained elevated, and glycosaminoglycans were even more elevated.
Conclusion
Extracellular matrix changes and altered cell distribution occur early in the degenerative meniscus. There is a close relationship between changes in the articular cartilage and the menisci at the onset of secondary OA.





Similar content being viewed by others
References
Kuettner KE, Goldbeg VM (1995) Introduction. In: Kuettner KE, Goldbeg VM (eds) Osteoarthritic disorders. American Academy of Orthopedic Surgeons, Rosemont, pp xxi–xxv
Hauser N, Paulsson M, Kale AA, Di Cesare PE (1995) Tendon extracellular matrix contains pentameric thrombospondin-4 (TSP-4). FEBS Lett 368:307–310
Rosenberg K, Olsson H, Mörgelin M, Heinegård D (1998) Cartilage oligomeric matrix protein shows high affinity zinc-dependent interaction with triple helical collagen. J Biol Chem 273:20397–20403
Di Cesare PE, Fang C, Leslie MP et al (1999) Localization and expression of cartilage oligomeric matrix protein by human rheumatoid and osteoarthritic synovium and cartilage. J Orthop Res 17:437–445
Holden P, Meadows RS, Chapman KL, Grant ME, Kadler KE, Briggs MD (2001) Cartilage oligomeric matrix protein interacts with type IX collagen, and disruptions to these interactions identify a pathogenetic mechanism in a bone dysplasia family. J Biol Chem 276:6046–6055
Hedbom E, Antonsson P, Hjerpe A et al (1992) Cartilage matrix proteins. An acidic oligomeric protein (COMP) detected only in cartilage. J Biol Chem 267:6132–6136
DiCesare PE, Mörgelin M, Mann K, Paulsson M (1994) Cartilage oligomeric matrix protein and thrombospondin 1. Purification from articular cartilage, electron microscopic structure, and chondrocyte binding. Eur J Biochem 223:927–937
Thur J, Rosenberg K, Nitsche DP et al (2001) Mutations in cartilage oligomeric matrix protein causing pseudoachondroplasis and multiple epiphyseal displasia affect binding of calcium and collagen I, II, and IX. J Biol Chem 276:6083–6092
Guo Y, Bozic D, Malashkevich VN, Kammerer RA, Schulthess T, Engel J (1998) All-transretinol, vitamin D and other hydrophobic compounds bind to the axial pore of the five-stranded coiled-coil domain of cartilage oligomeric matrix protein. EMBO J 17:5265–5272
Chen H, Deere M, Hecht JT, Lawler J (2000) Cartilage oligomeric matrix protein is a calcium-binding protein, and a mutation in its type 3 repeats causes conformational changes. J Biol Chem 275:26538–26544
Forslind K, Eberhardt K, Jonsson A, Saxne T (1992) Increased serum concentrations of cartilage oligomeric matrix protein. A prognostic marker in early rheumatoid arthritis. Br J Rheumatol 31:593–598
Sharif M, Saxne T, Shepstone L et al (1995) Relationship between serum cartilage oligomeric matrix protein levels and disease progression in osteoarthritis of the knee joint. Br J Rheumatol 34:306–310
Neidhart M, Hauser N, Paulsson M, Di Cesare PE, Michel BA, Häuselmann HJ (1997) Small fragments of cartilage oligomeric matrix protein in synovial fluid and serum as markers for cartilage degradation. Br J Rheumatol 36:1151–1160
Petersson IF, Boegård T, Dahlström J, Svennson B, Heinegård D, Saxne T (1998) Bone scan and serum markers of bone and cartilage in patients with knee pain and osteoarthritis. Osteoarthritis Cartilage 6:33–39
Petersson IF, Boegård TB, Svensson D, Heinegård D, Saxne T (1998) Changes in cartilage and bone metabolism identified by serum markers in early osteoarthritis of the knee joint. Br J Rheumatol 37:46–50
Otterness IG, Swindell AC, Zimmerer RO, Poole AR, Ionescu M, Weiner E (2000) An analysis of 14 molecular markers for monitoring osteoarthritis: segregation of the markers into clusters and distinguishing osteoarthritis at baseline. Osteoarthritis Cartilage 8:180–185
Vilím V, Olejárová M, Macháček S, Gatterová J, Kraus VB, Pavelka K (2002) Serum levels of cartilage oligomeric matrix protein (COMP) correlate with radiographic progression of knee osteoarthritis. Osteoarthritis Cartilage 10:707–713
Wisłowska M, Jabłońska B (2005) Cartilage oligomeric matrix protein in serum in systemic lupus erythematosus and knee osteoarthritis. Preliminary communication. Rheumatol Int 25:373–378
Gómez-Barrena E, Lindroos L, Čeponis A et al (2006) Cartilage oligomeric matrix protein (COMP) is modified by intra-articular liposomal clodronate in an experimental model of arthritis. Clin Exp Rheumatol 24(6):622–628
Rimoin DL, Rasmussen IM, Briggs MD et al (1994) A large family with features of pseudoachondroplasia and multiple epiphyseal dysplasia: exclusion of seven candidate gene loci that encode proteins of the cartilage extracellular matrix. Hum Genet 93:236–242
Stevens JW (1999) Pseudoachondroplastic dysplasia: an Iowa review from human to mouse. Iowa Orthop J 19:53–65
Kennedy J, Jackson G, Ramsden S et al (2005) COMP mutation screening as an aid for the clinical diagnosis and counselling of patients with a suspected diagnosis of pseudoachondroplasia or multiple epiphyseal dysplasia. Eur J Hum Genet 13:547–555
Yoshioka M, Coutts RD, Amiel D, Hacker SA (1996) Characterization of a model of osteoarthritis in the rabbit knee. Osteoarthritis Cartilage 4(2):87–98
Meachim G (1972) Light microscopy of Indian ink preparations of fibrillated cartilage. Ann Rheum Dis 31(6):457–464
Adams ME, Billingham ME, Muir H (1983) The glycosaminoglycans in menisci in experimental and natural osteoarthritis. Arthritis Rheum 26:69–76
Herrero-Beaumont G, Guerrero R, Sánchez-Pernaute O et al (2001) Cartilage and bone biological markers in the synovial fluid of osteoarthritic patients after hyaluronan injections in the knee. Clin Chim Acta 308:107–115
Moon MS, Kim JM, Ok IY (1984) The normal and regenerated meniscus in rabbits. Morphologic and histologic studies. Clin Orthop Relat Res 182:264–269
Hellio Le Graverand MP, Vignon E, Otterness IG, Hart DA (2001) Early changes in lapine menisci during osteoarthritis development (Part I): cellular and matrix alterations. Osteoarthritis Cartilage 9:56–64
Hellio Le Graverand MP, Vignon E, Otterness IG, Hart DA (2001) Early changes in lapine menisci during osteoarthritis development (Part II): molecular alterations. Osteoarthritis Cartilage 9:65–72
Ochi M, Kanda T, Sumen Y, Ikuta Y (1997) Changes in the permeability and histologic findings of rabbit menisci after immobilization. Clin Orthop Relat Res 334:305–315
Nishida M, Higuchi H, Kobayashi Y, Takagishi K (2005) Histological and biochemical changes of experimental meniscus tear in the dog knee. J Orthop Sci 10:406–413
Hellio Le Graverand MP, Sciore P, Eggerer J, Rattner JP, Vignon E, Barclay L et al (2001) Formation and phenotype of cell clusters in osteoarthritic meniscus. Arthritis Rheum 44(8):1808–1818
Hashimoto S, Takahashi K, Ochs RL, Coutts RD, Amiel D, Lotz M (1999) Nitric oxide production and apoptosis in cells of the meniscus during experimental osteoarthritis. Arthritis Rheum 42:2123–2131
Herwig J, Egner E, Buddecke E (1984) Chemical changes of human knee joint menisci in various stages of degeneration. Ann Rheum Dis 43:635–640
Buckwalter JA, Mankin HJ (1997) Instructional course lecture. The American academy of orthopaedic surgeons—articular cartilage. Part II: degeneration and osteoarthrosis, repair, regeneration and transplantation. J Bone Joint Surg A 79:612–632
Nguyen BQ, Fife RS (1986) Vitreous contains a cartilage related protein. Exp Eye Res 43:375–382
Riessen R, Fenchel M, Chen H, Axel DI, Karsch KR, Lawler J (2001) Cartilage oligomeric matrix protein (thrombospondin-5) is expressed by human vascular smooth muscle cells. Arterioscler Thromb Vasc Biol 21:47–54
Xiao Y, Kleeff J, Guo J et al (2004) Cartilage oligomeric matrix protein expression in hepatocellular carcinoma and the cirrhotic liver. J Gastroenterol Hepatol 19:296–302
Gagarina V, Carlberg AL, Pereira-Mouries L, Hall DJ (2008) Cartilage oligomeric matrix protein protects cells against death by elevating members of the IAP family of survival proteins. J Biol Chem 283:648–659
Hashimoto Y, Tomiyama T, Yamano Y, Mori H (2003) Mutation (D472Y) in the type 3 repeat domain of cartilage oligomeric matrix protein affects its early vesicle trafficking in endoplasmic reticulum and induces apoptosis. Am J Pathol 163:101–110
Oldberg A, Antonsson P, Lindblom K, Heinegård D (1992) COMP (cartilage oligomeric matrix protein) is structurally related to the thrombospondins. J Biol Chem 267:22346–22350
Di Cesare PE, Carlson CS, Stollerman ES, Chen FS, Leslie M, Perris R (1997) Expression of cartilage oligomeric matrix protein by human synovium. FEBS Lett 412:249–252
Fang C, Johnson D, Leslie MP, Carlson CS, Robbins M, Di Cesare PE (2001) Tissue distribution and measurement of cartilage oligomeric matrix protein in patients with magnetic resonance imaging detected bone bruises after acute anterior cruciate ligament tears. J Orthop Res 19:634–641
Acknowledgments
The authors thank Dr. Carmen Gómez-Guerrero and Guadalupe Ortiz-Muñoz for the excellent technical assistance in histological aspects of the study. The research complied with national legislation and with the National Institute of Health Guide for the Care and Use of Laboratory Animals.
Conflict of interest
The authors did not receive payments or other benefits or a commitment or agreement to provide such benefits from a commercial entity.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
López-Franco, M., López-Franco, O., Murciano-Antón, M.A. et al. An experimental study of COMP (cartilage oligomeric matrix protein) in the rabbit menisci. Arch Orthop Trauma Surg 131, 1167–1176 (2011). https://doi.org/10.1007/s00402-011-1332-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-011-1332-3